Bacterial skin and soft-tissue infections (SSTIs) are a major health concern worldwide because of their high prevalence, frequent recurrence, and increasing antibiotic resistance. Recent research indicates that SSTI incidence can reach up to 77.5 cases per year in high-income settings, while prevalence rates may be as high as 66% among hospitalised patients in resource-limited regions. Recurrence rates range from 7%-45% depending on comorbidities, treatment adequacy, and host variables. Methicillin-resistant Staphylococcus aureus (MRSA) and other resistant bacteria reduce the efficacy of traditional treatments. Novel antibiotics such as dalbavancin, tedizolid, oritavancin, and delafloxacin are effective against resistant SSTIs, but their use varies regionally. This review summarises recent research on SSTI epidemiology, recurrence, and resistance patterns, emphasising diagnostic gaps, treatment problems, and potential therapeutic methods. Enhanced surveillance, uniform reporting, and newer antimicrobials are critical for reducing the SSTI burden and directing future research.
Pyogenic Infections, Soft Tissue Infections, Antibiotic Resistance, Bacterial Infection
The global increase in bacterial infections resistant to numerous antibiotics threatens to return contemporary medicine to the pre-antibiotic era. Although new antibiotics are still being produced, the cost of developing them is high, and bacteria will eventually develop resistance to them. Breaking the cycle of bacterial infection requires non-antibiotic techniques.1 Recent reviews of experimental antibiotics indicate that medication development is substantially slower than the emergence and spread of resistant bacteria. Only two new antibiotic classes, dalbavancin and oritavancin, have entered the market over the past 30 years. This standstill has prompted research into new chemicals and anti-infective treatments.
Antimicrobial peptides, antivirulence drugs, bacteriophages, and antibodies show promise as replacements or adjuncts to standard antibiotics. Although the pipeline for phase II and III antibacterial drugs is still restricted, progress is possible through the combination of computational techniques with chemical and biological innovation.2,3 Antibiotics have saved lives since their introduction in the 1940s, but rising resistance has swung the advantage back to bacteria. With high rates of emergent resistance and limited progress in antibiotic discovery, the demand for therapeutic methods other than conventional chemotherapy is increasing.
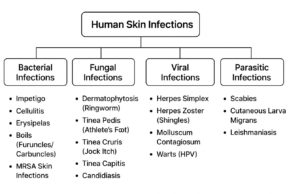
Human skin infections
The skin is the body’s primary defence against microbial invasion, acting as a physical barrier and releasing low-pH secretions and fatty acids that prevent pathogen growth. Its indigenous microbiota further limits the colonisation of dangerous pathogens.4,5 The skin not only protects against diseases but also controls temperature, prevents fluid loss, and aids in sensory awareness. Pathogens that break this barrier cause tissue injury and an inflammatory response.6,7
Types of human skin infections
Primary infections
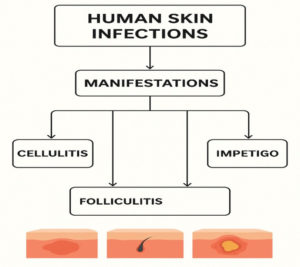
Primary skin infections appear in normally healthy skin, are caused by a single pathogen, and exhibit a specific clinical pattern (Figure 1). Examples include impetigo, folliculitis, and boils.8
Secondary infections
Secondary infections develop in diseased skin. Their clinical course varies depending on underlying diseases (Figure 1). Examples include intertrigo and toe-web infections.9
Bacterial skin infections
Bacterial skin infections are among the most common illnesses in hospitals and are sometimes referred to as skin and soft tissue infections (SSTIs) or skin and soft structure infections (SSSIs). Erythema, warmth, induration, and discomfort are examples of local or systemic inflammatory reactions caused by microbial invasion of the epidermal and subcutaneous tissue.10 SSTIs can range from severe, potentially fatal necrotising fasciitis to minor, self-limiting illnesses.11,12 According to Mngqibisa et al.,13 SSTIs are classified as deep (affecting the subcutaneous tissue, fascia, and muscle) or superficial (affecting the epidermis and dermis). Deeper infections are typically polymicrobial, whereas superficial infections often include a single pathogen. Cellulitis penetrates the dermis, whereas conditions such as folliculitis, erysipelas, and impetigo affect the epidermis. Fasciitis or myonecrosis may develop when the infection spreads to deeper tissues.14,15
Fungal skin infections
Dermatophytosis and cutaneous candidiasis are examples of prevalent superficial fungal diseases. Dermatophytic infections on different body sites lead to tinea capitis, tinea corporis, tinea cruris, tinea manuum, and tinea pedis. Other dermatophytes (tinea unguium) and fungal species (onychomycosis) cause fungal nail infections.16 Another common fungal illness is candidiasis, which is primarily caused by Candida albicans.17
Viral skin infections
Childhood chickenpox is caused by primary infection with the varicella-zoster virus (VZV), and the reactivation of latent VZV causes herpes zoster. Herpes simplex virus (HSV)-2 mostly causes genital herpes, whereas HSV-1 causes cold sores, stomatitis, and ocular infections. Human papillomavirus (HPV) often cause warts in children, teenagers, and older adults.18,19 A poxvirus illness called molluscum contagiosum is prevalent throughout the world, particularly in tropical regions and in children.20
Parasitic skin infections
Parasitic skin infections continue to contribute substantially to the global dermatologic disease burden especially in tropical, subtropical and resource†limited regions. Scabies, caused by the mite Sarcoptes scabiei, remains one of the most common and burdensome skin diseases worldwide. Current estimates indicate that hundreds of millions of people are affected each year globally. Treatment challenges and emerging resistance remain a concern. For instance, a 2025 report documented a case of CLM refractory to conventional therapy with Ivermectin.21
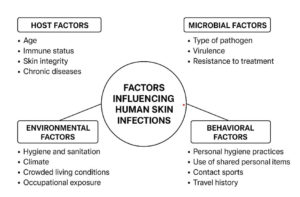
Factors influencing human skin infections
The prevalence of pyoderma is greatly influenced by various socioeconomic and environmental factors, including poverty, poor hygiene, overcrowding, malnutrition, and climate.22,23 Climate, genetics, age, sex, stress, diet, and hospitalisation affect the microbial makeup of skin infections.24 Because of physiological skin changes and lowered immunity, older adults are especially vulnerable. Approximately 70% of adults over 70 have at least one skin condition, and skin and soft tissue infections cause greater morbidity and death in this age range.25
Age-related skin thinning, decreased sebaceous gland activity, and dehydration increase infection susceptibility and hinder recovery (Figure 2). Skin fragility worsens with prolonged UV exposure.26,27 Proteus mirabilis and Pseudomonas aeruginosa colonise older adults more frequently, partially because of their weakened immune systems.28 The risk of recurring or severe SSTIs is further increased by comorbid conditions such as diabetes, renal disease, obesity, HIV infection, and immunosuppressive medication.29 The likelihood of infection also increases due to lymphedema and surgical lymph node excision.30
Occurrence of human skin infections
In India, bacterial skin infections are common and are a major reason for dermatology consultations. Because of widespread and improper antibiotic usage, illnesses that were previously treatable are becoming less responsive to widely used antibiotics (Table). Of the 178 lab-confirmed cases in an analysis of clinically suspected SSTIs, 66.4% of samples tested positive for SSTI pathogens.31
Table:
Epidemiology and recurrence of skin and soft tissue infections (SSTIs)
Measure/Outcome |
Estimate |
Sample/Numerator & CI |
Study Context |
Ref. |
|---|---|---|---|---|
Incidence of SSTI episodes (USA) |
77.5 per 1,000 person-years |
9.1 million SSTI episodes in 5.4 million patients; 95% CI 77.4-77.5 per 1,000 PYO. (OUP Academic) |
Retrospective claims-data cohort (Optum Clinformatics), 2010-2020, USA. (PMC) |
37 |
Proportion of recurrent SSTI (USA cohort) |
26.3% of index cases |
Among ~2.98 million index SSTI cases, 783,963 had a recurrence (26.3%). (OUP Academic) |
Same Optum cohort (2010-2020). (PubMed) |
37 |
Prevalence of SSTI among suspected hospitalized cases (Uganda) |
66.4% |
268 randomly selected reports; prevalence 66.4% (95% CI ~60.7-72.1). (PubMed) |
Retrospective study at Jinja Regional Referral Hospital, Uganda (2019-2021). (makir.mak.ac.ug) |
38 |
Recurrent skin and soft tissue infection (RSSTI) rate |
7%-45% |
Reported recurrence range in recent literature; risk depends on comorbidities/ host factors. (Lippincott Journals) |
Review of risk factors & management for recurrent SSTIs. (Lippincott Journals) |
39 |
Recurrence risk (by risk-score stratification) |
High-risk group: ~16.6% recurrence |
Based on BRRISC score stratification: high-risk (score 6–15) had recurrence of 16.6% (95% CI 13.3–20.4%). (PMC) |
Model / risk-stratification in cellulitis recurrence — part of the recurrence review. (PMC) |
39 |
According to a major health system dataset, the annual incidence of SSTIs is approximately 77.5 per 1,000 people. A 2024 epidemiological investigation identified 77.5 SSTI episodes per 1,000 people during the study period (5.4 million patients with 9.1 million SSTI episodes). This estimate reflects healthcare utilisation based on a large dataset.
An estimated 14 million instances of cellulitis, a prevalent bacterial SSTI, occur in the United States each year.32 SSTIs are among the most prevalent hospital/healthcare-associated infections (HCAIs). A national systematic review and meta-analysis in Nigeria reported an overall HCAI prevalence of approximately 15.8% (pooled data). The most common HCAIs in the included studies were surgical site infections and SSTIs.33
MRSA prevalence among SSTI isolates varies greatly by region. In Asia, documented regional prevalence ranged from 7.3% to 74% depending on country, environment, and year, highlighting regional variations in the burden of resistance.34
According to recent evaluations of recurrence risk, cellulitis has a recurrence rate of approximately 14% within one year and 45% within three years, which greatly increases the burden on the healthcare system.35
According to several recent regional studies (2023-2024), children living in high-burden environments show a high prevalence of impetigo, with prevalence estimates varying by context, season, and monitoring technique.36
Causative agents of human skin infections
Bacteria, fungi, viruses, and parasites cause skin infections. Most pyogenic skin infections are caused by bacterial pathogens, including Staphylococcus aureus and Streptococcus pyogenes.
Agents of bacterial causation
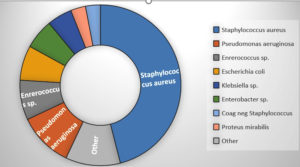
Because the skin is constantly exposed to the environment, it hosts a variety of microbiota, primarily bacteria (Figure 3). Gram-positive bacteria such as S. aureus, S. pyogenes, S. epidermidis, and Corynebacterium spp. are more common above the waist.40 Both Gram-positive and Gram-negative bacteria, including Enterobacteriaceae species, are more prevalent below the waist.41 The following Figure illustrates the overall occurrence of bacterial causal agents of human skin diseases, as suggested by Maraki et al.42
Gram-positive organisms are the most prevalent pathogens in complicated skin and skin structure infections (CSSSIs) and uncomplicated skin and skin structure infections (USSSIs). Figure 4 illustrates the most common Gram-positive and Gram-negative microorganisms that cause skin diseases. S. aureus was the most common (46%), followed by P. aeruginosa (11%), Enterococcus sp. (8%), E. coli (7%), Enterobacter sp. (6%), Klebsiella sp. (5%), P. mirabilis (3%), coagulase-negative Staphylococcus sp. (3%), and miscellaneous species (11%). S. aureus and S. pyogenes are the usual causes of USSSIs, and historically, empirical treatment has primarily targeted these Gram-positive bacteria.43 However, anaerobic and Gram-negative bacteria are becoming more common.44
SSTIs are characterised by different microbiology depending on how they enter the skin.45,46 According to a 2024 study conducted in northern India, 48.36% of pus samples from SSTIs were culture-positive during the study period, whereas 36.33% of the samples were culture-negative.47 Among the positive clinical isolates, 54.04% were Gram-negative bacilli and 45.96% were Gram-positive cocci. The top six microbes accounted for approximately 90% of all infections: S. aureus (38.05%), E. coli (17.39%), Pseudomonas sp. (11.82%), Acinetobacter sp. (10.16%), Klebsiella sp. (6.72%), and Coagulase-negative Staphylococcus species (5.50%). Other isolates included Proteus species (2.26%), Enterococcus species (2.19%), β-haemolytic streptococci (Group A) (0.18%), and S. pneumoniae (0.03%). S. aureus was a frequent pathogen in over 70% of all SSTIs and in 50% of community cellulitis cases. MRSA was present in 10.9% of patients with bacterial skin infections in South India.48 In a hospital-based prospective investigation conducted among children in north India in 2024, the incidence of community-acquired (CA) MRSA was 6.9%.49
Methicillin-resistant S. aureus (MRSA)
MRSA initially appeared in hospitals in the 1960s before spreading to the general public. Prevalence differs greatly by country (1%-74%).50 CA-MRSA is a leading cause of SSTIs worldwide, especially in children.51 S. aureus was responsible for 76% of SSTIs in a major emergency department research, with MRSA accounting for 59% of these infections.52 The Panton-Valentine leukocidin (PVL) toxin, which causes tissue necrosis and leukocyte death, is commonly present in CA-MRSA strains.53
Manifestations of human skin infections
The ability of an organism to infiltrate tissues, proliferate, and elude host defences is known as pathogenicity. Adhesins, enzymes, and toxins are among the many variables that affect microbial virulence, which differs between species.54
The epidermis, dermis, subcutaneous and adipose tissues, muscular fascia, and other layers of the skin architecture are colonised by bacteria, initially in small quantities. When the integumentary barrier is compromised, bacteria proliferate, leading to invasion and the development of SSTIs. (Figure 4). Finally, involvement of deeper skin structures may lead to fasciitis and myositis.55 These pathways allow native flora and normal skin flora introduced by the penetrating tool to enter.56-59
Bacterial infection
Three processes are necessary for the establishment of an SSTI: bacterial adhesion to host cells, tissue invasion with host defence evasion, and toxin generation. Most pathogenic bacteria have virulence genes that encode proteins providing these characteristics. Toxins are the strongest contributors to clinical illness among the bacterial virulence factors.60-62 Endotoxins and exotoxins are the two primary categories of toxins. Gram-negative bacterial cell walls are rich in lipopolysaccharide chains known as endotoxins. Lipopolysaccharides may be advantageous in small amounts because they stimulate the immune system.
By stimulating the production of costimulatory molecules, they increase T-lymphocyte activation and the release of chemoattractants. However, excessive lipopolysaccharide elaboration may cause harmful overstimulation of the host’s inflammatory and immunological systems.63 Conversely, exotoxins are actively released proteins that cause tissue injury or dysfunction via several pathways.
Inflammation
The host’s reaction to tissue invasion and damage is the other part of the infection process. The body uses inflammation as a defensive response to begin tissue repair and eliminate the causative organisms. Microbial invasion and damage to the skin or soft tissues alter vascular tone, increasing blood flow to the affected area. Further alterations in the microvasculature facilitate the extravasation of leukocytes and plasma proteins. At the damaged site, these cells and proteins move, gather, and become active. When cells are activated, they phagocytose and eliminate foreign objects, dead tissue, and microorganisms. The fever response is caused by certain exotoxins or pyrogenic cytokines. Ultimately, the injured area is cleansed, quarantined, and restored.64
Depth of infection
The potential depths of SSTI involvement and the corresponding diagnoses are shown in the following Figure. Cellulitis extends into the dermis, whereas superficial infections such as erysipelas, impetigo, folliculitis, furuncles, and carbuncles are found in the epidermis. Deeper infections progress to fasciitis or myonecrosis after passing through the subcutaneous tissue.65 Determining the level of infection through examination can be challenging, but laboratory testing can aid this evaluation.66
Coagulase, staphylokinase, lipase, and β-lactamase are among the virulence factors found in 90% of S. aureus strains. Factors that prevent phagocytosis include the polysaccharide slime layer and protein A on the cell surface. Certain strains produce cytolytic toxins, leukocidins, exfoliative toxins, epidermal cell differentiation inhibitors, and toxic shock syndrome toxins.67 Panton-Valentine leukocidin (PVL) is frequently linked to enhanced virulence in CA-MRSA.68 By producing the powerful chemotactic proteins interleukin-8 and leukotriene B4, this cytotoxin destroys leukocytes and promotes tissue necrosis.69,70
Additionally, S. aureus secretes enterotoxins, which are superantigens that activate T cells non-specifically and circumvent normal immune system pathways, causing a large release of cytokines. Research has shown that enterotoxins are produced by up to 50% of S. aureus isolates recovered from SSTIs.71,72
Antibiotic Treatment and Development of Drug Resistance
Antibiotic therapy
Antimicrobial medications can be either broad-spectrum or narrow-spectrum and act through various mechanisms: inhibition of cell wall synthesis (e.g., β-lactams, vancomycin), cell membrane disruption (e.g., polymyxins), inhibition of protein synthesis (e.g., tetracyclines, macrolides, aminoglycosides), inhibition of nucleic acid production (e.g., rifampin, quinolones), and suppression of metabolic pathways (e.g., trimethoprim, sulfonamides).73,74
Antibiotic policy
β-Lactams are first-line therapy for streptococcal and methicillin-sensitive S. aureus infections, with benzylpenicillin preferred for susceptible patients. Clindamycin is used for rapidly progressing infections, while newer fluoroquinolones (levofloxacin, moxifloxacin) and other systemic agents including cephalosporins, carbapenems, macrolides, linezolid, glycopeptides, glycylcyclines, and topical drugs like retapamulin broaden treatment options. Susceptibility patterns of skin isolates have been extensively studied.75
Development of drug resistance
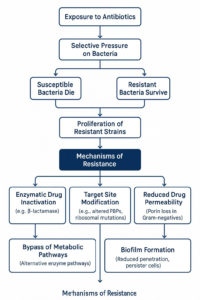
Drug resistance occurs when initially susceptible microbes stop responding to antibiotics (Figure 5). Resistance may be non-genetic, for example, a temporary change into L-forms. R-plasmid acquisition and chromosomal mutations are examples of genetic resistance. To withstand antibiotics, microorganisms employ five primary strategies: modified enzymes, drug-inactivating enzymes, reduced membrane permeability, changed drug targets, and avoidance of metabolic processes.76 Shortly after penicillin became widely used, S. aureus strains that produced penicillinase appeared.77 MRSA emerged by 1961, despite the introduction of methicillin to combat β-lactamase.78 The mecA gene, which codes for PBP2a and decreases β-lactam binding, is the primary resistance mechanism of MRSA.79
Current resistance trends
Studies conducted in India have revealed high rates of resistance to co-trimoxazole, erythromycin, and penicillins.80-83 E. coli and Klebsiella frequently produce extended-spectrum β-lactamase (ESBL).84-86 Common pathogens are becoming more resistant to aminoglycosides, macrolides, and fluoroquinolones. Treatment is becoming more difficult in many areas because MRSA strains exhibit multidrug-resistance and possess the PVL toxin.
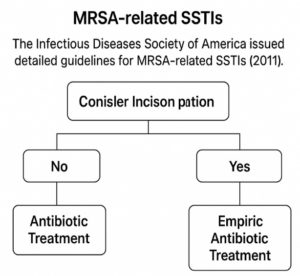
Current treatment guidelines
The Infectious Diseases Society of America published comprehensive guidelines for treating MRSA-related SSTIs.87-89 (Figure 6). These guidelines recommend using topical mupirocin (2%) for mild infections and incision and drainage alone for simple abscesses. Clindamycin, trimethoprim-sulfamethoxazole, doxycycline/minocycline, and linezolid are recommended as empirical treatments for purulent cellulitis caused by community-acquired MRSA. If non-purulent cellulitis does not improve after 48-72 hours, it is recommended to start with β-lactams and add MRSA coverage. Options for severe SSTIs that require hospitalisation include ceftaroline (active against MRSA and Gram-negative bacteria), vancomycin, linezolid, daptomycin, tigecycline, and telavancin. Although vancomycin has long been the main treatment for MRSA, other drugs such as linezolid, daptomycin, and glycopeptide derivatives have been developed because of delayed bacterial clearance and toxicity concerns.90-93
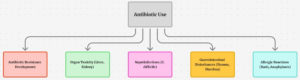
Adverse Effects of Antibiotic Treatment
Several adverse events are linked to antibiotic therapy (Figure 7). Clindamycin is linked to diarrhoea (particularly colitis caused by C. difficile); trimethoprim-sulfamethoxazole is associated with hyperkalaemia risk, particularly in older adults; long-term linezolid use can cause hematologic toxicity, neuropathy, and lactic acidosis; daptomycin is linked to GI problems, renal impairment, and muscle soreness; telavancin causes GI symptoms, taste abnormalities, and nephrotoxicity; ceftaroline is associated with electrolyte imbalance and increased liver enzymes; and fluoroquinolones are linked to GI distress and glycaemic abnormalities. Certain MRSA strains are less susceptible to vancomycin, which increases morbidity and causes treatment failure.94-100
Skin and soft tissue infections (SSTIs) continue to pose a significant clinical and public health burden worldwide. The prevalence of SSTIs varies widely, ranging from 77.5 per 1,000 person-years in the USA to 66% among hospitalized patients in resource-limited settings, highlighting the need for region-specific surveillance and management strategies. Methicillin-resistant Staphylococcus aureus (MRSA), particularly community-associated strains, remains a major contributor to morbidity, with prevalence rates ranging from 1%-74% across different countries.
Future research should focus on combating antimicrobial resistance (AMR) by investigating alternative therapeutic strategies, including novel antimicrobials, phage therapy, antimicrobial peptides, and immunomodulatory approaches. Additionally, studies evaluating the efficacy of preventive measures, rapid diagnostics, and stewardship interventions are crucial. Maintaining updated epidemiological data with standardized reporting of prevalence and resistance patterns will be essential to guide evidence-based clinical management and reduce the global burden of SSTIs.
ACKNOWLEDGMENTS
None.
FUNDING
None.
DATA AVAILABILITY
Not applicable.
ETHICS STATEMENT
This article does not contain any studies on human participants or animals performed by any of the authors.
- Di Gennaro F. Prescriptive appropriateness of dalbavancin in acute bacterial skin and skin structure infections in adults: an integrated approach between clinical profile, patient- and health system-related factors and focus on environmental impact. Front Antibiot. 2024;3:1405401.
Crossref - Parruti G, Polilli E, Coladonato S, et al. Safety and efficacy of Dalbavancin in real life: Retrospective analysis of a large monocentric case series of patients treated for Skin/Soft Tissue and other Difficult-to-Treat infections. Antibiotics. 2024;13(11):1063.
Crossref - De Vito A, Fiore V, Colpani A, et al. The current and future off-label uses of dalbavancin: a narrative review. Eur Rev Med Pharm Sci. 2023;27(3):1222-1238.
Crossref - Chong KJ, Feng H, Letchumanan V, et al. Tackling Microbial Resistance and Emerging Pathogens with Next-Generation Antibiotics. Progress In Microbes & Molecular Biology. 2024;7(1).
Crossref - Ahiskali A, Rhodes H. Oritavancin for the treatment of complicated gram-positive infection in persons who inject drugs. BMC Pharmacol Toxicol. 2020;21(73)
Crossref - Raza ZA, Giannini A, Bongiovanni M. Oritavancin for Gram-Positive Bone and Joint Infections: A Comprehensive Review of the Literature. Antibiotics. 2026;15(2):226.
Crossref - Velasco de la Fuente S, Fernández-Martinez M, Rodríguez Lozano J, Pablo-Marcos D, Siller M, Calvo J. In vitro activity of ceftobiprole and dalbavancin against a collection of coagulase-negative staphylococci isolates from clinical samples with reduced susceptibility to daptomycin and/or resistant to linezolid or glycopeptides. Enferm Infecc Microbiol Clin (Engl Ed). 2023;41(9):567-570.
Crossref - Scarano SM, Bruzzese E, Poeta M, Del Bene M, Guarino A, Lo Vecchio A. Dalbavancin for Acute Bacterial Skin and Skin Structure Infections in Pediatrics: Insights from Continuation Therapy Experience. Antibiotics. 2024;13(4):327.
Crossref - Poliseno M, Bavaro DF, Brindicci G, et al. Dalbavancin efficacy and impact on Hospital Length-of-Stay and treatment costs in different Gram-Positive bacterial infections. Clin Drug Investig. 2021;41(5):437-448.
Crossref - Iqbal K, Milioudi A, Wicha SG. Pharmacokinetics and pharmacodynamics of tedizolid. Clin Pharmacokinet. 2022;61(4):489-503.
Crossref - Zasheva A, Batcheva E, Ivanova KD, Yanakieva A. Differences in patient access to newly approved antibacterial drugs in EU/EEA countries. Antibiotics. 2024;13(11):1077.
Crossref - McCool R, Gould IM, Eales J, et al. Systematic review and network meta-analysis of tedizolid for the treatment of acute bacterial skin and skin structure infections caused by MRSA. BMC Infect Dis. 2017;17(1):39.
Crossref - Mngqibisa R, Fofanov O, Grazioso CF, et al. A Phase 3 Study of the Safety and Efficacy of Tedizolid Phosphate in Patients <12 Years of Age With Acute Bacterial Skin and Skin Structure Infections. Pediatr Infect Dis J. 2025;44(6):533-538.
Crossref - Athanasiou V, Ragias D, Tzikopoulou M, Zenios M, Falagas ME. Tedizolid for osteoarticular infections: Evaluation of the published evidence. Eur J Pharmacol. 2025;998:177458.
Crossref - Samajdar S, Chatterjee R, Mukherjee S, et al. Tedizolid: A comprehensive review of pharmacology, clinical applications and future directions in antimicrobial resistance. Afro-Egyptian Journal of Infectious and Endemic Diseases. 202515(3):242-246.
Crossref - Crocker J, Derrick C, Justo JA, et al. P-1102. Real-World Effectiveness of dalbavancin for invasive Gram-Positive bacterial infections. Open Forum Infect. 2025;12(Supplement_1).
Crossref - Baltin CT, Wulf C, Rongisch R, et al. Outpatient care concept and potential inpatient cost savings associated with the administration of dalbavancin – A real-world data and retrospective cost analysis. Journal of Infection and Public Health. 2023;16(6):955-963.
Crossref - Crapis M, Venturini S, Callegari A, et al. Usefulness of dalbavancin in early discharge and non-hospitalization. It’s time to throw your heart over the obstacle? Global & Regional Health Technology Assessment. 2024;11(Suppl. 2):18-21.
Crossref - Baiardi G, Caviglia MC, Piras F, et al. The clinical efficacy of multidose oritavancin: a systematic review. Antibiotics. 2023;12(10):1498.
Crossref - Moñino-Dominguez L, Aguado-Paredes A, Cordero-Ramos J. [Translated article] Therapeutic drug monitoring of dalbavancin: A systematic review of strategies and clinical applications in the treatment of complex infections. Farm Hosp. 2025;49(6):T396-T406.
Crossref - Currie BJ, Hoopes J, Cumming B. Cutaneous Larva Migrans Refractory to Therapy with Ivermectin: Case Report and Review of Implicated Zoonotic Pathogens, Epidemiology, Anthelmintic Drug Resistance and Therapy. Trop Med Infect Dis. 2025;10(6):163.
Crossref - Anastasio PJ, Wolthoff P, Galli A, Fan W. Single-Dose Oritavancin compared to standard of care IV antibiotics for acute bacterial skin and skin structure infection in the outpatient setting: a retrospective Real-World study. Infect Dis Ther. 2017;6(1):115-128.
Crossref - Bassetti M, Della Siega P, Pecori D, Scarparo C, Righi E. Delafloxacin for the treatment of respiratory and skin infections. Expert Opin Investig Drugs. 2015;24(3):433-442.
Crossref - Giordano PA, Pogue JM, Cammarata S. Analysis of pooled phase III efficacy data for delafloxacin in acute bacterial skin and skin structure infections. Clin Infect Dis. 2019;68(Supp_3):S223-S232.
Crossref - Diehl C, Pinzan CF, de Castro PA, Delbaje E, García Carnero LC, Sánchez-León E, Bhalla K, Kronstad JW, Kim D-g, Doering TL, Alkhazraji S, Mishra NN, Ibrahim AS, Yoshimura M, Vega Isuhuaylas LA, Pham LTK, Yashiroda Y, Boone C, Dos Reis TF, Goldman GH. Brilacidin, a novel antifungal agent against Cryptococcus neoformans. mBio. 2024;15(7):e0103124.
Crossref - Konwar AN, Hazarika SN, Bharadwaj P, Thakur D. Emerging Non-Traditional Approaches to Combat Antibiotic Resistance. Curr Microbiol. 2022;79(11):330.
Crossref - Ademi A, Qyra S, Dika-Haxhirexha F, Dogjani A, Biberaj P, Haxhirexha K. Contemporary Guidelines for the treatment and Prevention of Nosocomial Infections: Critical analysis and Practical Perspectives. Albanian Journal of Trauma and Emergency Surgery. 2025;9(2):1869-1876.
Crossref - Larson PJ, Zhou W, Santiago A, et al. Associations of the skin, oral and gut microbiome with aging, frailty and infection risk reservoirs in older adults. Nature Aging. 2022;2(10):941-955.
Crossref - Darlow CA, Parsons J, Lucy D, et al. Experience of use of dalbavancin for the treatment of unlicensed indications in a UK tertiary infectious diseases setting. Infection. 2025;53(6):2503-2509.
Crossref - Leong HN, Kurup A, Tan MY, Kwa ALH, Liau KH, Wilcox MH. Management of complicated skin and soft tissue infections with a special focus on the role of newer antibiotics. Infect Drug Resist. 2018;11:1959-1974
Crossref - Tuan JJ, Kayani J, Fisher A, Kotansky B, Dembry LM, Datta R. Clinical outcomes following dalbavancin administration in patients with barriers to outpatient parenteral antimicrobial therapy. Antimicrob Steward Healthc Epidemiol. 2022;2(1):e83.
Crossref - Zhang W, Liu F, Li J, Zhang L, Hu T. Surveillance of Antibiotic Resistance and Molecular Epidemiology of Staphylococcus aureus in Baotou, Inner Mongolia, China. Infect Drug Resist. 2025;18:4887-4900
Crossref - Scoble PJ, Reilly J, Tillotson GS. Real-World Use of Oritavancin for the Treatment of Osteomyelitis. Drugs Real World Outcomes. 2020;7(Suppl 1):46-54.
Crossref - Antosz K, Al-Hasan MN, Lu ZK, et al. Clinical Utility and Cost Effectiveness of Long-Acting Lipoglycopeptides Used in Deep-Seated Infections among Patients with Social and Economic Barriers to Care. Pharmacy. 2021;10(1):1.
Crossref - Machado E, Sousa JC. New antibiotics for treating infections caused by Multidrug-Resistant bacteria. Antibiotics. 2025;14(10):997.
Crossref - Saka B, Kassang P, Gnossike P, et al. Prevalence of skin Neglected Tropical Diseases and superficial fungal infections in two peri-urban schools and one rural community setting in Togo. PLoS Negl Trop Dis. 2022;16(12): e0010697.
Crossref - Vella V, Derreumaux D, Aris E, et al. The Incidence of Skin and Soft Tissue Infections in the United States and Associated Healthcare Utilization Between 2010 and 2020. Open Forum Infect Dis. 2024;11(6):ofae267.
Crossref - Lwigale F, Kibombo D, Kasango SD, et al Prevalence, resistance profiles and factors associated with skin and soft-tissue infections at Jinja regional referral hospital: A retrospective study. PLOS Glob Public Health. 2024;4(8):e0003582.
Crossref - Toschi A, Giannella M, Viale P. Recurrence of skin and soft tissue infections: identifying risk factors and treatment strategies. Curr Opin Infect Dis. 2025;38(2):71-77.
Crossref - De Rosa FG, Lupia T, Fornari V, et al. Medical source control with oritavancin in complicated gram-positive bacterial infections: A case series. Int j Infect Dis. 2025;161:108111.
Crossref - Cooper MM, Preslaski CR, Shihadeh KC, Hawkins KL, Jenkins TC. Multiple-Dose dalbavancin regimens as the predominant treatment of Deep-Seated or endovascular infections: A scoping review. Open Forum Infect Dis. 2021;8(11):ofab486.
Crossref - Maraki S, Mavromanolaki VE, Stafylaki D, Iliaki-Giannakoudaki E, Hamilos G. In Vitro Activities of Ceftobiprole, Dalbavancin, Tedizolid and Comparators against Clinical Isolates of Methicillin-Resistant Staphylococcus aureus Associated with Skin and Soft Tissue Infections. Antibiotics. 2023;12(5):900.
Crossref - Geremia N, Giovagnorio F, Colpani A, et al. Fluoroquinolones and Biofilm: A narrative review. Pharmaceuticals. 2024;17(12):1673.
Crossref - Salvatore D, Moore J, Steuber TD. Long-acting lipoglycopeptides compared to standard-of-care for the treatment of complicated gram-positive infections: A systematic review and meta-analysis. Int J Antimicrob Agents. 2025;66(5):107581.
Crossref - Pontali E, Baiardi G, Del Puente F, Mattioli F. Long-Acting Antibiotics: New opportunities beyond acute bacterial skin and skin structure infections (ABSSSIs)! Antibiotics. 2025;14(2):164.
Crossref - Lan SH, Lai CC, Lu LC, Chang SP, Huang HT. Efficacy and safety of delafloxacin in the treatment of acute bacterial skin and skin structure infections: a systematic review and meta-analysis of randomized controlled trials. Infect Drug Resist. 2019;12:1415-1423
Crossref - Amara S, Adamson RT, Lew I, Huang X. Clinical response at Day 3 of therapy and economic outcomes in hospitalized patients with acute bacterial skin and skin structure infection (ABSSSI). Curr Med Res Opin. 2013;29(7):869-877.
Crossref - Ashraf H, Nadeem ZA, Rehman KA, et al. Safety and Efficacy of Outpatient Parenteral Antibiotic therapy (OPAT) in patients with infective endocarditis: A Systematic Review and Meta-Analysis. Clin Cardiol. 2025;48(5):e70147.
Crossref - Hussein M, Barclay J, Baker M, et al. A Comparative review of the pharmacology of dalbavancin and oritavancin for Gram-Positive Infections: Birds of a Feather or Apples and Oranges? Infect Dis Ther. 2025;14(10):2221-2246.
Crossref - LoVecchio F, McCarthy MW, Ye X, et al. Single intravenous dose Dalbavancin pathway for the treatment of acute bacterial skin and skin structure infections: Considerations for emergency department implementation and cost savings.
J Emerg Med. 2024;67(2):e217-e229.
Crossref - Harkins CP, Pichon B, Doumith M, et al. Methicillin-resistant Staphylococcus aureus emerged long before the introduction of methicillin into clinical practice. Genome Biol. 2017;18(1):130.
Crossref - Moran GJ, Krishnadasan A, Gorwitz RJ, et al. Methicillin-Resistant S. aureus Infections among Patients in the Emergency Department. N Eng J Med. 2006;355(7):666-674.
Crossref - Changchien CH, Chen SW, Chen YY, et al. Antibiotic susceptibility and genomic variations in Staphylococcus aureus associated with Skin and Soft Tissue Infection (SSTI) disease groups. BMC Infect Dis. 2016;16:276.
Crossref - Belev N, Tein A, Mangialardi G, et al. A randomized, Observer-Blinded, Active-Controlled, Phase IIIB study to compare IV/Oral Delafloxacin Fixed-Dose monotherapy with best available treatments in a microbiologically enriched population with surgical site infections: the DRESS Study. Open Forum Infect Dis. 2025;12(9):ofaf476.
Crossref - Huband MD, Fedler KA, Mendes RE, et al. Surveillance of omadacycline against 35,000 bacterial clinical isolates from the United States (2019-2023). Diagn Microbiol Infect Dis. 2025;111(3):116711.
Crossref - Liang W, Yin H, Chen H, Xu J, Cai Y. Efficacy and safety of omadacycline for treating complicated skin and soft tissue infections: a meta-analysis of randomized controlled trials. BMC Infect Dis. 2024;24(1):219.
Crossref - Kong WQ, Shu YF, Tang J, Li SQ, Zhong H, Zhang XY. Omadacycline for the treatment of acute bacterial skin and skin structure infections: a systematic review and network meta-analysis. BMC Infect Dis. 2025;25(1):1460.
Crossref - Rodriguez GD, Warren N, Yashayev R, Chitra S, Amodio-Groton M, Wright K. Intravenous Versus Oral Omadacycline or Linezolid for Acute Bacterial Skin and Skin Infections: A post hoc Analysis of the OASIS Trials. Infect Dis Ther. 2024;13(12):2637-2648.
Crossref - Gao W, Yang J, Zhang X, et al. Omadacycline for diverse infections in China: A Real-World Analysis of Efficacy and safety. Infect Dis Ther. 2024;13(12):2509-2526.
Crossref - File TM, Ramirez JA, Wilde AM. New Perspectives on Antimicrobial Agents: Omadacycline for community-acquired pneumonia, skin and soft tissue infections, and nontuberculous mycobacteria (focus on M. abscessus). Antimicrob Agents Chemother. 2025;69(2):e0108724.
Crossref - Carbonara S. The efficacy of dalbavancin and impact on hospitalization and treatment costs in patients with ABSSSI. Glob Reg Health Technol Assess. 2024;11(Suppl. 2):13-17.
Crossref - Rappo U, Puttagunta S, Shevchenko V, et al. Dalbavancin for the treatment of osteomyelitis in adult patients: multicenter experience. Open Forum Infect Dis. 2022;6(1):ofy331.
Crossref - Treu CN, Stilwell AM, Cheon E, Acquisto NM. Use of lipoglycopeptides for moderate to severe ABSSSI in the emergency department. Am J Emerg Med. 2024;87:44-50.
Crossref - Chang AJ, Mannix MK, Kenney PO, et al. Case Series: Use of Dalbavancin for Invasive Gram-Positive Infection in Children. J Pediatr Pharmacol Ther. 2026;31(1):88-101.
Crossref - Cattaneo D, Fusi M, Colaneri M, Fusetti C, Genovese C, Giorgi R, Matone M, Merli S, Petri F, Gori A. Therapeutic Drug Monitoring of Dalbavancin in Real Life: A Two-Year Experience. Antibiotics. 2023;13(1):20.
Crossref - Tran TT, Villegas SG, Aitken SL, et al. New perspectives on antimicrobial agents: Long-Acting Lipoglycopeptides. Antimicrob Agents Chemother. 2022;66(6):e0261420.
Crossref - Bongiovanni M, Thoueille P, Barda B, et al. Oritavancin use in patients with recurrent bone infections by methicillin-resistant Staphylococcus aureus with monitoring of concentrations. Eur J Clin Microbiol Infect Dis. 2024;43(7):1503-1504.
Crossref - Załęska-Kocięcka M, Mielczarek S, Góral P, et al. Oritavancin in LVAD related infections – a chance for shortening therapy and improving outcomes. BMC Infect Dis. 2025;25(1):1358.
Crossref - Deck DH, Jordan JM, Holland TL, et al. Single-Dose Oritavancin Treatment of Acute bacterial skin and skin structure infections: SOLO Trial Efficacy by Eron Severity and Management Setting. Infect Dis Ther. 2016;5(3):353-361.
Crossref - Linz MS, Mattappallil A, Finkel D, Parker D. Clinical Impact of Staphylococcus aureus Skin and Soft Tissue Infections. Antibiotics. 2023;12(3):557.
Crossref - Lodise TP, Gunter K, Mu F, et al. Real-world effectiveness of omadacycline and impact of unapproved omadacycline prescription claims among adult outpatients with community-acquired bacterial pneumonia or acute bacterial skin and skin structure infections. J Manag Care Spec Pharm. 2023;29(8):952-964.
Crossref - Bhavnani SM, Zhang L, Ambrose PG, Flamm RK, Cammarata SK, Rubino CM. Population Pharmacokinetic (PPK) and Pharmacokinetic-Pharmacodynamic (PK-PD) Target Attainment Analyses for Delafloxacin to Support Dose Selection for the Treatment of Patients with Acute Bacterial Skin and Skin Structure Infections (ABSSSI). Open Forum Infect Dis. 2017;4(suppl_1):S530.
Crossref - Gulyás D, Kamotsay K, Szabó D, Kocsis B. Investigation of Delafloxacin Resistance in Multidrug-Resistant Escherichia coli Strains and the Detection of E. coli ST43 International High-Risk Clone. Microorganisms. 2023;11(6):1602.
Crossref - Gavaruzzi F, Granata G, Capone A, Chinello P, Cicalini S. Long-Term Therapy with Long-Acting Lipoglycopeptide Antibiotics in the Treatment of Cardiovascular Prosthetic Infections: A Systematic Review. Antibiotics. 2025;14(11):1130.
Crossref - Sivori F, Cavallo I, Kovacs D, et al. Role of Extracellular DNA in Dalbavancin Activity against Methicillin-Resistant Staphylococcus aureus (MRSA) Biofilms in Patients with Skin and Soft Tissue Infections. Microbiology Spectrum. 2022;10(2):e0035122.
Crossref - Abdelsattar AS, Abutaleb NS, Seleem MN. A novel peptide mimetic, brilacidin, for combating multidrug-resistant Neisseria gonorrhoeae. PLoS ONE. 2025;20(6):e0325722.
Crossref - Wu Y, Liu T, Jin L, Wang C, Zhang D. Antimicrobial peptides for skin wound healing. Biomolecules. 2025;15(11):1613.
Crossref - Donà D, Barbieri E, Brigadoi G, et al. State of the art of antimicrobial and diagnostic stewardship in pediatric setting. Antibiotics. 2025;14(2):132.
Crossref - Carter LL, Yu MA, Sacks JA, et al. Global genomic surveillance strategy for pathogens with pandemic and epidemic potential 2022-2032. Bull World Health Organ. 2022;100(4):239-239A.
Crossref - Biggs HM, Li R, Jackson KA, et al. Trends in Incidence and Epidemiology of Methicillin-Resistant Staphylococcus aureus Bacteremia, Six Emerging Infections Program Surveillance Sites, 2005–2022. Open Forum Infect Dis. 2025;12(6):ofaf282.
Crossref - Almogbel Y, Mobark MA, Almutairi MS, et al. The appropriateness of empirical uses of antibiotics based on antimicrobial susceptibility results for inpatients at a tertiary hospital in Saudi Arabia. Antibiotics. 2024;13(9):824.
Crossref - Steuber TD, Gipson H, Boyett B, Belk M, Thayer B, Edwards J. Head-to-head comparison of multi-dose oritavancin and dalbavancin for complicated infections: A propensity score-matched analysis. Int J Antimicrob Agents. 2024;63(6):107165.
Crossref - Jordan H, Kozierowski K, Pickles R, Davis JS. Oritavancin for the Treatment of Staphylococcus aureus Bacteremia—A Retrospective Single-arm Cohort Study. Open Forum Infect Dis. 2025;12(7):ofaf333.
Crossref - Chaldoupis AE, Petrakis V, Ioannou P, et al. Dalbavancin for infective endocarditis: A systematic review of effectiveness, safety, and dosing. Eur J Clin Microbiol Infect Dis.
Crossref - Radakovich N, Prasad P, Escobar D, et al. A machine learning model to predict optimal antibiotic use in hospital medicine patients. Antimicrob Steward Healthc Epidemiol. 2025;5(1):e238.
Crossref - Zurawski DV, Serio AW, Black C, et al. A review of omadacycline for potential utility in the military health system for the treatment of wound infections. Mil Med. 2023;189(5-6):e1353-e1361.
Crossref - Petropoulou C, Ioannou P, Eleftherakis G, et al. Dalbavancin for bone and joint infections: A Two-Center Greek Real-World retrospective study. Pathogens. 2025;14(11):1109.
Crossref - Kubicskó A, Kamotsay K, Banczerowski P, Sipos L, Szabó D, Kocsis B. Variable In Vitro Efficacy of Delafloxacin on Multidrug-Resistant Pseudomonas aeruginosa and the Detection of Delafloxacin Resistance Determinants. Antibiotics. 2025;14(6):542.
Crossref - Russo A, Trecarichi EM, Torti C. The role of Gram-negative bacteria in skin and soft tissue infections. Curr Opin Infect Dis. 2021;35(2):95-102.
Crossref - Al Jalali V, Zeitlinger M. Clinical Pharmacokinetics and Pharmacodynamics of Telavancin Compared with the Other Glycopeptides. Clin Pharmacokinet. 2018;57(7):797-816.
Crossref - Orlikowski C, Torney N, Hunt A, Britton W, Nichols C. P-758. Single-Dose long-acting lipoglycopeptide Clinical Pathway for treatment of cellulitis in the emergency Department: A how-to guide based on Real-World implementation at a community hospital health system. Open Forum Infect Dis. 2026;13(Supp_1).
Crossref - Texidor WM, Miller MA, Molina KC, et al. Oritavancin as sequential therapy for Gram-positive bloodstream infections. BMC Infect Dis. 2024;24(1):127. Doi: 10.1186/s12879-023-08725-8
- De Pablo-Miró M, Pujol-Ruiz S, Iftimie S, et al. Comparative Analysis of Dalbavancin versus Other Antimicrobial Options for Gram-Positive Cocci Infections: Effectiveness, Hospital Stay and Mortality. Antibiotics. 2021;10(11):1296.
Crossref - Bassetti M, Magnè F, Giacobbe DR, Bini L, Vena A. New antibiotics for Gram-negative pneumonia. Eur Respir Rev. 2022;31(166):220119.
Crossref - Bai F, Mazzitelli M, Silvola S, et al. Cost analysis of dalbavancin versus standard of care for the treatment of acute bacterial skin and skin structure infections (ABSSSIs) in two Italian hospitals. JAC-Antimicrobial Resistance. 2023;5(2).
Crossref - Prokocimer P, De Anda C, Fang E, Mehra P, Das A. Tedizolid Phosphate vs Linezolid for Treatment of Acute Bacterial Skin and Skin Structure Infections. JAMA. 2013;309(6):559.
Crossref - Sato VAH, Neves PDMM, Nunes RAB, Grassioli L, Piastrelli F. Delafloxacin as a New Therapeutic Tool for the Treatment of Skin and Soft Tissue Infections: Experience in a Single Center. Preprint. Research Square.
Crossref - John CM, Otala SA, Jarvis GA. Cyclization increases bactericidal activity of arginine-rich cationic cell-penetrating peptide for Neisseria gonorrhoeae. Microbiol Spectr. 2024;12(9):e0099724.
Crossref - Rahman A, Debnath T, Kundu D, et al. Machine learning and deep learning-based approach in smart healthcare: Recent advances, applications, challenges and opportunities. AIMS Public Health. 2024;11(1):58-109.
Crossref - Athanasakis K, Petrakis I, Ollandezos M, et al. Antibacterial Treatment of Meticillin-Resistant Staphylococcus aureus Complicated Skin and Soft Tissue Infections: a Cost and Budget Impact Analysis in Greek Hospitals. Infect Dis Ther. 2014;3(2):257-268.
Crossref
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.