ISSN: 0973-7510
E-ISSN: 2581-690X
Inefficiency of antibiotics because of emerging resistance, to reduce use of chemical preservatives and to alleviate other gastrointestinal or urogenital disorders- using probiotics get prioritized in recent times. We investigated probiotic potentiality of L. coryniformis subsp. torquens MTi1 and L. coryniformis MTi2 isolated from Nile Tilapia. A set of in vitro probiotic parameters (antibacterial activity, temperature sensitivity, pH, bile salt, NaCl tolerance, antibiotics resistance) were evaluated by performing disc diffusion, agar well diffusion methods and several other microbiological tests. Both isolates demonstrated antibacterial activity against Escherichia coli, Salmonella Typhii, Bacillus subtilis and Pseudomonas aeruginosa whereas L. coryniformis MTi1 also active against Staphylococcus aureus. They could grow at wide range of temperatures(27-45°C) and pH (2-9); had tolerance to 0.5-2% bile salt and 1-7% NaCl; ferment lactose. L. coryniformis subsp. torquens MTi1 was not killed by Cefixime(5µg) whereas L. coryniformis MTi2 neither killed by Cefixime(5µg) nor Streptomycin(10µg) in antibiotic susceptibility testing. Best antibacterial production of isolates found at 37°C, pH 2 and after 72 hours of incubation. We observed approximately 42% and 40% more antibacterial production from L. coryniformis subsp. torquens MTi1 and L. coryniformis MTi2 respectively after 72 hours of incubation instead of 24 hours. L. coryniformis MTi2 could produce bacteriocin or bacteriocin like substances active against E. coli (15mm) and B. subtilis (14mm). The study concludes based on empirical observations that, the isolates have potentiality to be efficient probiotic which will eventually contribute to promote public health.
Probiotics, Lactobacillus coryniformis, Nile Tilapia, Antibacterial activity
Probiotics are widely recognized as helpful bacteria as they play role to keep our gut healthy. FAO and WHO (2001) defined probiotic as “Live microorganisms which when administered in adequate amounts confer a health benefit on the host”1. Injudicious or haphazard use of antibiotics to challenge frightful pathogens, extensive application of chemical preservatives to preserve foods and food products and increase in the prevalence of allergic diseases due to environmental hazards have imposed a great threat to human health2. To overcome those challenges, scientists and regulatory authorities are looking for alternative living agents and thus probiotics get prioritized.
Probiotics have been used to reduce the duration of infectious diarrhea, shortening the initial phase of watery stool, increase the amount of IgA antibody and decrease virus shedding in viral diarrhea3. Probiotics have also been reported to control lactose intolerance, irritable bowel syndrome, and reduction of gastric ulcers4,5. To balance the disequilibrium in the immune response causing the allergic response and to reduce the levels of blood cholesterol by consumption of probiotics containing dairy foods have also been demonstrated2,6. To substitute the employment of antibiotics which are used to maintain a good balance of the gastrointestinal tract (GIT) microflora, to eliminate pathogenic microorganisms, to facilitate the reduction of gastrointestinal upsets probiotics are proven to be effective7. Lactobacillus spp. are ubiquitous in nature, generally regarded as safe and most often used as probiotics. There are several salient features behind its popularity: lactobacilli are found naturally in the human gut; particularly common in the small bowel; grow in the absence of oxygen or small amounts of oxygen; they feed sugars as a source of carbon which are plenty in the intestine; require a range of minor nutrients to enable them to live and grow, as there is a wide range of food in that part of the intestine so they are prominent there. Besides intestine, they are also found in the vagina where they are the dominant resident bacteria and they protect against vaginal infection8,9. Lactobacillus coryniformis, have been studied for its potential probiotic efficiency. L. coryniformis CECT 5711 has the ability to produce both reuterin and cobalamin10. It has been found to survive when exposed to conditions found in the gastrointestinal tract, and particularly low pH and bile, which seems to be a critical pre-requisite for a probiotic strain10. The strain seemed to adhere to the intestinal mucosa, a pre-requisite for gut colonization that may lead to the competitive exclusion of pathogenic bacteria, displayed b-galactosidase activity that can exert beneficial effects in the host11. L. coryniformis have been reported to modulate the immune response against allergic reactions. Study conducted by Martinez et al. (2009) showed that the consumption of a probiotic product containing L. coryniformis CECT5711 along with L. gasseri CECT5714 induced beneficial effects on immunological parameters in allergic children2. Obesity is associated with intestine dysbiosis and is characterized by a low-grade inflammatory status, which affects vascular function. Animal model experiment revealed that an endothelial-protective effect of probiotic L. coryniformis CECT5711 in obese mice by increasing NO bioavailability, suggesting the therapeutic potential of this gut microbiota manipulation to prevent vasculopathy in obesity12.
Considering those facts and discussed perspectives, this study focused on isolation of Lactobacillus spp., from intestine of Nile Tilapia (Oreochromis niloticus) fishes and in vitro evaluation of their probiotic potentiality.
Sample collection
Healthy Nile Tilapia (Oreochromis niloticus) fishes were collected from local ponds in Chittagong region, Bangladesh. The fishes were washed thoroughly with sterilized distilled water and processed in lab within two hours of collection.
Isolation, selection and identification of Lactobacillus spp.
For isolation of Lactobacillus, selective media for Lactic acid bacteria (LAB), MRS (De Man Rogosa Sharpe) was used and serial dilution agar plate technique was implemented. About 10g of fish intestine was cut aseptically and homogenized with 90 mL normal saline in a homogenizer and serially diluted up to 10-5. About 1 mL of each dilution of the sample was pipetted into sterile petri plates. MRS agar media were poured and incubated at 37°C for 24-48 hours13.
As probiotic Lactobacillus should be good antimicrobial producer, the isolated Lactobacillus were first screened for antibacterial activity against five selected test organisms. 200mL of MRS broth was autoclaved at 121ºC for 15 minutes and inoculated with the colonies Lactobacillus and incubated at 37ºC for 2-3 days under stationary condition. Then it was centrifuged at 9000 rpm for 15 minutes at 4°C. The supernatant was then filtered through Whatman No. 1 filter paper to remove residual cells. Petri-plates were prepared by pouring sterile molten Muellar Hinton medium and allowed it to solidify. 100µL of each standardized test microorganisms were spread on agar plates. Two wells (each 7mm in diameter) made into agar plates with sterile borer. The wells were loaded with 100µL of filtered cell free supernatant (CFS) and 100µL sterile broth. Plates were incubated at 37°C for 24 hours. After incubation, diameter of zone of inhibition was observed and measured14.
Finally, the good antibacterial producer Lactobacillus spp. were identified by characterization on the basis of their morphological characteristics including size and shape of the organism, arrangement of the cells, presence or absence of the spores, regular or irregular forms, acid fastness, gram reaction etc.; cultural and physiological characteristics including H2S production, nitrate reduction, deep glucose agar test, fermentation of different carbohydrates etc. All these characteristics were then compared with the standard description of ‘‘Bergey’s Manual of Determinative Bacteriology’’, 8th edition15.
Test microorganisms
The test pathogenic organisms used in this study were Escherichia coli ATCC25922, Salmonella Typhii AE14296. Staphylococcus aureus ATCC25923, Bacillus subtilis IFSTIM22, Pseudomonas aeruginosa CRL(ICDDR,B). The test microorganisms were standardized by using 0.5 Mc Farland standard solutions which gives approximate cell density of 1.5 x 108 CFU/mL, having absorbance of 0.132 at wavelength of 600nm16.
Assessment of in vitro potentiality of Lactobacillus spp. based on evaluating following parameters
Temperature sensitivity
Lactobacillus spp. separately inoculated in MRS broth was incubated at varying temperatures, i.e., 27, 37, and 45ºC for 24-48 hours. After incubation, the absorbance of MRS broths were taken at 600nm by a spectrophotometer to measure microbial load.
pH tolerance
As like above experiment, the Lactobacillus cultures were separately inoculated into sterile MRS broth tubes of varying pH (2, 4, 7 and 9) incubated at 37ºC for 24-48 hours. The absorbance of MRS broths was taken at 600nm by a spectrophotometer to measure microbial load.
Bile salt tolerance
The MRS broth media with different concentrations of bile salt (0.5, 1.0 and 2.0%) were inoculated separately with each Lactobacillus sp. and incubated at 37ºC for 48 hours. Then the absorbances of MRS broths were taken at 600nm by a spectrophotometer for measuring microbial load.
NaCl tolerance
The MRS broth media prepared with varying concentrations of NaCl (1, 3 and 7%) were inoculated separately with each Lactobacillus sp. and incubated at 37ºC for 48hours. Then the absorbances of MRS broths were taken at 600nm by a spectrophotometer for measuring microbial load.
Response to some common antibiotics
Sensitivity of Lactobacillus spp. to some commonly used antibiotics was determined by antibiotic susceptibility testing performed according to Kirby-Bauer discs diffusion method on MRS agar plates. The used antibiotics were Penicillin G(10units), Chloramphenicol(30µg), Erythromycin(15µg), Cefixime(5µg), Cephradine(30µg), Streptomycin(10µg) and Rifamycin(5µg).
Lactose utilization
The acid production by Lactobacillus spp. was detected by observing the change in color of the medium red to yellow. Sterilized fermentation medium (Peptone 10g, NaCl 15g, phenol red 0.018g, lactose 5g, for 1L distilled water and final pH 7.0) was inoculated with Lactobacillus cultures and incubated at 37ºC for 24-48 hours. Change in color from red to yellow indicates the production of acid17.
Characterization of probiotic Lactobacillus spp. by determining optimum temperature, pH and incubation period for getting best antibacterial production
Optimum Temperature
To determine the optimum temperature for getting maximum antibacterial production by the Lactobacillus spp., MRS broth tubes were prepared for each isolate and inoculated with Lactobacillus sp. Then the MRS broth tubes were incubated at three different temperatures (27ºC, 37ºC and 45ºC) for 24-48 hours. Then the culture broth was centrifuged at 9000rpm for 15minutes and filtered through Whatman No. 01 filter paper. Then antibacterial activity of the cell free supernatant (CFS) was assayed against respective test organism (previously showed antagonistic activity) by agar well diffusion method. (100µL CFS in each well)
Optimum pH
MRS broth were adjusted for four different pH (2, 4, 7, 9) using 0.1N HCl and 0.1N NaOH. Each Lactobacillus sp. was inoculated separately and incubated at optimized temperature for 24-48hours. Then the culture broth was centrifuged at 9000rpm for 15minutes and filtered through Whatman No. 01 filter paper. Then antibacterial activity of the CFS was assayed against respective test organism by agar well diffusion method. (100µL CFS in each well)
Optimum incubation period
MRS broth tubes were prepared and the pH was adjusted to optimum for each isolates. Each Lactobacillus sp. was inoculated separately and incubated at optimized temperature for 24, 48, 72 and 96hours. After completing each incubation period broth was centrifuged at 9000rpm for 15minutes and then filtered through Whatman No. 01 filter paper. Then antibacterial activity of the culture supernatant was assayed against respective test organism by agar well diffusion method. (100µL CFS in each well)
Determination of reasons behind antibacterial activity of Lactobacillus spp.
To determine cause(s) (Lactic acid/acetic acids, H2O2 or Bactericins/Bacteriocin like substance) of antibacterial activity, 1mL of frozen Lactobacillus isolate was cultured overnight in 20mL MRS broth. Then 1mL of this culture was sub-cultured overnight in 20mL fresh MRS broth. Cells were removed by centrifuging at 9000rpm for 15minutes. The supernatant was filtered through a sterile Whatman No. 1 filter paper and 100µL of the pH unadjusted aliquot of cell free supernatant (CFS) was added to the first well. The remaining CFS was adjusted to pH 6.0 with 1M/lN NaOH in order to rule out possible inhibitory effects due to organic acids. 100µL of the pH adjusted CFS was filtered and added to the second well. The neutralized CFS was then treated with 1mg/mL of catalase (Merck KGa A, Germany) at 25°C for 30min to eliminate the possible inhibitory action of H2O2 and filtered. Then 10µL catalase treated CFS was placed in the third well. If inhibition zone were found in the third well, the isolates were considered to be able to produce bacteriocin or bacteriocin like substances)18.
Isolation, identification and selection of Lactobacillus spp.
Fifteen (15) strains of Lactobacillus spp. have been isolated from intestine of Nile Tilapia fish. Among them only 2(13%) isolates showed potential antibacterial activity against five test pathogenic organisms. Finally, the two isolates were identified as Lactobacillus coryniformis subsp. torquens MTi1 and L. coryniformis MTi2 based on conventional methods using cultural, morphological characteristics and biochemical reactions as described in ‘‘Bergey’s Manual of Determinative Bacteriology’’, 8th edition15.
Lactose utilization
The acid production by utilizing lactose in the medium by the selected Lactobacillus isolate was detected by observing the change in the color of the medium from red to yellow. Both isolates showed the capability of producing lactose in test medium.
Characterization of probiotic L. coryniformis subsp. torquens MTi1 and L. coryniformis MTi2.
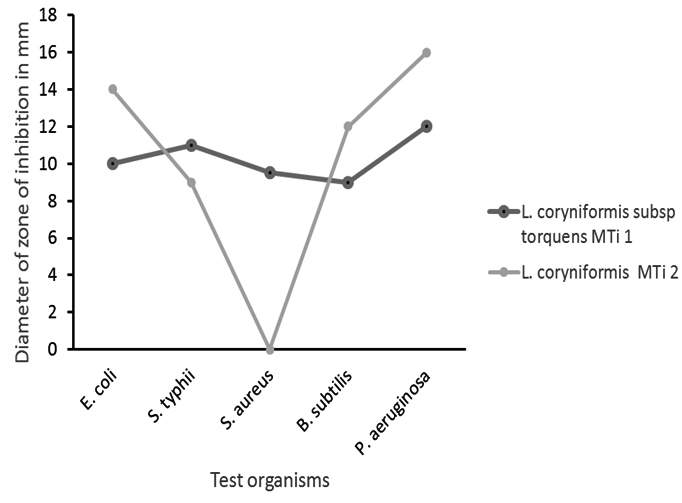
The aim of this study was to isolate Lactobacillus spp. having potentiality to be used as probiotic from intestine of Nile Tilapia. To be an efficient probiotic, Lactobacillus spp. must have potential antimicrobial activity against gut pathogens, survival capability at intestinal pH and temperatures, could utilize lactose, tolerance to bile salt and NaCl; may have resistance to commonly used antibiotics. Our isolated L. coryniformis subsp. torquens MTi1 and L. coryniformis MTi2 demonstrated antibacterial activity against E. coli, S. Typhii, B. subtilis and P. aeruginosa. L. coryniformis MTi1 also additionally showed antibacterial activity against S. aureus (9.5mm). We observed that L. coryniformis subsp. torquens MTi1 showed better antibacterial activity against S. Typhii (11 vs 9mm) than L. coryniformis MTi2 while L. coryniformis MTi2 has comparatively greater antibacterial activity against E. coli (14 vs 10mm), B. subtilis (12 vs 9mm), P. aeruginosa (16 vs 12mm) (Fig. 1). Martin et al., (2005) reported that L. coryniformis CECT 5711, a strain isolated from a goat’s milk cheese, displayed a broad-spectrum antimicrobial activity; could produce lactic acid, acetic acid, reuterin and cobalamin, a cofactor required for conversion of glycerol to 3-HPA through a glycerol dehydratase10. Nair and Surendran (2009) also reported that lactic acid bacteria isolated from fish and prawn have inhibitory activity against Listeria monocytogenes, Bacilus cereus, E. coli, S. aureus and S. Typhii19.
Fig. 1. Antibacterial activity of L. coryniformis subsp. torquens MTi1 and L. coryniformis MTi2 against five test organisms
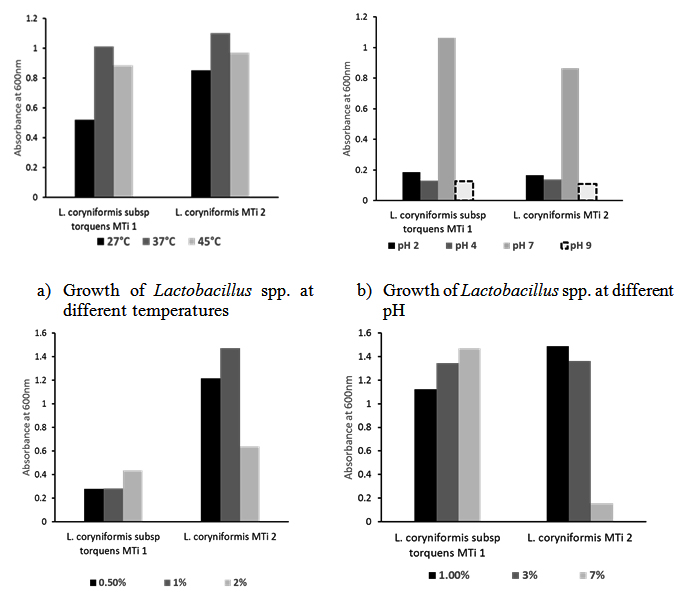
Both isolates could grow at wide range of temperatures (27-45°C) though best growth found at 37°C (Fig. 2a), and showed satisfactorily tolerance to wide range of pH (2-9) in vitro growth medium. Although they are grow less in pH 2, 4 or 9 but maximum growth found at neutral pH (7) (Fig. 2b). Resistance to low pH is an important selection criterion for probiotic microorganisms as gastric juice in the stomach destroys most microorganisms ingested. Many other studies showed that the exposure of Lactobacillus strains to pH values of 2.5–4.0 does not influence their survival rate, but it dropped at lower pH values. The ability of lactobacilli to survive the passage through media with physiological pH of 2-3 (to mimic the stomach environment) was reported to be variable and strain dependent, but with a survival rate of approximately 85%, which is very significant for the probiotics20.
In this study, L. coryniformis subsp. torquens MTi1 and L. coryniformis MTi2 were able to survive at 0.5 to 2% bile concentration in test media. The best growth of L. coryniformis subsp. torquens MTi1 found with 2% bile salt whereas L. coryniformis MTi2 at 1%. It is apparent that growth ability of L. coryniformis MTi2 is better than L. coryniformis subsp. torquens MTi1 in presence of bile salt (Fig. 2c). Tolerance to bile salts is a prerequisite for colonization and metabolic activity of bacteria in the small intestine of the host which eventually help Lactobacilli to reach the small intestine and colon and contribute in balancing the intestinal microflora21. Both isolates also showed their growth capability in presence of 1-7% NaCl in growth medium though best growth found with 3% NaCl. The cell density indicates that NaCl may have promoting effect on the growth of the Lactobacillus isolates (Fig. 2d). Hoque et al., (2010) reported that Lactobacillus sp. isolated from yoghurts can tolerate 1–9% NaCl. Pundir et al., (2013) also made similar observations regarding the NaCl tolerance of Lactobacillus spp14.
Fig. 2. (a-d). Both L. coryniformis subsp. torquens MTi1 and L. coryniformis MTi2 could withstand at wide range of temperatures(27-45°C), pH (2-9), bile salt concentration (0.5-2%) and NaCl (1-7%) treatment
L. coryniformis subsp. torquens MTi1 and L. coryniformis MTi2 both could utilize lactose in growth medium which is another important selection criterion for probiotics. Lactose intolerant people cannot metabolize lactose due to the lack of essential enzyme a–galactosidase which resulting symptoms including abdominal pain, cramping and diarrhea when they consume milk or lactose containing products. If lactose passes through from the small intestine, it is converted to gas and acid in the large intestine by the colonic microflora. Other studies provide evidence that the addition of certain starter cultures (lactose utilizing Lactobacillus sp.) to milk products, allows the lactose intolerant people to consume those products without the usual rise of breath hydrogen or associated symptoms14. The oral administration of the probiotic strains combination of the probiotic strains L. gasseri CECT5714 and L. coryniformisCECT5711 led to an improvement of parameters such as the production of short chain fatty acids, the fecal moisture and the frequency and volume of the stools result a clear improvement in intestinal environment eventually make positive effect on healthy adults22,23.
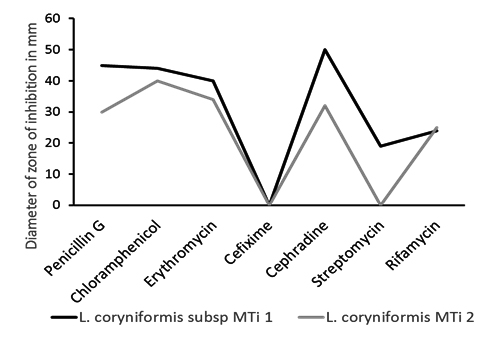
Fig. 3. Resistance pattern of L. coryniformis subsp torquens MTi1 and L. coryniformis MTi2 against some commonly used antibiotics
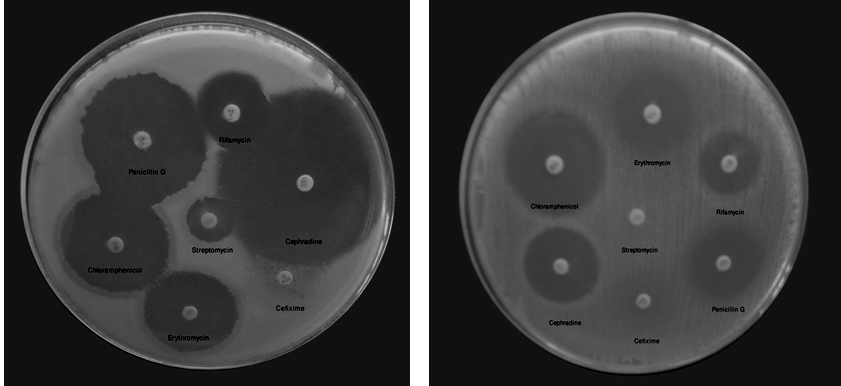
Fig. 4. Photograph of antibiotic susceptibility testing (AST) of L. coryniformis subsp. torquens MTi1 and L. coryniformis MTi2
Small scale antibiotic susceptibility testing revealed that both L. coryniformis subsp. torquens MTi1 and L. coryniformis MTi2 could survive against few commonly used antibiotics. We observed that L. coryniformis subsp. torquens MTi1 was not killed by Cefixime whereas L. coryniformis MTi2 neither killed by Cefixime(5µg) nor Streptomycin(10µg) (Fig. 3 and 4). Resistance of the probiotic strains to some antibiotics could be used for both preventive and therapeutic purposes in controlling intestinal infections; wide spectrum resistance of antibiotics indicated that if isolated probiotics induced in patients treated with antibiotic therapy may be helpful in faster recovery of the patients due to rapid establishment of desirable microbial flora24. The above mentioned data and scientific facts indicate that our isolated L. coryniformis subsp. torquens MTi1 and L. coryniformis MTi2 showed potential characteristics to be efficient probiotic though it still requires further additional confirmatory in vivo experiments to be performed.
We also tried to optimize temperature, pH and incubation time to get the best antibacterial production by the L. coryniformis subsp. torquens MTi1 and L. coryniformis MTi2 against respective test pathogens (previously showed antagonistic activity). Cell free supernatant (CFS) of the isolate was tested for antibacterial activity against test organisms by agar well diffusion method after letting the isolates grow at different temperatures (27, 37 and 45°C). The maximum average and least Coefficient of Variation (CV) of antibacterial production of L. coryniformis subsp. torquens MTi1 against test pathogens found at 37°C. Hence, 37°C is the optimum temperature for the antibacterial production. Keeping optimum temperature constant, the maximum average of antibacterial production of L. coryniformis subsp. torquens MTi1 found at pH 2 but the least CV found at PH 7, which means that consistent antibacterial production of L. coryniformis subsp. torquens MTi1 occurred at PH 7 even the maximum average antibacterial production was at PH 2. By keeping temperature at 37°C and pH at 2, the maximum antibacterial production from L. coryniformis subsp. torquens MTi1 found after 72 hours though the consistent antibacterial production against tested pathogens found just after 24 hours. Again, in case of L. coryniformis MTi2, the optimum temperature is found at 37°C since both maximum average and least CV of antibacterial production by the isolate found at 37°C. There is no optimum pH for best antibacterial production found for L. coryniformis MTi2 as maximum average found at pH 2 and least CV found at pH 4. The optimum incubation period for L. coryniformis MTi2 found after 72 hours as both maximum average and least CV of antibacterial production found after 72 hours (Table 1). After performing ANOVA (Analysis of Variance), the parameters temperature and incubation period for getting best antibacterial production by L. coryniformis subsp. torquens MTi1 are found to be significant (p<0.05), i.e., with the change in temperature and incubation period a significant change will occur in antibacterial production. With changing of pH, the changes in antibacterial production by L. coryniformis subsp. torquens MTi1 is insignificant, i.e., the change in antimicrobial activities is very less considerable. On the other hand, in case of L. coryniformis MTi2, with changing in pH and incubation period, a significant (P<0.05) change will occur in antibacterial production.
Table (1):
Determination of optimum temperature, pH and incubation period for getting best antibacterial production from probiotic L. coryniformis subsp. torquens MTi1 and L. coryniformis MTi2.
| Antibacterial activity against selected test organisms | L. coryniformis subsp. torquens MTi1 | L. coryniformis MTi2 | ||||
|---|---|---|---|---|---|---|
| Temperature (°C) | pH | Incubation time (Hours) | Temperature(°C) | pH | Incubation time (Hours) | |
| Maximum Average | 37 | 2 | 72 | 37 | 2 | 72 |
| Least CV# | 37 | 7 | 24 | 37 | 4 | 72 |
# CV is the Coefficient of Variation
To determine which substance (Lactic acid/acetic acids, H2O2, Bacteriocins or Bacteriocin like substances) cause the antibacterial activity, CFS of the L. coryniformis subsp. torquens MTi1 and L. coryniformis MTi2 were first neutralized using NaOH/HCl to diminish the antibacterial activity of acid, then treated with catalase enzyme to reduce antibacterial activity of H2O2 and then antibacterial activity of CFS assayed by agar well diffusion method. It was found that the causes behind antibacterial activity of L. coryniformis subsp. torquens MTi1 are acids and H2O2, as it lost its antibacterial activity after adjusting to neutral pH and treated with catalase. On the other hand, L. coryniformis MTi2 retained its antibacterial activity against E. coli (15mm) and B. subtilis (14mm) even after adjusting to neutral pH and treated with catalase. This implicate that this isolate could produce bacteriocin or bacteriocin like substances (Table 2)18.
Table (2):
Determination of reasons behind antibacterial activity of L. coryniformis subsp. torquens MTi1 and L. coryniformis MTi2 by gradually eliminating antibacterial effect of acids and H2O2. (Diameter of zone of inhibition in mm).
| E. coli | S. Typhii | P. aeruginosa | B. subtilis | S. aureus | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| MTi1 | MTi2 | MTi1 | MTi2 | MTi1 | MTi2 | MTi1 | MTi2 | MTi1 | MTi2 | |
| Raw CFS | 17 | 23 | 17 | 20 | 24 | 22 | 18 | 24 | 19 | – |
| pH adjusted CFS | 10 | 19 | 12 | 15 | 17 | 11 | 12 | 19 | 18 | – |
| pH adjusted and Catalase treated CFS | – | 15 | – | – | – | – | – | 14 | – | – |
MTi1: L. coryniformis subsp. torquens MTi1; MTi2: L. coryniformis MTi2; CFS: Cell Free Supernatant. “-“: No antibacterial activity.
Searching new or better candidate of probiotic is focused and continuously progressing research interest. Based on empirical data gathered from in vitro experiments in this study, we can conclude that isolated L. coryniformis subsp. torquens MTi1 and L. coryniformis MTi2 have great potentiality to be used as probiotics after passing through other relevant in vitro and in vivo experiments.
ACKNOWLEDGMENTS
We gratefully acknowledge the all kind of logistical and technical supports from the Department of Microbiology, University of Chittagong, Bangladesh.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
- Probiotics. http://www.fao.org/es/ESN/probio/probio.htm, Accessed on 28 April 2007.
- Martínez Cañavate, A., Sierra, S., Lara Villoslada, F., Romero, J., Maldonado, J., Boza, J., Xaus, J. and Olivares, M. A probiotic dairy product containing L. gasseri CECT5714 and L. coryniformis CECT5711 induces immunological changes in children suffering from allergy. Pediatric Allergy and Immunology., 2009; 20(6): 592-600.
- Parvez, S., Malik, K.A., Kang, S.A., Kim, H.Y. Probiotics and their fermented food products are beneficial for health. J. of Appl. Microbiol., 2006; 100(6): 1171-1185.
- Montalto, M., Curigliano, V., Santoro, L., Vastola, M., Cammarota, G., Manna, R., Gasbarrini, A. and Gasbarrini, G. Management and treatment of lactose malabsorption. World journal of gastroenterology: WJG., 2006; 12(2):187.
- Levri, K.M., Ketvertis, K., Deramo, M., Merenstein, J.H. and D’amico, F. Do probiotics reduce adult lactose intolerance? A systematic review. The Journal of family practice., 2005; 54(7): 613-620.
- Simons, L.A., Amansec, S.G. and Conway, P. Effect of Lactobacillus fermentum on serum lipids in subjects with elevated serum cholesterol. Nutr. Metab. Cardiovasc Dis., 2006; 16(8): 531-535.
- Brizuela, M.A., Serrano, P. and Pérez, Y. Studies on probiotics properties of two lactobacillus strains. Brazilian Archives of Biology and technology., 2001; 44(1): 95-99.
- Versalovic, J. and Wilson, M. Therapeutic microbiology: probiotics and related strategies. ASM Press, 2008.
- Lactobacillus– the ‘pioneer’ of probiotics? https://www.protexin.com, Accessed on 14 May 2016
- Martin, R., Olivares, M., Marin, M.L., Xaus, J., Fernandez, L. and Rodriguez, J.M. Characterization of a reuterin-producing Lactobacillus coryniformis strain isolated from a goat’s milk cheese. Int. J. Food Microbiol., 2005; 104: 267– 277.
- Fernandez, M.F., Boris, S. and Barbes, C. Probiotic properties of human lactobacilli strains to be used in the gastrointestinal tract. J. Appl. Microbiol., 2003; 94: 449– 455.
- Toral, M., Gómez-Guzmán, M., Jiménez, R., Romero, M., Sánchez, M., Utrilla, M.P., Garrido-Mesa, N., Rodríguez-Cabezas, M.E., Olivares, M., Gálvez, J. and Duarte, J. The probiotic Lactobacillus coryniformis CECT5711 reduces the vascular pro-oxidant and pro-inflammatory status in obese mice. Clinical Science., 2014; 127(1): 33-45.
- Jayachitra, J. and Ramanathan, N. Isolation of bacteriocin producing lactic acid bacteria from fish and its antimicrobial activity. Int J. Recent Sci. Res., 2012; 3(7): 581-584.
- Pundir, R.K, Rana, S., Kashyap, N. and Kaur, A. Probiotic potential of lactic acid bacteria isolated from food samples: an in vitro study. J. Appl. Pharm. Sci., 2013; 3(3): 85-93.
- Buchanan, R. E., & Gibbson, N. E. Bergey’s Manual of Determinative bacteriology 8th edition The Williams & Wilkins company, 1974.
- Andrews, J.M. Determination of minimum inhibitory concentrations. J. Antimicrob. Chemother., 2001; 48(1): 5-16.
- Ahmed, T. and Kanwal, R. Biochemical characteristics of lactic acid producing bacteria and preparation of camel milk cheese by using starter culture. Pak Vet. J., 2004; 24: 87-91.
- Yang, E., Fan, L., Jiang, Y., Doucette, C. and Fillmore, S. Antimicrobial activity of bacteriocin-producing lactic acid bacteria isolated from cheeses and yogurts. AMB Express., 2012; 2(1): 48.
- Nair, P.S. and Surendran, P.K. Biochemical characterization of lactic acid bacteria isolated from fish and prawn. J. Culture Collections., 2005; 4: 48-52.
- Belicová, A., Mikulášová, M. and Dušinský, R. Probiotic potential and safety properties of Lactobacillus plantarum from Slovak Bryndza cheese. BioMed research international, 2013.
- Tambekar, D.H. and Bhutada, S.A. Studies on antimicrobial activity and characteristics of Bacteriocins produced by Lactobacillus strains isolated from milk of domestic animals. Internet J. Microbiol., 2010; 8(2): 1-7.
- Olivares Martín, M., Díaz-Ropero, M.P., Gómez, N., Lara-Villoslada, F., Sierra Ávila, S., Maldonado Jurado, J.A., Martín, R., Rodríguez, J.M. and Xaus Pey, J. The consumption of two probiotic strains, Lactobacillus gasseri CECT5714 and Lactobacillus coryniformis CECT5711, boost the immune system of healthy adults. Int. Microbiol., 2006; 9: 47–52.
- Olivares Martín, M., Díaz-Ropero, M.P., Gómez, N., Lara-Villoslada, F., Sierra Ávila, S., Maldonado Jurado, J.A., Martín, R., Rodríguez, J.M. and Xaus Pey, J. Oral administration of two probiotic strains, Lactobacillus gasseriCECT5714 and Lactobacillus coryniformis CECT5711, enhances the intestinal function of healthy adults. Int. J. Food Microbiol., 2006; 107(2): 104-111.
- El-Naggar, M.Y.M. Comparative study of probiotic cultures to control the growth of E. coli 0157:H7 and Salmonella Typhimurium. Biotechnology., 2004; 3(2): 173-180.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.