ISSN: 0973-7510
E-ISSN: 2581-690X
Quick and reliable detection of bacterial species causing bovine sub-clinical mastitis is pivotal in determination of the antimicrobial treatment to minimize economic losses and ensure re-establishment of cattle health. The performance of conventional bacterial culture examination and real-time PCR based assay while screening cattle for sub-clinical mastitis is evaluated. 156 quarter milk samples (qms) (103 from sub-clinically affected (SCM) quarters and 53 from healthy quarters) were simultaneously subjected to PathoProof mastitis PCR assay and bacterial culture examination. On bacteriological culture examination, 93 (90.29 %) out of 103 qms from SCM quarters showed positive results, revealing either the presence of 1, 2 or 3 bacterial species. However, PathoProof mastitis PCR assay showed presence of one or more bacterial species in all 103 SCM quarters. From healthy quarters, out of a total of 53 qms, 8 (15.09 %) were positive bacteriologically and by PathoProof mastitis PCR assay. Out of remaining 45 (84.91 %) qms, 42 (79.25 %) which showed negative results on bacterial culture showed presence of one or more bacterial species on PathoProof mastitis PCR assay. Overall, in 18 (11.54 %) out of 156 milk samples from healthy and sub-clinically mastitic quarters, both techniques provided identical results. In 78 (50%) qms, PathoProof mastitis PCR assay detected the species identified by bacterial culture, however, it also detected one or more additional species. In 8 (5.13 %) qms, both tests identified different species, while in 52 (33.33 %) qms which were negative on bacterial culture, PathoProof mastitis PCR assay detected one or more species. Thus, common mastitis causing bacteria may be present in large numbers, but are not detected by conventional bacterial culturing. The real-time PCR kit exemplified the advantages over conventional culturing in terms of speed, automated interpretation and increased sensitivity. Hence, this kit proves to be an indispensible parallel component in identification of pathogens. Quick and reliable identification of mastitis causing microorganism is central in management of disease and deciding the treatment with antimicrobials. In future, a similar kit can be designed which is affordable to dairy farmers across the world.
Real-time PCR; pathoproof mastitis PCR assay; bovine sub-clinical mastitis; bacteriological culture examination.
Bovine mastitis, intramammary infection (IMI), leads to significant decrease in milk quality and yield. It affects the cattle health and poses a major economical setback to dairy farmers and industry worldwide. Intramammary infections may translate into clinical mastitis whose manifestations are observed locally and systemically along with change in milk appearance. Alternatively, the infection may remain sub-clinical, where the animal does not show any clinical signs, but the SCC of milk increases (IDF 1999).
Reliable and timely detection of causative bacterial species is pivotal in disease management and formatting further line of treatment. Bacteriological culture examination has been the benchmark for detection of mastitis for several years (National Mastitis Council, 1996, 1999, 2004). Conventional culture encompasses incubation for 24 to 48 hours. Timely diagnosis of mastitis is crucial, since delay in detection expounds into increase in total duration of treatment or employing broad-spectrum antimicrobials (Pyorala 2002; Barkema et al. 2006). Additionally, the cultural examination has intrinsic challenges in interpreting the observations. Accurate bacteriological diagnosis of clinical samples is inevitable in veterinary medicine which entails to specify the analytical methods to be opted and to have information on the general performance of the laboratories. Pitkala et al. (2005) reported that the overall performance of the laboratories varied from 63 to 93% in different testing rounds. “No growth” samples have been reported to range from 25 to 45% of all milk samples even after 48 h of incubation (Makovec and Ruegg, 2003; Bradley et al. 2007; Koivula et al. 2007).
IMI diagnostics can become reliable with application of molecular techniques. Real-time PCR provides an edge in diagnostics, since it reduces the risk of cross-contaminations in the laboratories (Bustin, 2004). Developing an alternative technique for replacing conventional methods in mastitis can be attained only if the test is able to distinctly identify the causative bacterial species of IMI, since they are closely genetically related. Normally milk contains PCR-inhibiting substances. Hence a commercially available PCR-based mastitis tests, known as PathoProof Mastitis PCR Assay was developed by Finnzymes Oy, Finland. The assay requires raw milk without bacterial culturing and it identifies 11 mastitis causing bacterial species or species groups and the ²–lactamase gene (the blaZ gene) responsible for staphylococcal penicillin resistance.
Since the sensitivity of the real-time PCR assay is independent of the viability of the bacteria to grow in culture, it does not lead to false-positive detection thereby making it an optimal tool to study the bacteriological etiology of the no-growth milk samples (Koskinen et al. 2009). The current study evaluates performance between the bacterial culturing and real-time based identification of bacterial species from quarter milk samples (qms) of healthy and subclinical mastitic cattle.
Screening of Udder Quarters for Sub-Clinical Mastitis
Animal Distribution and Milk Collection
Bovine milk samples were collected from 69 cows comprising of 35 triple cross (Kankrej x Jersey x Holstein Friesian), 26 Kankrej and 8 Gir maintained at Livestock Research Station (LRS), Anand Agricultural University, Anand. These animals were housed, fed and managed under identical management practices as per the schedule followed at the LRS. Clinical examination of individual quarter of each animal included in this study was carried out for abnormalities in the shape and size of udder, udder oedema or atrophy, consistency i.e. lumps, fibrosis or hardening of udder. Similarly, each quarter milk sample was checked at the time of milking for any gross abnormality in the milk. On the basis of clinical examination of udder as well as gross examination of milk, only those animals having apparently healthy quarters were selected for the present investigation.
The milk samples were collected in sterile screw-capped vials from each quarter of individual cow for three consecutive days. Before the collection of milk samples, udder and teats were washed properly with 0.01 per cent potassium permanganate solution and teat orifices were scrubbed with 70 per cent ethanol. After discarding first 2-3 streams of milk, foremilk samples were collected from each quarter during the milking time.
Microbiological Culture Examination
Loopful of milk from foremilk sample from each quarter was streaked on blood agar plate (containing five per cent sheep blood) and simultaneously on MacConkey agar plate for primary bacterial isolation and the plates were incubated at 37 °C for 24 hours. Following incubation, the plates were examined for bacterial growth and the morphological characteristics of bacterial colonies were recorded. Characterization of bacterial isolates was performed as per the method described by Cowan and Steel (1970). The pure culture of each isolate was identified on the basis of morphological, cultural and biochemical characteristics.
Somatic Cell Count
Somatic cell count (SCC) of foremilk samples was carried out by using FossomaticTM minor cell counter (Foss Electric, Hillerod, Denmark) as per the method described by Gonzalo et al. (2003). The SCC value more than 500 cells/µl of milk indicated the presence of sub-clinical mastitis.
Identification of Infected Quarters
By correlating the results of SCC and bacteriological culture examination, infected quarters were identified following the Indian Dairy Federation (IDF) guidelines.
Distribution of Milk Samples
156 quarter milk samples (qms) were collected for 3 consecutive days and subjected to somatic cell count and bacteriological culture examination to categorize the quarters as healthy or sub-clinically infected (IDF, 1987).
Based on the results of screening, 103 out of 156 qms were categorized as from sub-clinically infected quarters, whereas 53 qms were categorized as from healthy quarters. All 156 qms were simultaneously subjected to PathoProof mastitis PCR assay, whereas the isolates recovered on bacterial culture were subjected to biochemical tests for genus and species level identification.
Detection of Mastitis Pathogen by Real-Time PCR Assay
The milk samples were simultaneously subjected to bacteriological culture examination and a commercial real-time PCR test kit (Pathoproof Mastitis PCR Assay, Thermo Fisher Scientific).
DNA was extracted from the milk samples and the PCR conditions were performed using the reagents and protocol instructions included in the manufacturer’s user manual. The kit protocol involved 4 separate multiplex real-time PCR reactions, which targeted 11 bacterial species and groups (covering more than 25 mastitis-causing species in total): Staphylococcus aureus, Staphylococcus spp. (including Staph. aureus and all relevant CNS), Enterococcus spp. (including Enterococcus faecalis and Enterococcus faecium), Corynebacterium bovis, Escherichia coli, Streptococcus dysgalactiae, Streptococcus agalactiae, Streptococcus uberis, Arcanobacterium pyogenes/Peptoniphilus indolicus, Klebsiella spp. (including Klebsiella oxytoca and Klebsiella pneumoniae), and Serratia marcescens.
The assay included internal amplification controls for confirmation of acceptable PCR reaction conditions. A negative control (distilled water) was included in every PCR run to control for cross-contamination in the laboratory. Additional protocol details were identical to those described in Koskinen et al. 2009.
Comparison of the Tests
All comparisons of bacterial culture and real-time PCR results were based on a double-blinded experimental design; that is, the culture results were not available to the PCR laboratory, and vice versa, before all laboratory work was finished.
Distribution of Total Number of Quarter Milk Samples
A total of 156 qms included in the present study were categorized as per IDF guidelines (IDF, 1987) as 103 from sub-clinically affected (SCM) quarters and 53 from healthy quarters. Based on the same guidelines, the 103 SCM quarters were divided as quarters with specific mastitis (SM), non-specific mastitis (NSM) and latent infection (LI). Number of quarters affected with SM, NSM and LI were 65, 8 and 30, respectively (Fig. 1). Subsequently, all these samples were subjected to bacteriological culture examination as well as PathoProof mastitis PCR assay.
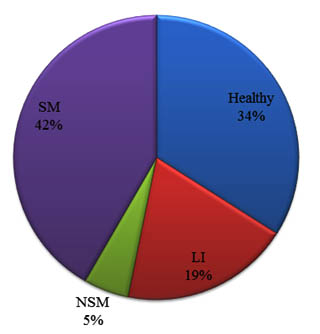
Fig. 1. Health status of quaters selected for collection of milk samples for bacterial culture and Pathoproof mastitis PCR assay
Milk samples from sub-clinically infected quarters
Bacterial culture detected 1, 2, 3/or more than 3 bacterial species in 69 (66.99 %), 22 (21.36 %) and 2 (1.94 %) SCM quarters, respectively. In contrast, PathoProof mastitis PCR assay detected 1, 2, 3/or more than 3 bacterial species in 15 (14.56 %), 8 (7.77 %) and 80 (77.67 %) SCM quarters, respectively (Table 1).
On bacteriological culture examination, 93 (90.29 %) out of 103 qms from SCM quarters showed positive results, revealing either the presence of 1, 2 or 3 bacterial species. However, PathoProof mastitis PCR assay showed presence of one or more bacterial species in all 103 SCM quarters. Therefore, all the qms from mastitic quarters were found positive on PathoProof mastitis PCR assay (Table 1).
Table (1):
Number of samples for which bacteriological culture (BC) and PathoProof mastitis PCR assay provided negative results, or identified one or more bacterial species
| Species Detected (n) | Samples from Sub-clinical Mastitis (n=103) | Samples from Healthy Quarters (n=53) | ||
|---|---|---|---|---|
| BC | Pathoproof PCR Assay | BC | Pathoproof PCR Assay | |
| 0 | 10 | 0 | 45 | 3 |
| 1 | 69 | 15 | 6 | 8 |
| 2 | 22 | 8 | 2 | 18 |
| ≥3 | 2 | 80 | 0 | 24 |
Both bacterial culture and PathoProof mastitis PCR assay detected Staphylococcus sp., Staphylococcus aureus, Streptococcus agalactiae, Escherichia coli and Klebsiella sp. in 82 (79.61 %), 17 (16.50 %), 2 (1.94 %), 1 (0.97 %) and 1 (0.97 %) qms, respectively (Table 4). Among SCM quarters, some quarters showed growth of bacteria on culture, however, PathoProof mastitis PCR assay could not detect the bacterial species. The negative results were obtained for Staphylococcus sp., Escherichia coli, Klebsiella sp., Serratia marcescens and Streptococcus agalactiae in 3 (2.91 %), 2 (1.94 %), 1 (0.97 %), 1 (0.97 %) and 1 (0.97 %) milk samples, respectively (Table 4).
In the present investigation, the PathoProof mastitis PCR assay detected additional bacterial species in SCM quarters other than those observed on bacterial culture (Table 4). For example, the PathoProof Mastitis PCR assay detected Staphylococcus aureus in 40 and Streptococcus agalactiae in 35 qms respectively from SCM quarters that were negative for these organisms on bacteriological culture (Table 4). Bacterial species unable to grow on culture but found positive by PathoProof mastitis PCR assay were Arcanobacterium pyogenes/ Peptoniphilus indolicus, Corynebacterium bovis, Escherichia coli, Enterococcus sp., Klebsiella sp., Staphylococcus aureus, Staphylococcus sp., Serratia marcescens, Streptococcus agalactiae, Streptococcus dysgalactiae and Streptococcus uberis which were detected in 4 (3.88 %), 55 (53.40 %), 40 (38.83 %), 17 (16.50 %), 7 (6.80 %), 40 (38.83 %), 14 (13.59 %), 8 (7.77 %), 35 (33.98 %), 5 (4.85 %) and 29 (28.16 %) SCM quarters, respectively (Table 4).
Milk Samples from Healthy Quarters
Out of a total of 53 qms from healthy quarters, 8 (15.09 %) were bacteriologically positive and showed presence of 1 and 2 bacterial species in 6 (11.32 %) and 2 (3.77 %) qms respectively. These qms were also found positive on PathoProof mastitis PCR assay (Table 1). Out of remaining 45 (84.91 %) qms from healthy quarters, which showed negative results on bacterial culture, 42 (79.25 %) showed presence of one or more bacterial species on PathoProof mastitis PCR assay (Table 1). Three qms from healthy quarters did not show presence of bacteria on PathoProof PCR assay.
PathoProof mastitis PCR assay detected presence of 1, 2, 3/or more than 3 bacterial species in 8 (15.09 %), 18 (33.96 %) and 24 (45.28 %) qms from healthy quarters, respectively (Table 1). The 42 qms showed presence of Staphylococcus sp., Staphylococcus aureus, Corynebacterium bovis, Escherichia coli, Enterococcus sp., Klebsiella sp., Streptococcus agalactiae, Streptococcus dysgalactiae and Streptococcus uberis in 31 (73.81 %), 10 (23.81 %), 21 (50 %), 12 (28.57 %), 6 (14.29 %), 7 (16.67 %), 7 (16.67 %), 1 (2.38 %) and 8 (19.05 %) samples, respectively (Table 3).
Milk samples from sub-clinically infected quarters showing no-growth on bacterial culture but found positive on PathoProof Mastitis PCR Assay
In the present investigation, 10 (9.71 %) out of 103 SCM quarters did not show growth on blood agar. However, these qms were positive for bacterial species on PathoProof mastitis PCR assay with detection of Staphylococcus aureus, Staphylococcus sp., Corynebacterium bovis, Serratia marcescens, Streptococcus agalactiae, Streptococcus uberis, Enterococcus sp. and Escherichia coli in 8 (80 %), 7 (70 %), 4 (40 %), 2 (20 %), 2 (20 %) 2 (20 %), 1 (10 %) and 1 (10 %) qms, respectively (Table 2).
Table (2):
Bacterial species found in the sub-clinical mastitis samples that provided negative results in culture, but positive results using the PathoProof mastitis PCR assay
Sr. No. |
Species Detected |
No. of Samples |
|---|---|---|
1) |
Staphylococcus sp. |
7 |
2) |
Staphylococcus aureus |
8 |
3) |
Enterococcus sp. |
1 |
4) |
Corynebacterium bovis |
4 |
5) |
Escherichia coli |
1 |
6) |
Serratia marcescens |
2 |
7) |
Streptococcus agalactiae |
2 |
8) |
Streptococcus uberis |
2 |
Table (3):
Bacterial species found in the healthy samples that provided negative results in culture, but positive results using the PathoProof mastitis PCR assay
Sr. No. |
Species Detected |
No. of Samples |
|---|---|---|
1) |
Staphylococcus sp. |
31 |
2) |
Staphylococcus aureus |
10 |
3) |
Corynebacterium bovis |
21 |
4) |
Escherichia coli |
12 |
5) |
Enterococcus sp. |
6 |
6) |
Klebsiella sp. |
7 |
7) |
Streptococcus agalactiae |
7 |
8) |
Streptococcus dysgalactiae |
1 |
9) |
Streptococcus uberis |
8 |
In the present study, 10 (9.71 %) out of 103 SCM quarters, did not show growth on blood agar (Table 1). From these, 1 sample showed presence of one organism, four samples showed presence of two organisms, three samples showed presence of three organisms and two samples showed presence of more than three samples. In the present investigation, 52 (33.33 %) qms from SCM quarters showed presence of one or more bacterial species by PathoProof mastitis PCR assay which failed to grow on conventional bacterial culture (Table 5).
Overall, in current investigation, it was observed that PathoProof mastitis PCR assay detected bacteria in 100 per cent SCM quarters against 90.29 % by conventional bacterial culture.
Detection of mastitis pathogens by bacterial culture and Real-Time PCR from QMS indicating compatibility between the two techniques
In the present study, in most of the cases PathoProof mastitis PCR assay identified species detected by culture examination, however, there were a large number of qms wherein PCR detected additional species not identified on culture (Table 4). In 15 (9.62 %) qms, there was same species identification by both the techniques whereas in 3 qms, both tests provided negative results. Therefore, in 18 (11.54 %) out of 156 milk samples from healthy and sub-clinically mastitic quarters, both techniques provided identical results (Table 5). In 78 (50%) qms, PathoProof mastitis PCR assay detected the species identified by bacterial culture, however, it also detected one or more additional species (Table 5). In 8 (5.13 %) qms, both tests identified different species, while in 52 (33.33 %) qms which were negative on bacterial culture, PathoProof mastitis PCR assay detected one or more species. Our study did not have a single sample that showed positive result on bacterial culture but produced negative result on PathoProof mastitis PCR assay (Table 5).
Table (4):
The total number of bacteria found in bacterial culture (BC) and in PathoProof mastitis PCR assay across the 103 milk samples from sub-clinically mastitic quarters
Microorganism |
BC positive / PCR positive |
BC positive / PCR negative |
BC negative / PCR positive |
BC negative / PCR negative |
|---|---|---|---|---|
Arcanobacterium pyogenes/ Peptoniphilus indol-icus |
0 |
0 |
4 |
99 |
Corynebacterium bovis |
0 |
0 |
55 |
49 |
Escherichia coli |
1 |
2 |
40 |
61 |
Enterococcus sp. |
0 |
0 |
17 |
86 |
Klebsiella sp. |
1 |
1 |
7 |
94 |
Staphylococcus aureus |
17 |
0 |
40 |
46 |
Staphylococcus sp. |
82 |
3 |
14 |
5 |
Serratia marcescens |
0 |
1 |
8 |
94 |
Streptococcus agalactiae |
2 |
1 |
35 |
66 |
Streptococcus dysgalactiae |
0 |
0 |
5 |
98 |
Streptococcus uberis |
0 |
0 |
29 |
74 |
In addition to the species listed, culture identified 2 more bacterial species that were not targeted or directly comparable by the PathoProof PCR mastitis assay. These included Micrococcus varians (n=2) and Enterobacter sp. (n=1).
Table (5):
Detection of mastitis pathogens in quarter milk samples (qms) by bacterial culture (BC) and real-time PCR (qRT-PCR) indicating compatibility between the two techniques (n=156)
Identical species detection by both tests (culture and qRT-PCR) |
Negative result by both tests (culture and qRT-PCR) |
qRT-PCR identified one or more additional species |
Detection of different species by both tests, no common identification |
Negative results by culture but detection of one or more species by qRT-PCR |
Bacterial culture positive but qRT-PCR negative results |
|
|---|---|---|---|---|---|---|
No. of samples (X) |
15 |
3 |
78 |
8 |
52 |
0 |
Per cent (X/156*100) |
9.62 |
1.92 |
50 |
5.13 |
33.33 |
0 |
For the management of bovine mastitis, which otherwise would lead to economic losses to farmers and dairy industry, the early and precise detection of causative bacterial species is crucial in determining the line of action for the treatment of cattle suffering from sub-clinical intra-mammary infections.
For over a long period of time, microbiological cultural examination was considered as the gold standard for detection of mastitis pathogen from the quarter milk samples. But recently several reports on milk samples from mastitic cows failing to show bacterial growth by culture examination have shifted attention to opt for more precise and less time consuming molecular detection methods (Taponen et al. 2009).
In case of clinical mastitis, the physical manifestations are more visible, viz. the swelling and redness of udder quarters followed by fever and change in milk quality. While in case of sub-clinical mastitis the bacterial infection is established and their presence is felt in milk, but the visual clinical manifestations are absent leading to failure in detection of SCM. The milk production is lowered. Hence, in order to mitigate further larger economic loss, periodic evaluation of milk for the presence of bacteria is eminent, where bacteriological culture examination along with other test is being employed.
In our study on prevalence of bovine sub-clinical mastitis, it was ensured that the same samples subjected to culture examination were also evaluated by a more precise Real-Time PCR based PathoProof Mastitis Assay. The results showed a stark difference in the detection of pathogens, where Real-Time PCR based PathoProof Mastitis Assay detected bacterial species in several samples which failed to show presence of bacteria through conventional culturing.
In the present study, 33.33% (52 samples) with “no-growth” on bacteriological culturing showed presence of one or more species by real-time PCR. Taponen et al. (2009) investigated bacteriological etiology of 79 “no-growth” (on conventional culture) mastitic milk samples from cows with clinical mastitis using the same diagnostic kit as the one used in our study. In our study, 9.71 per cent SCM quarters did not show any growth on culture. Generally bacteria fail to grow in conventional culture in a substantial proportion of mastitic milk samples. Samples with no bacterial growth are generally more common in sub-clinical mastitis rather than clinical mastitis. In two earlier studies on sub-clinical mastitis, the percentage of culture-negative samples have been reported to have ranged from 28.7 per cent (Koivula et al. 2007) to 38.6 per cent (Bradley et al. 2007). Makovec and Ruegg (2003) reported a figure of 49.7 per cent without differentiating between sub-clinical and clinical mastitis.
Several reasons have been implicated for such discrepancy. The foremost being the low concentration of bacteria in milk which fail to be detected during routine culture examination which employs approximately 0.01 mL of milk sample (Sears et al. 1990; Hogan et al. 1999). Few other parameters influencing the low detection includes pre-culture incubation, pre-culture freezing and application of large plate volumes reduced the proportion of samples with no growth (Schukken et al. 1989; Dinsmore et al. 1992).
Dead bacterial DNA may be one of the factors. Milk contains several antibacterial substances such as lactoferrin, lysozyme, lactoperoxidase, components of complement, and immunoglobulins to play their role (Rainard and Riollet, 2006). In healthy quarters they are at low concentration, but they increase in mastitic quarters. Additionally presence of external inhibitory substances may inhibit the bacterial growth. Thus, information about possible antibiotic treatment of the cow prior to sampling is crucial. In present study, the cows under antibiotic treatment were excluded; hence the later condition doesn’t affect the current investigation.
Milk samples from healthy quarters either do not have live bacteria or, if any, then in very low quantity. A very low concentration of bacteria can lead to a no growth result on bacterial culture. Methods based on PCR techniques are very sensitive and capable of detecting very low bacterial copy numbers. This has been corroborated by 100 per cent analytical specificity of the PCR test which was based on an extensive bacterial strain collection, representing 83 species originating from 6 countries across Europe and North America (Koskinen et al. 2009). The authors suggested that such data gives a strong indication that it is unlikely that the PCR test would deliver an erroneous positive result if DNA from the respective microorganism is absent in the sample. Instead, when such a high level of analytical accuracy is combined with the results obtained for the qms from healthy quarters, it can be said that PCR positive/culture negative samples from healthy quarters is that bacteriological culture provided false negative results. It is important to note that although culture is capable of detecting only viable cells, the PCR test targets DNA and will identify both viable and non-viable organisms. Therefore, the clinical relevance of PCR positive/culture negative results requires further study.
As a diagnostic method, the PCR assay can be useful to identify the bacterial species responsible for IMI even if the bacteria are already dead. Such quarters do not require antibiotic treatment, but they can provide information about bacteriological etiology.
We observed that PathoProof mastitis PCR assay could detect three or more bacterial species in 80 (77.67 %) qms from SCM quarters against two samples (1.94 %) by bacterial culture. Earlier, Koskinen et al. (2010) reported detection of 3 or more bacterial species by PCR in 5 (10.87 %) samples from sub-clinical mastitis contrary to bacterial culture that did not show a single milk sample resulting in the growth of 3 or more bacterial organisms.
In our study, in 3 (2.91 %) qms from SCM quarters a bacterial species not targeted by PathoProof mastitis PCR assay was detected. Earlier, Koskinen et al. (2010) reported identification of a bacterial species not targeted by PCR in 9 (19.57 %) out of 46 samples from SCM quarters.
Overall, we observed that PathoProof mastitis PCR assay detected bacteria in 100 per cent SCM quarters against 90.29 per cent by conventional bacterial culture. In a similar investigation, Koskinen et al. (2010) reported bacterial detection in 89 per cent clinical samples by PCR against 77 per cent by bacterial culture.
Thus, culture-negative samples constitute a substantial percentage of samples which are misjudged leading to loss in milk quality. Peal-time PCR based assay exhibited presence of pathogen in more than one third of overall milk samples. Thus, real-time PCR based assay offers a very high resolution in identification of mastitis pathogens. Since, the time taken to perform PCR-based assay is just 12-24% of conventional culturing, it ensures reduction of the total duration of treatment. While its precision ensures use of correct antibiotics to restore robust health. In the long-term, it also supports in decreasing the occurrence of multi-drug resistant bacterial species in the cattle.
ACKNOWLEDGMENTS
The authors are thankful to Government of India’s Rashtriya Krishi Vikas Yojana (RKVY) Stream – I Project (B.H. 18442) for providing financial support. The work presented was an integral part of KS’s post-doctoral research at Anand Agricultural University (A.A.U.). KS is appreciative of Dr. Vaibhav Bhatt, Dr. Pradeep Kumar Sharma and Mr. Piyush Valand for the invaluable interactions. KS expresses his gratitude towards the milkers of Livestock Research Station of A.A.U. for extending their support during the entire study.
- Barkema HW, Schukken YH, Zadoks RN. The role of cow, pathogen, and treatment regimen in the therapeutic success of bovine Staphylococcus aureus mastitis. J Dairy Sci, 2006; 89: 1877–1895
- Bradley AJ, Leach KA, Breen JE, Green LE, Green MJ. Survey on the incidence and aetiology of mastitis on dairy farms in England and Wales. Vet Rec, 2007; 160: 253–258
- Bustin SA. Quantification of nucleic acids by PCR. In: An A-Z Manual of Quantitative PCR. IUL Press, La Jolla, 2004; CA, pp 5–46
- Cowan ST, Steel JK. Manual for the identification of the medical bacteria. 3rd edn. Cambridge University Press, Cambridge 1970.
- Dinsmore RP, English PB, Gonzalez RN, Sears PM. Use of augmented cultural techniques in the diagnosis of the bacterial cause of clinical bovine mastitis. J Dairy Sci, 1992; 75: 2706– 2712
- Gonzalo C, Martinez JR, Carriedo JA, Primitivo FS. Fossomatic cell-counting on ewe milk: Comparison with direct microscopy and study of variation factors. J Dairy Sc, 2003; 86: 138-145
- Hogan JS, González RN, Harmon RJ, Nickerson SC, Oliver SP, Pankey JW, Smith KL. Laboratory Handbook on Bovine Mastitis. Rev. ed. National Mastitis Council, Madison, WI 1999.
- International Dairy Federation. Suggested interpretation of mastitis terminology. International Dairy. Fed. Bull. 1999; 338: 3–26
- International Dairy Federation. Bovine mastitis. Definition and guidelines for diagnosis. Brussels, Belgium. IDF Bul, 1987; 211: 24
- Koivula M, Mäntysaari EA, Pitkälä A, Pyörälä S. Distribution of bacteria and seasonal and regional effects in a new database for mastitis pathogens in Finland. Acta Agric. Scand. A, 2007; 57: 89–96
- Koskinen MT, Holopainen J, Pyorala S, Bredbacka P, Pitkala, A, Barkema HW, Bexiga R, Roberson J, Solverod L, Piccinini R, Kelton D, Lehmusto H, Niskala S, Salmikivi L. Analytical specificity and sensitivity of a real-time polymerase chain reaction assay for identification of bovine mastitis pathogens. J Dairy Sci, 2009; 92: 952-959
- Koskinen MT, Wellenberg GJ, Sampimon OC, Holopainen J, Rothkamp A, Salmikivi L, Haeringen WAV, Lam TJGM, Pyorala S. Field comparison of real-time polymerase chain reaction and bacterial culture for identification of bovine mastitis bacteria. J Dairy Sci, 2010; 93: 5707-5715
- Makovec JA, Ruegg, PL. Results of milk samples submitted for microbiological examination in Wisconsin from 1994 to 2001. J Dairy Sci, 2003; 86: 3466–3472
- National Mastitis Council. Current Concepts of Bovine Mastitis. 4th ed. National Mastitis Council, Madison, WI 1996.
- National Mastitis Council. Laboratory Handbook on Bovine Mastitis. National Mastitis Council, Madison, WI 1999.
- National Mastitis Council. Microbiological Procedures for the Diagnosis of Bovine Udder Infection and Determination of Milk Quality. 4th ed. National Mastitis Council, Madison, WI 2004.
- Pitkälä A, Gindonis V, Wallin H, Honkanen-Buzalski T, Interlaboratory proficiency testing as a tool for improving performance in laboratories diagnosing bovine mastitis. J Dairy Sci, 2005; 88: 553–559
- Pyörälä S. New strategies to prevent mastitis. Reprod Domest Anim, 2002; 37:211–216
- Rainard P, Riollet C. Innate immunity of the bovine mammary gland. Vet Res, 2006; 37: 369–400
- Schukken YH, Smit JAH, Grommers FJ, Vandegeer PL, Brand A. Effect of freezing on bacteriologic culturing of mastitis milk samples. J Dairy Sci, 1989; 72:1900–1906.
- Sears PM, Smith BS, English PB, Herer PS, González RN. Shedding pattern of Staphylococcus aureus from bovine intramammary infections. J Dairy Sci, 1990; 73: 2785–2789
- Taponen S, Salmikivi L, Simojoki H, Koskinen MT, Pyorala S, Real-time polymerase chain reaction-based identification of bacteria in milk samples from bovine clinical mastitis with no growth in conventional culturing. J Dairy Sci, 2009; 92: 2610–2617.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


