ISSN: 0973-7510
E-ISSN: 2581-690X
The devastating yield losses caused by rice blast can be mitigated by breeding resistant cultivars with QTLs enriched with resistance (R-gene) and defence related genes. These genes are the arsenal of the plant’s immune system against pathogen invasion. qBFR4 and qLBL5, are two stable QTLs known to contribute moderate level of resistance against blast disease. In this study we aim to characterize and understand the interconnectivity between the genes within these QTLs by producing a defence model based on the interplay of defence and resistance genes found within these QTLs. To achieve this end we identified defence and R-genes in qBFR4 and qLBL5 into functional groups and classes of R-genes, and classified their roles in mounting a defence against pathogens. Blast2GO analysis retrieved the description, gene ontology annotations and domains for 361 genes in qBFR4 and 617 genes in qLBL5. With this, it is concluded that qBFR4 (2.38 Mbps) has 27 R-genes (7.33%) and 14 (0.04%) defence- genes whereas qLBL5 (4.1 Mbps) has 25 R-genes (3.88%) and 17 defence genes (0.03%). R-genes and defence genes were classified into domains and functional groups respectively and directed acyclic graphs (DAG) were constructed for both QTLs explaining the role of these QTLs in quantitative resistance against rice blast. In conclusion, qBRF4 is was found to be more beneficial than qLBL5 based on the defence and resistance gene composition where SA/JA mediated signalling plays a crucial role in signal tranduction between R genes and the defence system. Apart from QTL pyramiding using the QTLs in this study, the major R-genes found within these QTLs can be subjected to cloning to develop resistant cultivars.
QTL, rice blast, Magnaporthe oryzae (M. oryzae), R-gene, defence-related genes
Rice has been an essential component of daily dietary intake as it serves as a primary source of nutrients. With almost 90% of rice being consumed by Asians, the increasing demand for rice is inevitable. However, the sustainable production of rice is often unachievable due to yield loss prompted by various factors such as poor management practices, diseases, drought, and flood. In Malaysia, disease like rice blast, sheath blight and bacterial blight cause devastating yield losses. Rice blast, caused by a pathogenic fungus known as M. oryzae1,2, is the number one disease that causes up to 60% yield loss worldwide. This disease has caused serious constraints in cereal crop production globally and due to its high genetic variability, poses a major challenge to rice breeders and pathologists3. Being a hemibiotophic organism, M. oryzae affects the crop at different growth stages and at different parts including leaf, stem, nodes, panicle and root4, leading to leaf blast, neck and panicle rot, collar rot and node blast 5. Common symptoms for this disease are the formation of diamond shaped lesions on the leaves, white to grey-green lesions or spots, with dark green borders on leaf and collar.
Apart from good management practices and application of fungicides, the development of resistant cultivars can curb this issue effectively. Two types of resistance known as qualitative and quantitative resistance are known to exist in resistant cultivars. Qualitative resistance is controlled by a single resistance gene known as R-gene and are race specific. Plant R-genes can detect the presence of foreign entity like pathogens by interacting with pathogen avirulent gene (Avr) upon attack and thence initiating an appropriate signalling cascades to activate the defence mechanism in the plant6, 7. These R-genes are classified into 8 distinct groups and differ in terms of the domain and repeats found in the N-terminal and C-terminal in the R-gene. Most of the R-genes are found with a nucleotide binding site domains (NBS) 8 and leucine rich repeats (LRR) at the C-terminal and with either a toll-interleukin-1-receptor (TIR) domain or putative coiled coil domain at the N-terminal. These R-genes are being increasingly investigated in the rice genome over the past few years and it is evident that most of it are found in rice based on past studies Several major R genes such as Pib, Pita, Pia, Pi1, Pikh, Pi2 and Pi4 have been successfully introgressed into rice lines through breeding programs9, 10. Some of these genes Pi-1, Pi2, Pi9, Pi20, Pi27, Pi39, Pi40 and Pikh are reported to have confered broad-spectrum resistance (BSR) and some of them including Pia, Pib, Pii, Pi-km, Pi-t, Pi12 and Pi19 confer race specific resistance7. Unfortunately, as the pathogen constantly evolves into a new race, the existing R-genes become obsolete and may not be so reliable which coerce the breeders to continuously mine for new R-genes to breed for durable and resistant cultivars corresponding to the evolution of the pathogen11. Besides, the identification of R-genes with broad spectrum resistance against wide array of pathogen will be an effective measure to eradicate this disease12.
Apart from that, disease resistance is also conferred through quantitative resistance which is controlled by a region known as quantitative trait loci (QTL) controlled by several major and minor genes that work together to provide resistance and defence in response to the attack imposed by the pathogen. For the past few years, several QTLs have been characterized based on experimental study on mapping population13. These QTLs may be enriched with genes involved in signalling, pathways and processes that interconnected to safeguard the plant from the attacks imposed by pathogens.
Two stable QTLs, namely qBFR4 and qLBL5 identified from previous studies were selected for analysis. QTL qBFR4 was identified in a F3 mapping population resulting from a cross between Ingngoppor-tinawon (IT) (resistant variety) and Koshikari (susceptible variety) with a phenotypic variation of 73.5%. The mapped position of qBFR4 shares the similar position to the Pi39-gene. This QTL is situated in a 2.38 Mbps region in chromosome 4 between the marker interval ID04_15 and RM384314. On the other hand, qLBL5 was identified from the F2 population of a cross between Akhanaphou (resistant varieties) and Leimaphou (susceptible varieties) for two consecutive years in two different conditions Rajendranagar and Manipur. with a phenotypic variation of 26.23%15. The mapping of this QTL revealed that it colocalizes with a meta-QTL reported in another study16. This QTL is flanked by the marker interval RM18408 and RM1888215 with an approximate size of 4.1 Mbps in chromosome 5. qLBL5 has demonstrated to confer resistance to leaf blast and neck blast in both types of conditions.
With the establishment of rice genomic map (Rice Genome Browser), it is much easier now to locate individual genes that may be involved in blast resistance based on the markers flanking the QTL region. Although a number of R genes and QTLs have been characterized for the past few years, the underlying mechanism on how these major R-genes work together with other defence-related genes is yet to be well elucidated. The intricate characterization of these genes may help in understanding the defence processes in plants. It also may provide an idea on the signalling network within the plant that initiates the defence processes. These may help in deciphering how defence and resistance mechanism work altogether. Apart from that, discovery of more putative R-genes will be a starting point in mining and characterizing novel R-genes. Hence this study aims (1) to identify and classify defence genes and R-genes in qBFR4 and qLBL5 according to functional groups and classes of R-genes, and (2) to explain the role of each defence gene and R-gene in the plant defence system by producing a defence mechanism model. With the characterization of genes along these QTLs and the generation of sufficient information on the defence processes, breeders can assess the suitability of the QTLs in this study to be selected for appropriate rice breeding programs. In addition, key genes found within these QTLs may be useful in marker assisted selection17.
The identification of the physical position of qBFR4 and qLBL-5
Bioinformatics based analysis are employed in retrieving the results for this study. Two QTLs, qBFR4 and qLBL-5 conferring resistance against rice blast were selected from published papers for analysis as these QTLs were found stable across different years and different environment. The forward and reverse primer sequences of SSR flanking markers of qBFR4 (ID04 _15 and RM3843), and qLBL-5 (RM18638-RM18894) were retrieved from the Gramene database (www.gramene.org/) and subjected to BLASTn in (https://www.ncbi.nlm.nih.gov/BLAST/) with the specification of search set against Oryza sativa (taxid:4530) nucleotide database. Hits with 100% match were selected and the physical positions of the QTLs were determined. The physical positions of qBFR4 (29308303-31683794) and qLBL5 (19724422-23842688) were viewed in Rice Genome Browser (rice.plantbiology.msu.edu/cgi-bin/gbrowse/rice/) and the first and last locus IDs of qBFR4 (LOC_Os04g49160-LOC_Os53210) and qLBL5 (LOC_Os05g33580- LOC_Os05g40650) were determined. The cDNA sequence in the FASTA format starting from the first locus ID until the last locus ID of the QTLs were downloaded from Rice Genome Annotation Project (rice.plantbiology.msu.edu/).
Proximity analysis of Transposable Elements
To explore the possible influence of transposable elements on both QTLs in conferring resistance against pathogen, the genes in proximity with transposable elements were investigated. The transposable element and genes found in the QTL were used as queries to generate an output file with calculated distance. The output is then filtered to retrieve the distances within 5kb. The visualization of transposable elements distribution with genes in proximity is viewed and captured using Rice Annotation Project Database (RAP-DB) Genome Browser (rapdb.dna.affrc.go.jp/)
Bioinformatic analysis using Blast2GO
The files with the FASTA sequences of the qBFR4 and qLBL-5 are then uploaded onto Blast2GO software (https://www.blast2go.com/) separately for further analysis. BLAST was performed on the uploaded sequences to get the descriptional annotations for a total of 453 genes in qBFR4 and 804 genes in qLBL-5 (Blast2GO > blast). These genes are then subjected to mapping and annotation analysis to obtain the gene ontologies in an attempt to identify genes associated to processes or functions that are associated to defence against fungal pathogens (Blast2GO > mapping > annotation). A function known as enzyme coding was utilized to retrieve the enzyme annotation for the genes (Blast2GO > Analysis > Enzyme Code and KEGG > Run GO-EnzymeCode Mapping). Then, domain analysis was conducted for each annotated gene to identify the presence of domains that are prevalent in R-genes (Blast2GO > Run InterProScan). Finally, a directed acyclic graph that explains the interconnecting pathways between the defence processes was mapped (Blast2GO > Graphs > Make GO Graph).
Overview of the gene distribution in qBFR4 and qLBL-5
A total of 368 genes and 644 genes were represented in the 2.38Mbp region of qBR4 and 4.12Mbp region of qLBL5 respectively. These genes are distributed at an average of 155/Mbp for qBFR4 and 156/Mbp for qLBL5 which shows that the density of genes in both QTLs are more or less the same regardless of the size of QTL region. The BLAST analysis via Blast2GO successfully retrieved the description annotations of 361 genes out of 368 genes in qBFR4 (Table S1) and 617 genes out of 644 in qLBL-5 (Table S2) (Supplementary data Table S1 and S2: https://drive.google.com/drive/folders/1ZLdMkzWpUKPffZZ_Y-msl9ZXD_ mSFN9M? usp=sharing).
Based on the results, transposable elements marks the highest percentage in both QTLs with a composition of 54 genes (14.67%) in qBFR4 and 114 genes (17.7%) in qLBL-5. The abundance of TE in these QTLs suggest the possible impact of TE insertion in driving the evolution of genes and possibly even R-gene or defence-related genes to counteract against the evolution of pathogen. Transposable elements (TE) also known as jumping genes, are mobile cluster of genes that move from one location of the genome to another18.
The transposable elements found in the current study were further dissected and two distinct classes of TE were observed. Class I TE known as retrotransposons were found abundantly in both QTLs, with a composition of 42 genes in qBFR4 and 78 genes in qLBL5. Retrotransposons are transposed in a “copy and paste” fashion where the DNA sequences are transcribed to an RNA intermediate which is then subjected to reverse transcription to create an identical DNA sequence and inserted back into the genome at a preferential site19. In contrast to retrotransposons, fewer DNA transposons were found in both QTLs. Having said that, a total of 12 DNA transposons were found in qBFR4 and 36 DNA transposon were found in qLBL5. DNA transposon are from class II TE and they are transposed using a “cut and paste” mechanism by a transposase that cut out the DNA segment from a site and insert it elsewhere in the genome.
TE insertion has a role in altering the regulation of nearby genes that results in differential expression of the genes20. Recently, it has been discovered that TE has a role in altering the genome expression that corresponds to the stress exerted on plants21. Hence, we are interested in investigating the pattern of insertion of this group of genes and how it may impact the genes in proximity specifically to provide resistance against pathogen. It is known that some R-genes constantly evolve to adapt to the rapidly evolving pathogen and perhaps the insertion of TE in a given position near to R-genes, may drive the alteration in the existing R-genes in accordance to the biotic stress posed.
Identification of genes in proximity with transposable elements
The proximity analysis on qBFR4 and qLBL5 retrieved all the genes within 5kb distance from transposable elements (see Supplementary Table S3 for qBFR4 and Supplementary Table S4 for qLBL5: https://drive.google.com/drive/folders/1ZLdMkzWpUKPffZZ_Y-msl9ZXD_ mSFN9M?usp=sharing). The visualization of TE distribution in qBFR4 and qLBL5 is depicted in Figure 1 and Figure 2 respectively. Out of 29 genes that were situated in proximity with TE in qBFR4, two genes (0.07%) were important in plant defence and resistance against pathogen. These genes were NBS-LRR-like and cathepsin B. NBS-LRR is a domain that is often associated to R-gene whereas, cathepsin B were shown to take part in hypersensitive response following the gene-gene interaction between the pathogen Cladosporium fulvum Avr4 and the R-gene in tomato known as Cf-4 22.
Fig. 1. Overview of TE distribution in qBFR4 (red box represent genes within 5kb distance with TE, black box represent TE, line between genes represent tandem repeats of the genes)
Fig. 2. Overview of TE distribution in qBFR4 (red box represent genes within 5kb distance with TE, black box represent TE, line between genes represent tandem repeats of the genes)
A total of 68 genes in qLBL5 were found to be in proximity with TE and out of these only six genes (0.08%) were associated to defence and resistance against disease. This includes inactive receptor kinase, LRR-receptor kinase, serine/threonine-protein kinase, LRR-F-box containing protein and powdery mildew resistance protein 5 (PMR 5). LRRs are ubiquitously known as an essential R-gene domain which is important in interaction between proteins especially during host-pathogen interaction6. Serine threonine kinase and receptor kinase on the other hand acts as receptors in transducing the signals after pathogen elicitor recognition for downstream defence mechanism23. Apart from that, F-box proteins in Arabidopsis were demonstrated in regulating a novel defence response that is independent of both salicylic acid and systemic acquired resistance24. Two PMR 5 genes found in proximity with TE were shown to demonstrate resistance against Erysiphe cichoracearum through the activation of a novel form of defence25.
From past studies, the existence of TE have been indicated in the cluster of disease genes in several plants including rice and shown to be induced by plant pathogens. Wang 2010 have successfully demonstrated the decreased level of resistance against the downy mildew disease after conducting the gene-knockout procedure on a retrotransposon found in Arabidopsis known as AtCOPIA4 26. In addition, RTE Tnt1A inserted in a tobacco resistance gene cluster has shown to drive partial transcription of the neighbouring disease resistance gene TNLL127. Hence, it can be hypothesized that the insertion of TE in qBR4 and qLBL5 may affect the neighbouring defence related genes or R-genes. Although the impact of TE insertion on the neighbouring defence/R-genes in the current study remains unknown, the findings may provide a basis to conduct a loss of function or gene knockout/silencing studies in future to validate whether the existence of TE among the putative R-genes or defence-related genes affects the resistance against pathogens in plants.
Identification and classification of disease resistance genes
The interaction between host plant and pathogen involves the recognition of pathogen effectors known as avirulence genes (Avr) by R-genes in host plants which in turn activates the weaponry of the plant defence mechanism28. This interaction is well represented in rice, where the Avr-Pita gene of M. grisea binds to the Pita gene, a well-characterised R-gene in rice for the subsequent activation of defence processes29. Fundamentally, R-genes are classified into 8 major classes based on the arrangement of domains mainly consisting NBS, LRR and kinases28. The results retrieved from InterProScan is analysed to identify genes with possible domains or motifs found in the different classes of R-genes. The results and discussion for this is provided in the following subsection.
Disease resistance genes in qBFR4
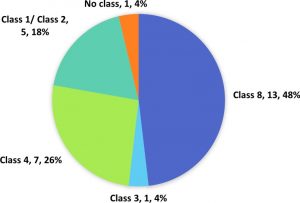
The output of the analysis unveiled 27 (7.33%) genes in qBFR4 with domains related to R-genes which has been classified according to relevant classes (Figure 3). Table 1 shows the locus ID and domains of all the putative R-genes in qBFR4. Based on our results, five genes in qBFR4 have NBS-LRR domains. However, it is unclear if these genes have an additional CC or TIR domain to be classified under class 1 or class 2 R-gene, hence these genes are classed as Class 1/Class 2 in Figure 3. One of the gene with LRR domain maybe a class 3 R-gene (Figure 3). Seven genes with LRR receptor-like serine/threonine kinase that encode protein kinase & LRR domain were classified into Class 4 and out of these seven genes, six have an additional malectin domain and based on a study, a malectin-like leucine-rich repeat receptor-like kinase in Arabidopsis contributes to downy mildew disease resistance30. R-genes from class 5-7 were not found in qBFR4. Ser/Thr Kinase and protein kinase domain with Ser/Thr activity were vastly found in qBFR4. Since protein kinase domain is an umbrella term for Ser/Thr Kinase, there is a strong possibility that the protein kinase domain with Ser/Thr Kinase activity is Ser/Thr Kinase domain. Thirteen (13) genes with these domains were classified under Class 8 R-gene. Three genes associated to kinase family did not return any InterProScan results and these gene were classified as putative R-genes without any class.
Table (1):
List of disease resistance genes in qBFR4.
Name/ Locus ID |
Description Annotation |
Domain |
|---|---|---|
LOC_Os04g49220 |
Probably inactive receptor kinase At2g46850 |
No IPR |
LOC_Os04g49460 |
Probable L-type lectin-domain containing receptor kinase |
Protein Kinase & Ser/Thr Kin |
LOC_Os04g49480 |
Probable L-type lectin-domain containing receptor kinase |
Legume-lectine domain |
LOC_Os04g49510 |
Calcium-dependent kinase |
Protein Kinase |
LOC_Os04g49690 |
Receptor kinase FERONIA |
Malectin |
LOC_Os04g51009 |
Wall-associated receptor kinase |
Protein Kinase |
LOC_Os04g51030 |
Wall-associated receptor kinase |
Protein Kinase |
LOC_Os04g51040 |
Wall-associated receptor kinase 5 |
Protein Kinase |
LOC_Os04g51050 |
Wall-associated receptor kinase 5 |
Protein Kinase |
LOC_Os04g51370 |
Serine threonine- kinase minibrain |
Protein Kinase |
LOC_Os04g51580 |
Plant intracellular Ras-group-related LRR 1 |
LRR |
LOC_Os04g51950 |
Kinase superfamily |
Protein Kinase & Ser/Thr Kin |
LOC_Os04g52140 |
Serine threonine- kinase CTR1 isoform X1 |
Protein Kinase & Ser/Thr Kin |
LOC_Os04g52590 |
Probable LRR receptor-like serine threonine- kinase At1g56130 |
Protein Kinase & Malektin & LRR |
LOC_Os04g52600 |
Probable LRR receptor-like serine threonine- kinase At1g56130 |
Protein Kinase & Malektin & LRR |
LOC_Os04g52606 |
Probable LRR receptor-like serine threonine- kinase At1g56140 |
Protein Kinase & Malektin & LRR |
LOC_Os04g52614 |
Probable LRR receptor-like serine threonine- kinase At1g56140 |
Protein Kinase & Malektin & LRR |
LOC_Os04g52630 |
Probable LRR receptor-like serine threonine- kinase At1g56140 |
Protein Kinase & Malektin & LRR |
LOC_Os04g52640 |
Probable LRR receptor-like serine threonine- kinase At1g56140 |
Protein Kinase & Malektin & LRR |
LOC_Os04g52780 |
LRR receptor-like serine threonine- kinase FLS2 |
Protein Kinase & LRR |
LOC_Os04g52840 |
Serine threonine- kinase |
Protein Kinase |
LOC_Os04g52860 |
Probable receptor kinase At1g30570 |
Protein Kinase & Ser/Thr Kin |
LOC_Os04g52970 |
Disease resistance RGA2-like |
NB-ARC & LRR |
LOC_Os04g53000 |
Disease resistance RGA2 |
LRR |
LOC_Os04g53030 |
Disease resistance RGA2-like |
NB-ARC & LRR |
LOC_Os04g53050 |
NBS-LRR-like resistance |
NB-ARC & LRR |
LOC_Os04g53120 |
NBS-LRR type resistance for bacteria |
NB-ARC & LRR |
Disease resistance genes in qLBL5
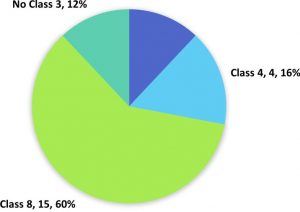
qLBL5 has 25 putative R-genes that makes up about 3.88% of the genes in the QTL (Figure 4). Table 2 shows the locus ID and domains of all the putative R-genes in qLBL5. Four genes with NBS LRR domains were classified into Class 1/Class 2 R-gene. These genes also lack CC or TIR domain which makes it impossible to differentiate these genes into Class 1 and 2. Besides, four genes annotated as LRR receptor-like serine threonine- kinase have been validated for the presence of LRR and protein kinase domain via InterProScan and these group of genes are classified into Class 4. Class 8 R-genes in qLBL5 consists of 16 kinase associated genes that are largely annotated as receptor like kinase, serine threonine kinase, calcium dependent kinase and CBL-interacting kinase. Three unclassified genes annotated as serine threonine receptor-like kinase, serine threonine- kinase and CBL-interacting serine threonine- kinase could be putative R-genes as well. Based on the annotation of these genes, it is suspected that they may be associated to R-gene although it didn’t return any results associated to R-gene domains through InterProScan.
Table (2):
List of disease resistance genes in qLBL5.
Name / ID Locus |
Gene Description |
Domain |
|---|---|---|
LOC_Os05g33690 |
LRR receptor-like serine threonine- kinase ERL1 |
Protein-kinase & LRR |
LOC_Os05g34220 |
Disease resistance RGA3 |
NB-ARC & LRR |
LOC_Os05g34230 |
Disease resistance RGA3 |
NB-ARC & LRR |
LOC_Os05g34270 |
Probably inactive leucine-rich repeat receptor kinase At5g48380 |
Ser-Thr Kin & LRR |
LOC_Os05g34390 |
G-type lectin S-receptor-like serine threonine- kinase At2g19130 |
Protein Kinase |
LOC_Os05g34950 |
Probable receptor kinase At1g11050 |
Ser-Thr Kin |
LOC_Os05g35760 |
Serine threonine kinase SAPK5 |
Protein kinase |
LOC_Os05g35770 |
Serine threonine kinase SAPK4 |
Protein kinase |
LOC_Os05g36050 |
Probable serine threonine- kinase At1g01540 |
Protein Kinase |
LOC_Os05g36960 |
Serine threonine- kinase Nek5 |
Protein kinase |
LOC_Os05g38020 |
Serine threonine receptor-like kinase NFP |
No IPR |
LOC_Os05g38070 |
CBL-interacting kinase 27 |
Protein kinase |
LOC_Os05g38770 |
Receptor-like kinase LIP2 |
Protein Kinase |
LOC_Os05g38800 |
Serine threonine- kinase ATM |
PWWP domain |
LOC_Os05g39080 |
Kinase superfamily |
Protein Kinase |
LOC_Os05g39090 |
Calcium-dependent kinase 13 |
Protein Kinase |
LOC_Os05g39860 |
CBL-interacting serine threonine- kinase 10 |
No IPR |
LOC_Os05g39870 |
CBL-interacting kinase 11 |
Protein Kinase |
LOC_Os05g39890 |
CBL-interacting kinase 28 |
Protein Kinase |
LOC_Os05g39900 |
CBL-interacting kinase 28 |
Protein Kinase |
LOC_Os05g40050 |
Probable LRR receptor-like serine threonine- kinase IRK |
Protein Kinase & LRR |
LOC_Os05g40150 |
Disease resistance RPP13 |
NB-ARC & LRR |
LOC_Os05g40180 |
Serine threonine- kinase chloroplastic |
Protein Kinase |
LOC_Os05g40270 |
Probable LRR receptor-like serine threonine- kinase At1g06840 isoform X1 |
Protein Kinase & LRR |
LOC_Os05g40540 |
Cyclin-dependent kinase B2-1 |
Protein Kinase |
Class 8 R-genes that comprise of kinases are prevalent in both QTLs. Receptor-like kinases (RLK) are particularly abundant in this group of kinases. RLK is a pathogen recognition receptor (PRR) that recognizes chitin, a type of elicitor present in especially fungal pathogen31,32. It is noteworthy that major diseases in rice like blast and sheath blight are caused by fungal pathogen and perhaps a myriad of RLKs is required in rice to detect the fungal pathogen for efficient activation of defence mechanism to halt the pathogen infection.
The identification and classification of defence-related genes
Defence-related genes in qBFR4
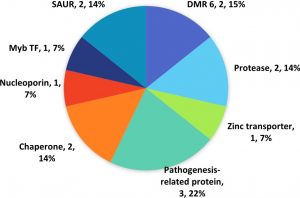
The mapping and GO annotation via Blast2GO provided the processes and functions carried out by individual genes and relevant genes that carry out defence-related processes and functions are handpicked for classification into respective functional groups. A total of 14 defence-related genes makes up about 0.04% of genes in qBFR4 (Table 3).
Table (3):
List of defence related genes in qBFR4 with descriptional and GO annotation.
Name/Locus ID |
Descriptional Annotation |
GO Annotation |
Enzyme Annotation |
Functional group |
|---|---|---|---|---|
LOC_Os04g49194 |
DOWNY MILDEW RESISTANCE 6-like |
Defence response to fungi |
Transferring phosphorus-containing groups |
DMR |
LOC_Os04g49210 |
DMR6-LIKE OXYGENASE 1 |
Defence response to fungi |
Transferring phosphorus-containing groups |
DMR |
LOC_Os04g50750 |
Subtilisin-like protease |
Signal transduction |
No |
Protease |
LOC_Os04g52310 |
Zinc transporter 3 |
Response to stimulus |
No |
Zinc transporter |
LOC_Os04g50184 |
Cathepsin B |
Defence response |
No |
Protease |
LOC_Os04g50700 |
S-norcoclaurine synthase |
Defence response |
No |
PR protein |
LOC_Os04g50710 |
S-norcoclaurine synthase |
Defence response |
No |
PR protein |
LOC_Os04g52880 |
BAG family molecular chaperone regulator 1-like |
Cell death |
No |
Chaperone |
LOC_Os04g52890 |
BAG family molecular chaperone regulator 1 |
Cell death |
No |
Chaperone |
LOC_Os04g51300 |
Thylakoid lumenal 29 kDa chloroplastic |
Peroxidase activity |
Peroxidase |
PR protein |
LOC_Os04g51900 |
Nuclear pore complex NUP88 |
Immune system process |
No |
Nucleoporin |
LOC_Os04g51130 |
Myb family transcription factor PHL11 |
Defence response |
No |
MYB TF |
LOC_Os04g52670 |
Auxin-responsive SAUR71 |
Response to auxin |
No |
SAUR |
LOC_Os04g52684 |
Auxin-responsive SAUR71-like |
Response to auxin |
No |
SAUR |
Two substilin-like proteases in qBFR4 are involved in signal transduction. Signal perception and transduction is probably the most crucial step to apprise the plant system to flick the switch to defence mode. The significance of subtilisin-like protease in transmitting signalling cascades has been indicated in past studies and were also shown to be expressed upon pathogen inoculation33. The pathogen recognition incident happens at the plant extracellular or cell surface and the accumulation of subtilisin-like protease in this location suggest that they may play a pivotal role in transducing the signal pertaining the pathogen recognition34. Besides, a nuclear pore complex NUP88 in qBFR4 is associated to immune response. In past studies, NUP88 is indicated in systemic acquired resistance (SAR) mediated by multiple R-genes, thus permitting resistance against wide range of pathogens35. The establishment of SAR is attributable to salicylic acid (SA) mediated signalling, a crucial part of signal transduction for defence responses.
A thylakoid lumenal 29 kDa chloroplastic gene in qBFR4 is enzyme coded as peroxidase and is involved in peroxidase activity. Peroxidases (PR-9) is essential in generating reactive oxygen species36. Rapid accumulation of ROS mediated by SA signals observed as one of the earliest event upon pathogen recognition. The involvement of ROS is not only restricted to direct antimicrobial activity but also has been implicated in signalling to establish hypersensitive response and activate other defence-related genes37. Two BAG family molecular chaperone regulator 1 in qBFR4 are involved in cell death and response to stress. Rapid cell death in plant cell is a type of hypersensitive response to halt the progression of infection in host plant38.
A zinc transporter that responds to stimuli was identified in qBFR4. The occupation of pathogen on host plant triggers some genes to exert response to stimuli. It is proposed that zinc transporter may transport zinc in response to this type of stimulus to trigger defence responses. A previous study suggested that zinc may play a role as regulatory factor in defence response as zinc is proven to induce JA/ETH signalling pathway which leads to enhanced PAD3 expression to provide resistance against Alternaria brassicicola in Arabidopsis thaliana39.
Apart from that, two auxin-responsive SAUR71 that responds to auxin were also present in qBFR4. Small Auxin Up RNAs (SAURs) is the largest family of auxin response genes. Auxin, an important plant hormone associated to biotic stress, regulates the expression of these genes in response to infection. Ghanashyam & Jain (2009) have shown that several auxin responsive genes which includes SAUR have responded to the attack of M. oryzae40. Two S-norcoclaurine synthase also regarded as pathogenesis-related proteins (PR proteins) classed as PR-10 protein41 were identified in current study. PR proteins are widely regarded as antifungal agents and directly associated to defence response which accumulates in host cell in response to SA-mediated signalling upon disease transmission by pathogen.
Table (4):
List of defence related genes in qLBL5 with annotations and functional groups.
Descriptional Annotation |
GO Annotation |
Enzyme Annotation |
Functional group |
|---|---|---|---|
Autophagy-related 18g |
Response to stress |
No |
Autophagy-related |
Glutathione transferase GST 23-like |
Signal transduction |
Glutathione transferase |
GST |
MLO 1 |
Defence response |
No |
GPCR |
Subtilisin-like protease |
Signal transduction |
Peptidase |
Protease |
Subtilisin-like protease |
Signal transduction |
Peptidase |
Protease |
Glucan endo-1,3-beta-glucosidase 14 |
Defence response |
No |
Pathogenesis related protein |
Pathogenesis-related 1-like |
Defence response |
No |
Pathogenesis related protein |
Zinc finger ZAT8 |
Response to chitin |
No |
Zinc finger |
Respiratory burst oxidase homolog H |
Defence response |
Peroxidase |
Pathogenesis related protein |
Probable WRKY transcription factor 70 |
Transcription factor activity |
No |
WRKY TF |
Probable WRKY transcription factor 71 |
Transcription factor activity |
No |
WRKY TF |
ACCELERATED CELL DEATH 6 |
Signal transduction |
No |
Plasma-membrane-localized protein |
Hydroxyproline-rich glyco -like |
Cell wall component |
No |
HPRG |
GPCR |
Signal transduction |
No |
GPCR |
Phosphatidylinositol:ceramide inositolphosphotransferase |
Response to stress |
No |
Transferase |
Coronatine-insensitive 1 |
Response to stress |
No |
F-box protein |
Abscisic acid receptor PYL4-like |
Signal transduction |
No |
Receptor |
In contrary to the above, a few genes were found to negatively regulate the defence response and increase the susceptibility of host plant to pathogen. On that premise, two genes annotated as downy mildew resistance 6 (DMR 6) were identified in qBRF-4. In a study, DMR 6 were upregulated when it was inoculated with the causative pathogen and only the mutant dmr6 were shown to increase the salicylic acid (SA) level42. This could mean that the defence pathway activated may have been SA independent and not one requiring SA.
To sum up the above discussion, after pathogen recognition by R-genes in qBFR4, signalling cascades will be relayed and mediated though SA-mediated pathway to induce defence responses such as rapid ROS production, hypersensitive response and. systemic acquired resistance (SAR). SAR involves the accumulation of PR proteins, and in the case of qBFR4, where PR-10 protein is involved. Aside from salicylic acid, plant hormone such as auxin may serve as an accessory to regulate the defence response through genes that are responsive to it. As a response to stimuli (pathogen invasion), zinc transporter may transport zinc to regulate other signalling pathways such as JA/ETH signalling pathway for enhanced resistance. However, the negative regulation of defence response by DMR6 may reduce the resistance against pathogen in the host plant42.
Defence-related genes in qLBL5
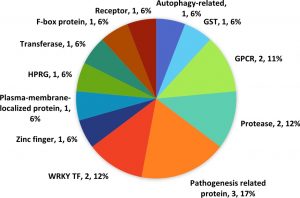
A total 17 (0.03%) defence related genes were found in qLBL5 and this sum comprises of a few transcription factors, PR proteins, autophagy related genes, and some enzymes.
A G-protein coupled receptor (GPCR) in qLBL5, is an important signal perceiving receptor43. As indicated earlier, signal transduction upon pathogen recognition is a prerequisite to actuate the responses from defence-related genes. G-protein coupled receptors (GPCRs) were implicated to mediate the extracellular signal to the intracellular environment for various physiological process including plant defence against pathogen. The signal transmitted by GPCR probably leads to SA mediated signalling pathway. Apart from that, a plasma membrane localized protein known as accelerated cell death 6 in qLBL5 regulates SA signalling pathway preceding cell death44. By acting in a positive feedback loop with SA signal, it is most definitely involved in the activation of other defence-related genes45.
Two subtilisin-like proteases in this QTL may be involved in signal transduction and they are proposed to function as a receptor for pathogen recognition to activate downstream signalling cascades46 as it was expressed rapidly right before the activation of SA responsive genes47. An isozyme in qLBL5 is known as glutathione transferase GST 23-like is also important in eliciting signalling cascades for defence responses48. Upon infection from Colletotrichum destructivum and C. orbiculare, GST genes were induced for transcription in Nicotiana benthamiana49.
One abscisic acid receptor, PYL4 was discovered in qLBL5 and is implicated in the crosstalk between abscisic acid and jasmonic acid (JA) signalling which elicits the biosynthesis of secondary metabolites when plant’s face biotic stress including pathogen invasion50. Defence signalling against biotrophic pathogen usually involve JA-mediated signalling whereas defence against necrotrophic pathogens involves SA-mediated signalling. Being a hemibiotroph, M. oryzae sustains its biotrophic lifestyle in living tissue and necrotrophic lifestyle in dead plant tissue. Hence, it is possible that both SA and JA mediated signalling utilized interchangeably by the host plant may be able to protect itself against a hemibiotroph like M. oryzae51.
The current study identified one peroxidase, annotated as respiratory burst oxidase homolog H suggesting the involvement of ROS in the defence system against fungal pathogen in qLBL5. These ROS species are often associated to hypersensitive response and also suggested to enhance other SA mediated defence response52. The production of the ROS species is largely contributed by peroxidase36. In a study, the formation of lignin to protect the cell wall of reed canary grass following the fungal penetration spiked the peroxidase activity level53.
Two WRKY transcription factor namely WRKY 70 and WRKY 71 in qLBL5 may carry out transcription activity to regulate expression of the defence-related signal and process. As a matter of fact, an increased expression of 15 WRKY genes in Oryza sativa japonica were observed upon the incompatible reaction between M. grisea and the host. Besides, WRKY 70 was reported to serve as a cross talk component in plant defence signalling network by activating SA mediated signals and repressing JA mediated signals54. The overexpression of WRKY71 in rice has upregulated the expression of defence-related genes induced by elicitors55.
Aside from that, one MLO1 gene in qLBL5 is directly associated to defence response against pathogen by preventing the penetration of fungus into the epidermal cell wall, and subsequently governs several processes for cell wall fortification. It may be regulated by Ca2+-dependent calmodulin binding and does not require heterotrimeric G proteins for signal transduction. Defence response of MLO1 varies between different pathogens where a homozygous mutant mlo in barley showed resistance towards powdery mildew but found susceptible in a very severe manner to M. oryzae, the causative pathogen of rice blast and vice versa for wild type MLO56. This shows that the wild type MLO1 is resistance or less susceptible to M. oryzae.
Three genes in qLBL5 responds to stress posed as pathogen infection by various means. An autophagy-related gene in qLBL5 executes cellular self-digestion to restrict the overspreading of pathogen-induced cell death to uninfected area. In the rice blast fungus M. oryzae, autophagic cell death is required for degradation of conidia and thus fungal pathogenicity57. Along with that, coronatine-insensitive protein 1 regulates the expression of plant genes during plant-pathogen interactions against Pseudomonas syringae and Alternaria brassicicol and also required for JA-mediated defence processes58, 59. One gene annotated as phosphatidylinositol:ceramide inositolphospho-transferase modulates plant programmed cell death (PCD) which has been indicated in Arabidopsis in conferring defence against Golovinomyces cichoracearum60.
Two PR proteins knowns as PR-1 and PR-2 were also found in qLBL5. PR-1 genes (OsPR1a and OsPR1b) were reported to be expressed in rice after blast infection8 as a consequence of ROS activation61. Apart from that, (PR-2 protein) comprises of beta-1,3-glucanase carries out anti-fungal activity by hydrolysing the 1,3-b-D-glucosidic linkages in b-1,3-glucans, an important fungal cell wall structural component62.
Briefly, the defence mechanism in qLBL5 is akin to qBFR4 to some extent. In addition to the ROS production, hypersensitive response and accumulation of PR proteins initiated by SA. In addition to the above strategies, response to stress is also triggered after pathogen invasion which is exhibited through cellular digestion, cell death and regulation of defence-gene expression. The defence process ends with the regulation of defence gene-expression which is modulated through activity of transcription factor activity.
The defence mechanism model using directed acyclic graph
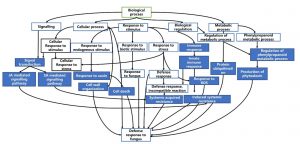
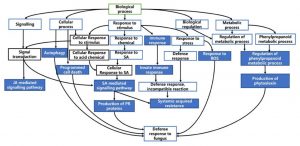
Directed acyclic graph (DAG) was produced using Blast2GO to visualize the gene ontologies associated with defence response. This graph portrays hierarchical structure of functional annotation related to defence processes.
Figure 7 and 8 shows the summarized version of DAG for biological processes related to defence for qBFR4 and qLBL5 respectively. Based on the DAG constructed, when the pathogen instigates infection, the host will mount the first line of defence through pathogen-associated molecular patterns (PAMP) triggered immunity (PTI), which happens at the cell surface. To vanquish the host PTI, the pathogen will produce effectors/elicitors and this will be recognized by R-gene in host which in turn activates the effector-triggered immunity 30 or also known as gene-gene resistance. ETI is mediated by R genes, such as NBS-LRR, receptor-like kinases (RLK) and serine/threonine kinase. After pathogen recognition and the incompatible reaction of R-gene with avr gene in pathogen, these R-genes will initiate and transduce signal to alarm the defence system in the host plant.
The signal transduction from R-genes will be relayed to other signal transmitting molecules for the activation of other defence responses. SA and JA mediated signalling pathway may be activated as a consequent of pathogen recognition. SA is a plant immune signal that induces systemic acquired resistance (SAR) whereas JA induces induced systemic resistance (ISR). SA-mediated signalling induces ROS production probably via peroxidase that carries out peroxidase activity. This will cause oxidative burst and some genes may respond to this oxidative stress for subsequent defence process. Thereafter, hypersensitive response (HR) will take place and this event is manifested as rapid cell death. Along with that, SAR activated through SA signal will lead to the production of pathogenesis-related (PR) proteins such as S-norcoclaurine synthase (PR-10) in qBFR4, PR-1 and beta-1,3-glucanase (PR-2) in qLBL5 which carry out anti-fungal activity in response to stress.
With the signal transmission, responses to stimuli, stress and plant hormone like auxin will be activated for the regulation of defence responses. Response to stress is also represented through cell death and cellular self-digestion (autophagy). The transcription activity conducted by the transcription factors like WRKY and MYB may regulate the expression of other defence-related genes 63.
Overall, it is evident that both QTLs exhibit a fairly low proportion of genes related to defence and resistance which may explain why they confer moderate levels of resistance to plants. However, qBFR4 (0.11%) has relatively higher percentage of R-genes and defence-related genes as compared to qLBL5 (0.07%). This suggests that qBFR4 is more beneficial and efficient than qLBL5 as it gives a better coverage of defence related genes for a small QTL region. In concordance with this, it is proposed that qLBL5 has to be fine-mapped to eliminate genes that are not of interest to reduce unnecessary drag effect during breeding. In terms of R-genes, RLKs from Class 8 R-genes were vastly found in both QTLs which is attributable to ability of RLK to recognize chitin in fungal pathogen which is the causative agent for the major diseases in rice. The characterization of genes in both QTLs gave a rough outline on resistance mechanism in both QTLs which sets off with pathogen recognition by R-genes and subsequent signalling mediated through several pathways such as SA and JA for the activation of downstream defence processes. Major R-genes found along the QTLs such as disease resistance RGA2 and RGA4 in qBFR4 and disease resistance RGA3 and RPP13 may be utilized in various molecular techniques such as cloning to develop resistant cultivar. The broad spectrum resistance given by qBFR4 and qLBL5 can be used to maximum potential through QTL pyramiding technique with other well characterized QTLs such as qSBR11-164 and qShb9-263 to confer multiple disease resistance (MDR) towards rice blast and sheath blight.
Acknowledgements
The work presented here is funded by Universiti Kebangsaan Malaysia, DCP-2017-004/1.
- Nadarajah K, Krishnan HK, Ali HZ. Cellular and molecular interactions of Magnaporthe oryzae S3 in rice. J Pure Appl Microbiol, 2014; 8 (5), 3481-91.
- Muni NM, Nadarajah K, editors. Morphological and molecular characterization of Magnaporthe oryzae (fungus) from infected rice leaf samples. AIP Conference Proceedings; 2014: AIP.
- Gowda M, Shirke MD, Mahesh H, Chandarana P, Rajamani A, Chattoo BB. Genome analysis of rice-blast fungus Magnaporthe oryzae field isolates from southern India. Genomics data, 2015; 5, 284-91.
- Marcel S, Sawers R, Oakeley E, Angliker H, Paszkowski U. Tissue-adapted invasion strategies of the rice blast fungus Magnaporthe oryzae. Plant Cell, 2010, tpc-110.
- Couch BC, Kohn LM. A multilocus gene genealogy concordant with host preference indicates segregation of a new species, Magnaporthe oryzae, from M. grisea. Mycologia, 2002; 94 (4), 683-93.
- Belkhadir Y, Subramaniam R, Dangl JL. Plant disease resistance protein signaling: NBS–LRR proteins and their partners. Current Opinion in Plant Biology, 2004; 7 (4), 391-9.
- McDowell JM, Simon SA. Recent insights into R gene evolution. Molecular Plant Pathology, 2006; 7 (5), 437-48.
- Agrawal GK, Rakwal R, Jwa N-S, Agrawal VP. Signalling molecules and blast pathogen attack activates rice OsPR1a and OsPR1b genes: a model illustrating components participating during defence/stress response. Plant Physiology and Biochemistry, 2001; 39 (12), 1095-103.
- Kiyosawa S. Gene analysis for blast resistance. Oryza, 1981; 18, 196-203.
- Koizumi S. Durability of resistance to rice blast disease. JIRCAS Work Rep, 2007; 53, 1-10.
- Miah G, Rafii M, Ismail M, Puteh A, Rahim H, Asfaliza R, et al. Blast resistance in rice: a review of conventional breeding to molecular approaches. Molecular biology reports, 2013; 40 (3), 2369-88.
- Rahim HA, Bhuiyan MAR, Saad A, Azhar M, Wickneswari R. Identification of virulent pathotypes causing rice blast disease (‘Magnaporthe oryzae’) and study on single nuclear gene inheritance of blast resistance in F2 population derived from Pongsu Seribu 2 x Mahshuri. Australian Journal of Crop Science, 2013; 7 (11), 1597.
- Rahim H, Bhuiyan M, Lim L, Sabu K, Saad A, Azhar M, et al. Identification of quantitative trait loci for blast resistance in BC. Genet Mol Res, 2012; 11, 3277-89.
- Mizobuchi R, Sato H, Fukuoka S, Yamamoto S, Kawasaki-Tanaka A, Fukuta Y. Mapping of a QTL for field resistance to blast (Pyricularia oryzae Cavara) in Ingngoppor-tinawon, a rice (Oryza sativa L.) landrace from the Philippines. Jpn Agric Res Q, 2014; 48 (4), 425-31.
- Aglawe SB, Bangale U, Satya RDSJ, Balija V, Pal BV, Kumar SS, et al. Identification of novel QTLs conferring field resistance for rice leaf and neck blast from an unique landrace of India. Gene Reports, 2017; 7, 35-42.
- Ballini E, Morel J-B, Droc G, Price A, Courtois B, Notteghem J-L, et al. A genome-wide meta-analysis of rice blast resistance genes and quantitative trait loci provides new insights into partial and complete resistance. Molecular Plant-Microbe Interactions, 2008; 21 (7), 859-68.
- Haramoto E, Kitajima M, Hata A, Torrey JR, Masago Y, Sano D, et al. A review on recent progress in the detection methods and prevalence of human enteric viruses in water. Water research, 2018.
- Pray L. Transposons, or jumping genes: Not junk DNA. Nature Education, 2008; 1 (1), 32.
- Todorovska E. Retrotransposons and their role in plant—genome evolution. Biotechnology & Biotechnological Equipment, 2007; 21 (3), 294-305.
- Xiao H, Jiang N, Schaffner E, Stockinger EJ, Van Der Knaap E. A retrotransposon-mediated gene duplication underlies morphological variation of tomato fruit. science, 2008; 319 (5869), 1527-30.
- Sahebi M, Hanafi MM, van Wijnen AJ, Rice D, Rafii M, Azizi P, et al. Contribution of transposable elements in the plant’s genome. Gene, 2018.
- Gilroy EM, Hein I, Van Der Hoorn R, Boevink PC, Venter E, McLellan H, et al. Involvement of cathepsin B in the plant disease resistance hypersensitive response. The Plant Journal, 2007; 52 (1), 1-13.
- Afzal AJ, Wood AJ, Lightfoot DA. Plant receptor-like serine threonine kinases: roles in signaling and plant defense. Molecular Plant-Microbe Interactions, 2008; 21 (5), 507-17.
- Kim HS, Delaney TP. Arabidopsis SON1 is an F-box protein that regulates a novel induced defense response independent of both salicylic acid and systemic acquired resistance. The Plant Cell, 2002; 14 (7), 1469-82.
- Vogel JP, Raab TK, Somerville CR, Somerville SC. Mutations in PMR5 result in powdery mildew resistance and altered cell wall composition. The Plant Journal, 2004; 40 (6), 968-78.
- Wang Y-H, Warren Jr JT. Mutations in retrotransposon AtCOPIA4 compromises resistance to Hyaloperonospora parasitica in Arabidopsis thaliana. Genetics and molecular biology, 2010; 33 (1), 135-40.
- Hernández-Pinzón I, de Jesús E, Santiago N, Casacuberta JM. The frequent transcriptional readthrough of the tobacco Tnt1 retrotransposon and its possible implications for the control of resistance genes. Journal of molecular evolution, 2009; 68 (3), 269-78.
- Gururani MA, Venkatesh J, Upadhyaya CP, Nookaraju A, Pandey SK, Park SW. Plant disease resistance genes: current status and future directions. Physiological and molecular plant pathology, 2012; 78, 51-65.
- Jia Y, McAdams SA, Bryan GT, Hershey HP, Valent B. Direct interaction of resistance gene and avirulence gene products confers rice blast resistance. The EMBO journal, 2000; 19 (15), 4004-14.
- Hok S, Danchin EG, Allasia V, Panabieres F, Attard A, Keller H. An Arabidopsis (malectin like) leucine rich repeat receptor like kinase contributes to downy mildew disease. Plant, cell & environment, 2011; 34 (11), 1944-57.
- Wu Y, Zhou JM. Receptor Like Kinases in Plant Innate Immunity. J Integr Plant Biol, 2013; 55 (12), 1271-86.
- Felix G, Regenass M, Boller T. Specific perception of subnanomolar concentrations of chitin fragments by tomato cells: induction of extracellular alkalinization, changes in protein phosphorylation, and establishment of a refractory state. The Plant Journal, 1993; 4 (2), 307-16.
- Tornero P, Conejero V, Vera P. Primary structure and expression of a pathogen-induced protease (PR-P69) in tomato plants: similarity of functional domains to subtilisin-like endoproteases. Proceedings of the National Academy of Sciences, 1996; 93 (13), 6332-7.
- Dixon RA, Lamb CJ. Molecular communication in interactions between plants and microbial pathogens. Annual review of plant biology, 1990; 41 (1), 339-67.
- Cheng YT, Germain H, Wiermer M, Bi D, Xu F, García AV, et al. Nuclear pore complex component MOS7/Nup88 is required for innate immunity and nuclear accumulation of defense regulators in Arabidopsis. The Plant Cell, 2009; 21 (8), 2503-16.
- O’Brien JA, Daudi A, Butt VS, Bolwell GP. Reactive oxygen species and their role in plant defence and cell wall metabolism. Planta, 2012; 236 (3), 765-79.
- Neill SJ, Desikan R, Clarke A, Hurst RD, Hancock JT. Hydrogen peroxide and nitric oxide as signalling molecules in plants. Journal of experimental botany, 2002; 53 (372), 1237-47.
- Morel J-B, Dangl JL. The hypersensitive response and the induction of cell death in plants. Cell death and differentiation, 1997; 4 (8), 671.
- Martos S, Gallego B, Cabot C, Llugany M, Barceló J, Poschenrieder C. Zinc triggers signaling mechanisms and defense responses promoting resistance to Alternaria brassicicola in Arabidopsis thaliana. Plant Science, 2016; 249, 13-24.
- Ghanashyam C, Jain M. Role of auxin-responsive genes in biotic stress responses. Plant signaling & behavior, 2009; 4 (9), 846-8.
- Lee E-J, Facchini P. Norcoclaurine synthase is a member of the pathogenesis-related 10/Bet v1 protein family. The Plant Cell, 2010, tpc. 110.077958.
- Zeilmaker T, Ludwig NR, Elberse J, Seidl MF, Berke L, Van Doorn A, et al. DOWNY MILDEW RESISTANT 6 and DMR6 LIKE OXYGENASE 1 are partially redundant but distinct suppressors of immunity in Arabidopsis. The Plant Journal, 2015; 81 (2), 210-22.
- Nadarajah K, Kumar IS, Sangapillai V, Omar NS. Recent advances in understanding the fungal G-protein-coupled receptors Malays J Microbiol, 2018.
- Rate DN, Cuenca JV, Bowman GR, Guttman DS, Greenberg JT. The gain-of-function Arabidopsis acd6 mutant reveals novel regulation and function of the salicylic acid signaling pathway in controlling cell death, defenses, and cell growth. The Plant Cell, 1999; 11 (9), 1695-708.
- Zhang Z, Shrestha J, Tateda C, Greenberg JT. Salicylic acid signaling controls the maturation and localization of the Arabidopsis defense protein ACCELERATED CELL DEATH 6. Mol Plant, 2014; 7 (8), 1365-83.
- Figueiredo A, Monteiro F, Sebastiana M. Subtilisin-like proteases in plant–pathogen recognition and immune priming: a perspective. Frontiers in plant science, 2014; 5, 739.
- Ramírez V, López A, Mauch-Mani B, Gil MJ, Vera P. An extracellular subtilase switch for immune priming in Arabidopsis. PLoS Pathogens, 2013; 9 (6), e1003445.
- Gullner G, Kômíves T. Defense reactions of infected plants: roles of glutathione and glutathione S-transferase enzymes. Acta Phytopathologica et Entomologica Hungarica, 2006; 41 (1-2), 3-10.
- Dean J, Goodwin P, Hsiang T. Induction of glutathione S-transferase genes of Nicotiana benthamiana following infection by Colletotrichum destructivum and C. orbiculare and involvement of one in resistance. Journal of Experimental Botany, 2005; 56 (416), 1525-33.
- Kazan K, Manners JM. Jasmonate signaling: toward an integrated view. Plant physiology, 2008; 146 (4), 1459-68.
- Park J-Y, Jin J, Lee Y-W, Kang S, Lee Y-H. Rice blast fungus (Magnaporthe oryzae) infects Arabidopsis via a mechanism distinct from that required for the infection of rice. Plant physiology, 2009; 149 (1), 474-86.
- Shirasu K, Nakajima H, Rajasekhar VK, Dixon RA, Lamb C. Salicylic acid potentiates an agonist-dependent gain control that amplifies pathogen signals in the activation of defense mechanisms. The Plant Cell, 1997; 9 (2), 261-70.
- Vance CP, Anderson JO, Sherwood RT. Soluble and cell wall peroxidases in reed canarygrass in relation to disease resistance and localized lignin formation. Plant physiology, 1976; 57 (6), 920-2.
- Phukan UJ, Jeena GS, Shukla RK. WRKY transcription factors: molecular regulation and stress responses in plants. Frontiers in Plant Science, 2016; 7, 760.
- Chujo T, Kato T, Yamada K, Takai R, Akimoto-Tomiyama C, Minami E, et al. Characterization of an elicitor-induced rice WRKY gene, OsWRKY71. Bioscience, biotechnology, and biochemistry, 2008; 72 (1), 240-5.
- Piffanelli P, Zhou F, Casais C, Orme J, Jarosch B, Schaffrath U, et al. The barley MLO modulator of defense and cell death is responsive to biotic and abiotic stress stimuli. Plant Physiology, 2002; 129 (3), 1076-85.
- Veneault-Fourrey C, Barooah M, Egan M, Wakley G, Talbot NJ. Autophagic fungal cell death is necessary for infection by the rice blast fungus. Science, 2006; 312 (5773), 580-3.
- van Wees SC, Chang H-S, Zhu T, Glazebrook J. Characterization of the early response of Arabidopsis to Alternaria brassicicola infection using expression profiling. Plant Physiology, 2003; 132 (2), 606-17.
- He P, Chintamanani S, Chen Z, Zhu L, Kunkel BN, Alfano JR, et al. Activation of a COI1 dependent pathway in Arabidopsis by Pseudomonas syringae type III effectors and coronatine. The Plant Journal, 2004; 37 (4), 589-602.
- Wang W, Yang X, Tangchaiburana S, Ndeh R, Markham JE, Tsegaye Y, et al. An inositolphosphorylceramide synthase is involved in regulation of plant programmed cell death associated with defense in Arabidopsis. The Plant Cell, 2008; 20 (11), 3163-79.
- Nadarajah K, Ali HZ, Muni NM. The host pathogen interactions observed between’Fusarium’sp. F2 and rice. Plant Omics, 2015; 8 (6), 572.
- Xu X, Feng Y, Fang S, Xu J, Wang X, Guo W. Genome-wide characterization of the â-1, 3-glucanase gene family in Gossypium by comparative analysis. Scientific reports, 2016; 6, 29044.
- Chanthran SSD, Cheah BH, Nadarajah K. In silico analysis of disease resistance and defence-related genes for a major sheath blight qShb 9-2 QTL in rice. Malaysian Journal of Microbiology, 2018.
- Kumar IS, Cheah BH, Nadarajah K. In silico identification and classification of disease resistance genes and defense-related genes against sheath blight from QTL qSBR11-1 in rice (Oryza sativa L.). Undergrad Res J Integ Biol, 2017; 1.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.