Biofilms are surface-attached bacterial communities that are widespread in nature. A key factor in their development is quorum sensing (QS), a mechanism of intercellular communication among bacteria. Biofilm formation serves as a pivotal mechanism in the establishment of multidrug-resistance, significantly undermining the efficacy of conventional treatment strategies. The development of novel antimicrobial compounds that can disrupt or prevent biofilm formation has emerged as a critical strategy to mitigate the clinical impact of biofilm-associated infections. Medical devices are also receiving much attention in the healthcare sector to address this issue. Emerging studies suggests that both herbal formulations and synthetic agents are extensively explored for their prospective anti-biofilm efficacy. This review summarizes the mechanisms behind biofilm formation of commonly reported bacterial pathogens and regular techniques in medical device fabrication to control biofilm formation. Additionally, it focuses on the information about reported natural compounds as quorum quenching agents. Through an integrative analysis of biofilm mechanisms, anti-biofilm agents, and device fabrication techniques, this review offers critical perspectives for the development of clinically viable multi-targeted interventions for managing biofilm-driven medical complications.
Health-care Associated Infection, Quorum Sensing, Quorum Quenching, Medical Devices, Polymicrobial Colonies, Antibiotic Resistance
Biofilms play a pivotal role in nosocomial or Healthcare-associated infections (HAIs), especially those associated with medical implants such as orthopedic implants, urinary catheters, and intravascular catheters.1 Similarly, nosocomial infections associated with intensive care unit (ICU) have also been a major concern in the health sector. Commonly reported cases are Bloodstream infections (BSIs) and Urinary tract infections (UTIs). According to the U.S. Centers for Disease Control and Prevention (CDC), urinary catheters are the primary source of catheter-associated urinary tract infections (CAUTIs), while central venous catheters (CVCs) are commonly linked to bloodstream infections. The major risk factors were reported as the duration of catheterization and anatomical location of CVCs.2 According to the European Sepsis Group, around 28% of CVCs were reported every year from ICUs. A 2008 report by the European Centre for Disease Prevention and Control (ECDC) estimated that around 4.1 million patients worldwide are affected by healthcare-associated infections (HAIs). Surgical site infections are reported mainly from orthopedic implants. The rising incidence of orthopedic implant infections is largely attributed to the increasing population of elderly and disabled individuals undergoing surgical procedures. The unsupervised and excessive administration of antibiotics has substantially contributed to the rapid emergence and proliferation of antimicrobial resistance.3
Given the complexity of multi-species biofilms in device-associated infections and the growing challenge of antibiotic resistance, ongoing research into effective microbial eradication strategies is essential. Future therapeutics should ideally incorporate a combination of agents capable of targeting both Gram-positive and Gram-negative bacteria. Coating surfaces with one or a combination of antimicrobial agents is a frequent approach to controlling biofilms on device surfaces. In recent years, these devices have come to be classified medicated devices, owing to the incorporation of therapeutic agents for enhanced clinical performance. A significant limitation of these devices is the initial burst of antibiotic release, which is followed by prolonged low-concentration release phases.4 The successful development of catheters with long-term antimicrobial activity depends on both technological design approach and the choice of materials used in their production. The basic concept of designing the material is that it can absorb the antibiotics with gradual release for long-term treatment. Functionalized polymers with either antibiotic or metal coating are the best option which is widely investigated under different trials.5 Here, we review the common bacterial nosocomial pathogens and highlight the most successful and promising control approaches, ranging from traditional methods to advanced strategies for future application.
Quorum sensing
Intercellular communication is fundamental to the quorum sensing (QS) mechanisms in both Gram-positive and Gram-negative bacteria. QS-regulated genes are responsible for the production of various virulence factors, including exoenzymes, proteases, elastases, and pyocyanin. The enzyme hyaluronidase S (HysA) is a significant virulence determinant in Staphylococcus aureus, facilitating the hydrolysis of hyaluronic acid (HA) and contributing to tissue invasiveness. Hyaluronic acid is a major component of mammalian extracellular matrix. It will act as a barrier against microbial entry. Degradation of HA and tissues will allow the entry of S. aureus by breaking the barrier. Later hyaluronic acid will be involve in the dispersal of biofilm. Degraded products will be used as nutrients by the pathogens in the host body. Gram-positive bacteria have auto-inducer peptides (AIPs) that will be secreted to the environment. In Gram-positive bacteria, the signaling molecules are recognized by regulatory elements such as accessory gene regulators (Agr) and RNAIII. In contrast, Gram-negative bacteria typically utilize acylhomoserine lactones (AHLs) as their quorum sensing signals. The LuxI/LuxR system is induced by AHLs leading to the production of virulence factors such as pyocyanin, lectin, elastase, and protease.6
Biofilm: resistant enemies
Microbial biofilms are poly-microbial communities, they are self-embedded on living or non-living surfaces by producing Extracellular matrix (ECM) which is rich in DNA, proteins, carbohydrates etc. Sessile cells are commonly associated with ECM-production more than planktonic cells. Sessile cells are highly resistant to the environment as well as to antimicrobials. A concerning feature is that biofilm communities serve as favourable niches for plasmid exchange and bacterial conjugation. Thus the exchange of genes will enhance the multidrug-resistance capacity. The mechanisms behind the drug resistance are the following:
- Slow or incomplete penetration of antimicrobial agents through biofilm matrix
- Heterogeneous nature of biofilm environment and physiological responses of microorganisms. Each layers of biofilm has a different environment, e.g. accumulation of toxic substances, oxygen depletion, etc.
- Dormant condition of a few bacterial cells in the community
- Poly-microbial community infections
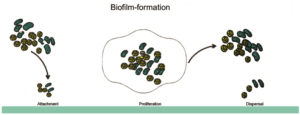
The microbial adhesion strongly depends on physicochemical properties and a combination of materials, hydrophilic properties and surface charges are the most important factors among them. Microbial attachment is a biphasic process; the first phase is reversible binding with weak Van der Waals forces, and the second stage is irreversible. The adhered cells will duplicate and communicate with each other and will produce many layers of cells. Different stages of biofilm formation is represented in Figure 1. This process is collectively called the “building blocks”, which starts from attachment to a suitable surface, maturation, and dispersal.7
ESKAPE pathogen biofilm formation and antimicrobial resistance
The ESKAPE group of pathogens which includes Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter spp. is recognized as a major cause of hospital-acquired infections worldwide. These pathogenic organisms demonstrate extensive multidrug-resistance capabilities, contributing to their persistence and posing significant challenges to existing treatment strategies. A significant contributor to their resistance is the overproduction of specific β-lactamase enzymes, notably TEM, SHV, and CTX-M types. For example, TEM-1 has been identified in Enterobacteriaceae, P. aeruginosa, and Escherichia coli, while SHV-type enzymes are predominantly found in K. pneumoniae. CTX-M enzymes have been detected in K. pneumoniae, A. baumannii, P. aeruginosa, and Enterobacter spp. These enzymes collectively confer resistance to β-lactam antibiotics such as ceftazidime, ceftriaxone, piperacillin, and amoxicillin.8
More than 20 Enterococcus species are considered clinically important, and in recent years, several have developed resistance to vancomycin, leading to the emergence of Vancomycin-Resistant Enterococcus (VRE). This resistance is categorized into six genotypic groups: VanA –High resistance against vancomycin and teicoplanin, VanB– resistant against vancomycin but susceptible for teicoplanin, VanC– low level resistance to vancomycin, VanD– Moderate to high level resistance against vancomycin and low to moderate resistance to teicoplanin, VanG– low level resistance to vancomycin and susceptible to teicoplanin. In K. pneumoniae, the presence of the blaNDM-1 gene encodes the NDM-1, a potent resistance mechanism. A. baumannii exhibits resistance via two major enzymes: metallo-β-lactamases encoded by blaNDM-1 and oxacillinase-type serine β-lactamases encoded by blaOXA genes responsible for resistance in Gram-negative bacteria and these gens present bacterial chromosomes and in plasmid. Meanwhile, P. aeruginosa is often associated with resistance through the production of the VIM enzyme, encoded by blaVIM.
Natural anti-biofilm agents against microbes and their mechanism
Natural anti-biofilm agents function through several mechanisms, including: (A) inhibition of polymer matrix development, (B) prevention of bacterial adhesion and surface attachment, (C) disruption of extracellular matrix (ECM) production, and (D) suppression of virulence factor expression. This highlights the need for innovative strategies to identify novel quorum sensing inhibitors (QSIs) with minimal side effects. Biofilm formation has been observed in both Gram-positive and Gram-negative bacteria, and various natural extracts have demonstrated potential as effective QSIs. Details of different pathogens and their reported active QSI compounds are as follows.9,10
Pseudomonas aeruginosa
Pseudomonas aeruginosa can be considered as one of the opportunistic microorganisms associated with healthcare-associated infections, including ventilator-associated pneumonia (VAP), infections from ICUs, bloodstream infections and surgical infections. They secrete exopolysaccharides, matrix proteins, extracellular polymeric substances etc. These compounds facilitate the adhesion process, enabling the aggregation of microbial cells and promoting the intercellular interactions necessary for biofilm establishment. Different natural substances have been reported for biofilm inhibition of P. aeruginosa. Saponin, curcumin, eugenol are commonly reported natural agents to control biofilm in P. aeruginosa by the inhibiting autoinducer production. One of the predominantly reported natural agents is garlic extract, which has also been shown to reduce biofilm formation by weakening the QS signal system and virulence factors.11
In a mouse UTI model, garlic extract was tested and reported to potently reduce the quorum sensing system through N-heptylsulfanylacety-l-L-homosrine lactone.12 This metabolite was also reported to suppress the regulators LuxR/LasR. A study conducted by Fu et al.13 focused on the effectiveness of different herbal plants on P. aeruginosa to control six genes reported for biofilm formation. The findings revealed that among the various plant extracts tested, Herba patriniae exhibited notable antibiofilm activity by significantly reducing polysaccharide production. The use of hordenine has a significant effect with a concentration-based reduction in biofilm formation in food-borne pathogen of P. eruginosa.14 Isovitexin and parthenolide from Dendrobium chrysotoxum controlled the formation of virulence factors associated with P. aeruginosa thereby controlling microbial biofilm formation.15 According to Kazemian et al.,16 Chamaemelum nobile demonstrated antibiofilm activity against the Pseudomonas aeruginosa PAO1 strain, which was isolated from hospitalized patients.
Escherichia coli
The commonly reported nosocomial infection from E. coli is UTIs. BSIs are also reported from E. coli. These microorganisms have the capacity to produce virulent toxins that may induce severe toxicological effects and pose significant risk to life. Ginkgo biloba extract has been shown to inhibit biofilm formation by Escherichia coli O157:H7 isolated from glass, polystyrene, and nylon membrane surfaces, with effective activity observed at a concentration of 100 µg/mL. The reported mechanism reveals that ginkgolic acid downregulates the transcriptional activity of curli and prophage genes leading to a significant reduction in fimbrial production.17 Another researcher reported that cinnamaldehyde suppresses biofilm formation and inhibits the motility of E. coli. Phloretin is abundantly present in apples and is reported to control biofilm formation in E. coli O157:H7 by reducing motility. The compound also demonstrated a significant reduction in the adhesion of the same strain to human epithelial cells, potentially limiting its colonization and pathogenicity. Phloretin has been reported to suppress the expression of several key genes in Escherichia coli O157:H7 biofilm cells, including toxin genes hlyE and stx,2 autoinducer-2 importer genes (lsrACBDF), curli fiber genes (csgA and csgB), and certain prophage-associated genes.18
Staphylococcus aureus
Methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant Staphylococcus aureus (VRSA) are among the most concerning pathogens in healthcare settings and are frequently implicated in hospital-acquired infections. S. aureus small colony variants (SCVs) demonstrate frequent phenotypic variation, that plays a pivotal role in their resistance to standard treatments. In immunocompromised people, infections are commonly observed with medical devices. The commonly reported genes associated with biofilm formation in S. aureus are agr, sarA, saeR/S and arlRS. Cell wall-associated proteins also help in the binding on biotic or abiotic surfaces through electrostatic and hydrophobic interactions. Opuntia ficus-indica caused a major reduction in biofilm formation in S. aureus. Methanolic extracts prepared from dried plant material were utilized in the study and demonstrated a significant reduction in extracellular polymeric substance (EPS) production in Staphylococcus aureus, along with decreased specific biofilm formation (SBF). The presence of flavonoids for inhibition. The secondary metabolite usnic acid derived from lichen has been shown to exhibit anti-biofilm activity and is widely incorporated into medical device coatings to mitigate S. aureus colonization.19 Ocimum lamiifolium, Catharanthus roseus, Rosmarinus officinalis, Moringa stenopetala and Azadirachta indica were reported with anti-biofilm activity against methicillin-resistant S. aureus. The ethanolic extract of dried plant samples demonstrated a substantial decrease in slime production, which was attributed to the inhibition of S. aureus biofilm formation.20 Phloretin, an antioxidant, has also been reported to have biofilm inhibition activity in S. aureus RN4220 through targeting efflux proteins. The ethanol extract of Rhodomyrtus tomentosa Hassk. leaves demonstrated inhibitory activity against biofilm formation by Staphylococcus aureus.21
Streptococcus mutants
Biofilm formation by dental carries is considered a worldwide issue related to oral cavity formation in children and adults. Lack of proper treatment and care leads to enhanced pathogenicity. It is reported that S. mutants is the primary etiologic agent involved in this condition. This organism plays a significant role in the biofilm formation on the oral mucosa and tooth enamel. Demineralization of the tooth will also take place by the produced acids from pathogens. Glucosyltransferase B will be produced by S. mutants, an extracellular enzyme responsible for glucan formation from sucrose sucrose-rich diet. The produced glucan supports the attachment of pathogens to tooth enamel adhesion and produces much resistance against the body’s immunity mechanism. The Camellia sinensis extract is formulated against biofilm formation on a tooth by S. mutants as a natural agent. This plant is rich in epigallocatechin (EGC), epicatechin gallate (ECG), epicatechin (EC) and epigallocatechin gallate (EGCG). EGCG is reported to inhibit or reduce the activity of Glucosyltransferase B by suppressing the activity of gtfs (gtfB, gtfC, gtfD), which reduces biofilm inhibition.
Honey is reported as one of the effective antibiofilm agents against S .mutants. The inhibition mechanism reported that honey is rich in hydrogen peroxide, flavonoids, and methylglyoxal and all of these have antibiofilm activity. Similarly, the high concentration of sugar content will develop hypertonic conditions that will lead to the lysis of the cell wall of pathogens. Propolis is a natural sticky agents extracted from honeybees (Apis mellifera L.) and reported with multiple applications. Propolis typically consists of approximately 50% plant resins, 30% waxes, 10% essential oils, 5% pollen, and 5% miscellaneous compounds.22 Propolis controls biofilm formation by S. mutants by suppressing the activity of genes such as LuxS and Gtf-B.23 Ethanol extract of propolis (EEP) is widely reported with anti-biofilm activities. Apigenin is a type of flavonoid known as 4′,5,7-trihydroxyflavone and present in honey. Apigenin exhibits multiple bioactive activities and the most effective report is that it can control the expression of gtf gene. It also reduces the accumulation and polysaccharide production by the pathogens. The gene gbpC is associated with biofilm formation through adhesion and dextran production. The suppression of the same gene by apigenin was reported with reduced biofilm activity.24
Advanced method of biofilm coating on devices
The adhesion of microorganisms is substantially influenced by the physicochemical characteristics of both the microbial surface and the materials utilized in substrate fabrication. Hydrophobic and electrostatic surface properties constitute key determinants that modulates the dynamics and effectiveness of the finishing process in the substrates. The use of anti-biofilm agents, mechanical methods and surface modifications are the common approaches used in finishing methods of medical devices. Commonly used methods of finishing implants are discussed below. The laser surface texturing technique functions as a microstructural approach for biofilm inhibition by regulating surface, specifically specifically size and shape features to prevent biofilm establishment. This is mainly achieved by modifying the surface texture and roughness of the material. Nano materials coated with medicine will reduce the attachment of microbes and control biofilm formation. Plasma immersion ion implantation (PIII) or pulsed plasma dropping (pulsed PIII) is an advanced method that accelerates ions through changing voltage of surface to reduce or inhibit biofilm formation.
Table (1):
Mode of action of anti- biofilm agents30
Process |
Mechanism |
|---|---|
Inhibition of biofilm attachment |
Arrest the attachment of flagella like cell appendages. This process is aided through the surface modification using anti-bactericidal agents, e.g. polysaccharides from Antarctic sponge.25 |
Breaking or interfering signals |
Inhibit N-acyl homo-serine lactones (AHL-QS) system.26 |
Inhibition of cyclic di-guanosine monophosphate (c-di-GMP) |
Cyclic di-GMP plays a key regulatory role in biofilm formation in both Gram-positive and Gram-negative bacteria. The enzyme diguanylate cyclase synthesizes c-di-GMP. Inhibition of the activity of this enzyme will terminate biofilm formation.27 |
Inhibition of genetic bio diversification |
Arresting the horizontal gene transfer.28 |
Biofilm dispersal inducers |
Dispersal of biofilm leads to the attachment of cells to new surface.29 |
Table (2):
Quorum quenching agents31
Group /class of agents |
Examples |
|---|---|
Antimicrobial peptide |
Subtilosin,9 Hordenine,10 Falcarindiol,11 |
Enzymatic inhibitors |
Avibactam,14 7 hydroxytropolone,15 Plazomicin16 |
Efflux pump inhibitors |
MC-207, 110 (17a), MC-04124 (17b) |
Quaternaryammonium compounds |
MC-207, MBX231919 |
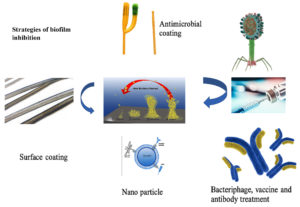
Tables 1 and 2 summarizes the action of different biofilm inhibitors and reported quorum quenching agents. Figure 2 illustrated reported treatments or strategies to control biofilm formation.
Hydrophilic coatings on implants
Negatively charged zwitterionic MEDSAH surfaces reduced biofilm formation of P. aeruginosa by inhibiting the motility and attachment. Hyaluronic acid is a hydrophilic polymer and poly-N-vinylpyrrolidone is frequently used as a coating agent on silicon catheters and silicon shunts respectively and reported with a reduction in the adhesion of Streptococcus epidermis.32 Ureteral stents and hydrogel coating are widely used because of their anti-biofilm properties and hydrophilic characteristics.32 The application of Heparin-functionalized coatings are reported to exhibit anti-adhesive properties that inhibit the initial attachment and proliferation of microbial colonies. Heparin reduces the deposition of fibronectin on the vascular catheter and turns the surface to a negative charge. This property is highly useful in reducing catheter-related infections.33 Silver alloy-coated latex catheters are presently utilized in clinical settings and have been shown to reduce biofilm formation. Nitrofurazone-coated silicon-latex catheters are also widely used in hospitals to control biofilm formation. The ethylene–vinyl acetate and polyethylene oxide demonstrated sustained antimicrobial efficacy for up to 30 days against Klebsiella pneumoniae and Proteus vulgaris. Catheter impregnated with minocycline and rifampicin showed a reduced rate of Gram-positive bacteria-associated biofilm infection.34
Antimicrobial finished urinary catheters
Frequently identified biofilm-forming microorganisms on catheters include Enterococcus faecalis, Pseudomonas aeruginosa, Escherichia coli, and Proteus mirabilis. To control this mixed microbial population silver alloy-coated catheter is most advisable.35 Other studies reported effective coating with nitrofurazone, gentamicin, rifampicin and norofloxacin. Silver-alloy-latex catheters are promising agents with significant inhibition and few of them are modified with nitrofurazone coating.36 Both are reported to be effective in controlling biofilm formation. Catheters coated with a combination of copolymers, including ethylene-vinyl acetate (EVA) and polyethylene oxide (PEO), have demonstrated effective biofilm inhibition for up to 30 days against Klebsiella pneumoniae and Proteus vulgaris. Additionally, coatings with minocycline and rifampicin (MR) have been shown to significantly reduce adhesion of Gram-positive bacteria. In clinical settings, MR-coated catheters were also associated with a notable decrease in Gram-negative infections, such as bacteriuria and candiduria.37
Antimicrobial impregnated orthopedic implants
Despite adherence to strict surgical protocols and antibiotic prophylaxis, the knee and hip remain the most affected sites in joint replacement-associated infections. For several decades, research has been directed toward developing antibiotic-loaded polymethylmethacrylate (PMMA) bone cements and spacers. These materials are tailored to adsorb and subsequently release antibiotics that is facilitated by the aqueous solubility of the incorporated therapeutic agents. Many antibiotic-coated implants and their combinations are under investigation. The most suitable method selected was gentamycin-loaded polymethylmethacrylate beads with novel biodegradable gentamycin-releasing polytrimethylene carbonate. This can inhibit the growth of S. aureus for around 80% up to 14 days. It is one of the advisable systems for local treatment of osteomyelitis.
Antimicrobial finished central venous catheters (CVCs)
The incorporation of chlorhexidine and silver sulfadiazine as dual coatings on CVCs confers temporary resistance to microbial colonization, thereby offering short-term anti-biofilm functionality. Minocycline and rifampicin (MR)-coated CVCs were effective for ICU patients for three days. It was also reported that there was a reduction in catheter-related bloodstream infections (CRBSIs) up to 60 days.38 Similarly, a decrease in Candida sp. colonization was also reported with MR coating on ICU patients for three days. At the same time, it was also observed silver alloy or any metal alloy-coated MR CVCs are not effective in reducing CRBSIs.
Biofilms are identified as potential threats in health care due to their increased resistance to antibacterial treatments. Treatment and control of biofilm are causing a substantial challenge on human health care. As indicated in this review, natural sources serves as the best libraries for screening potent anti-biofilm agents. To date, numerous studies have reported effective pre-clinical findings to control biofilm formation through gene suppression. Many of these studies have identified active metabolites that are progressing through the regulatory approval process. Future research must aim to deeply investigate the biological mechanisms underlying biofilm formation to develop innovative and effective strategies to suppress the current scenario. Gaining a better understanding of the molecular mechanism involved in microbial colonisation will facilitate the fabrication of medicated devices and pharmaceuticals to provide resistance against colonization. Similarly, advancement of rapid detection technologies is also essential to detect early colonization, enabling timely preventive measures. Even though numerous scientific and technological approaches were reported in past decades, many have proven ineffective or have yet to be implemented. To address this critical issue, collaboration between research institutes, healthcare sectors and industries is very important. This partnership can accelerate the development of advanced solutions for combating biofilm-related infections.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
Not applicable.
- Sharma S, Kaushik V, Tiwari V. Role of biofilms in hospital-acquired infections (HAIs). In: Das S, Kungwani NA (eds). Understanding Microbial Biofilms Fundamentals to Applications. Academic Press. 2023:209-245.
Crossref - Alberti C, Brun-Buisson C, Burchardi H, et al. Epidemiology of sepsis and infection in ICU patients from an international multicentre cohort study. Intensive Care Med. 2002;28(2):108-121.
Crossref - Klevens RM, Edwards JR, Richards Jr CL, et al. Estimating health care-associated infections and deaths in US hospitals, 2002. Public Health Rep. 2007;122(2):160-166.
Crossref - Munson EL, Heard SO, Doern GV. In vitro exposure of bacteria to antimicrobial impregnated-central venous catheters does not directly lead to the emergence of antimicrobial resistance. Chest. 2004;126(5): 1628-1635.
Crossref - Hentzer M, Wu H, Andersen JB, et al. Attenuation of Pseudomonas aeruginosa virulence by quorum sensing inhibitors. EMBO J. 2003;22(15):3803-3815.
Crossref - Holm AB. Institutional context and e-recruitment practices of Danish organizations. Employee Relations, 2014;36(4):432-455
Crossref - Aslam S. Effect of antibacterials on biofilms. AJIC. 2008;6(10):S175-e9.
Crossref - Lin Q, Deslouches B, Montelaro RC, Di YP. Prevention of ESKAPE pathogen biofilm formation by antimicrobial peptides WLBU2 and LL37. Int J Antimicrob Agents. 2018;52(5):667-672
Crossref - Rutherford ST, Bassler BL. Bacterial quorum sensing: its role in virulence and possibilities for its control. Cold Spring Harb. Perspect Med. 2012;2(11): a012427.
Crossref - Papenfort K, Bassler BL. Quorum sensing signal–response systems in Gram-negative bacteria. Nat Rev Microbiol. 2016;14(9):576-588.
Crossref - Harjai K, Kumar R, Singh S. Garlic blocks quorum sensing and attenuates the virulence of Pseudomonas aeruginosa. FEMS Immunol. Med. Microbiol 2010;58(2):161-168.
Crossref - Persson T, Hansen TH, Rasmussen TB, Skindersf ME, Givskov M, Nielsen J. Rational design and synthesis of new quorum-sensing inhibitors derived from acylated homoserine lactones and natural products from garlic. Org Biomol Chem. 2005;3(2):253-262.
Crossref - Fu B, Wu Q, Dang M, et al. Inhibition of Pseudomonas aeruginosa biofilm formation by traditional Chinese medicinal herb Herba patriniae. Biomed Res Int.
2017(1):9584703.
Crossref - Zhou JW, Luo HZ, Jiang H, Jian TK, Chen ZQ, Jia AQ. Hordenine: a novel quorum sensing inhibitor and antibiofilm agent against Pseudomonas aeruginosa. J Agric Food Chem. 2018;66(7):1620-1628.
Crossref - Kalia M, Yadav VK, Singh PK, Sharma D, Narvi SS, Agarwal V. Exploring the impact of parthenolide as anti-quorum sensing and anti-biofilm agent against Pseudomonas aeruginosa. Life Sci. 2018;199:96-103.
Crossref - Kazemian H, Ghafourian S, Heidari H, et al. Antibacterial, anti-swarming and anti-biofilm formation activities of Chamaemelum nobile against Pseudomonas aeruginosa. Rev Soc Bras Med Trop. 2015;48(4):432-436.
Crossref - Lee JH, Kim YG, Ryu SY, Cho MH, Lee J. Ginkgolic acids and Ginkgo biloba extract inhibit Escherichia coli O157: H7 and Staphylococcus aureus biofilm formation. Int J Food Microbiol. 2014;174:47-55.
Crossref - Lee JH, Regmi SC, Kim JA, et al. Apple flavonoid phloretin inhibits Escherichia coli O157: H7 biofilm formation and ameliorates colon inflammation in rats. Infect. Immun. 2011;79(12):4819-4827.
Crossref - Zheng Y, He L, Asiamah TK, Otto M. Colonization of medical devices by Staphylococci. Environ Microbiol. 2018;20(9):3141-3153.
Crossref - Manilal A, Sabu KR, Shewangizaw M, et al. In vitro antibacterial activity of medicinal plants against biofilm-forming methicillin-resistant Staphylococcus aureus: efficacy of Moringa stenopetala and Rosmarinus officinalis extracts. Heliyon. 2020;6(1):1-8.
Crossref - Rudrapal M, Sarkar B, Deb P, Bendale A, Nagar A. Addressing antimicrobial resistance by repurposing polyphenolic phytochemicals with novel antibacterial potential. In: Rudrapal M (eds.) Polyphenols: Food, Nutraceutical, and Nanotherapeutic Applications. John Wiley & Sons. 2023;260-289.
Crossref - Nassar HM, Li M, Gregory RL. Effect of honey on Streptococcus mutans growth and biofilm formation. AEM. 2012;78(2):536-540.
Crossref - Pensantes-Sangay SJ, Calla-Poma RD, Requena-Mendizabal MF, Alvino-Vales MI, Millones-Gomez PA. Chemical composition and antibacterial effect of Plantago Major Extract on periodontal pathogens. Pesqui Bras Odontopediatria Clín Integr. 2020;20:1-10.
Crossref - Ramalingam K, Amaechi BT. Phytochemicals: Their Therapeutic Potential Against Dental Caries. In: Kamalanand K, Thayumanavan B, Jawahar PM (eds). Computational Techniques for Dental Image Analysis. Advances in Medical Technologies and Clinical Practice Book Series.; 2018:238-275.
Crossref - Asadi A, Razavi S, Talebi M, Gholami M. A review on anti-adhesion therapies of bacterial diseases. Infection. 2019;47(1):13-23.
Crossref - Case RJ, Labbate M, Kjelleberg S. AHL-driven quorum-sensing circuits: their frequency and function among the Proteobacteria. ISME J. 2008;2(4):345-349.
Crossref - Hengge R, Grundling A, Jenal U, Ryan R, Yildiz F. Bacterial signal transduction by cyclic di-GMP and other nucleotide second messengers. J Bacteriol. 2016;198(1):15-26.
Crossref - Martin-Rodriguez AJ, Romling U. Nucleotide second messenger signaling as a target for the control of bacterial biofilm formation. Curr Top Med Chem. 2017;17(17):1928-1944
- Blazanin M, Turner PE. Community context matters for bacteria-phage ecology and evolution. ISME J. 2021;15(11):3119-3128.
Crossref - Nadar S, Khan T, Patching SG, Omri A. Development of antibiofilm therapeutics strategies to overcome antimicrobial drug resistance. Microorgs. 2022;10(2):303.
Crossref - Francolini I, Donelli G. Prevention and control of biofilm-based medical-device-related infections. FEMS Immunol Med Microbiol. 2010;59(3):227-238.
Crossref - John T, Rajpurkar A, Smith G, Fairfax M, Triest J. Antibiotic pretreatment of hydrogel ureteral stent. J Endourol. 2007;21(10):1211-1216.
Crossref - Appelgren P, Ransjo U, Bindslev L, Espersen F, Larm O. Surface heparinization of central venous catheters reduces microbial colonization in vitro and in vivo: results from a prospective, randomized trial. Crit Care Med. 1996;24(9):1482-1489.
Crossref - Jain G, Allon M, Saddekni S, Barker JF, Maya ID. Does heparin coating improve patency or reduce infection of tunneled dialysis catheters? Clin J Am Soc Nephrol. 2009;4(11):1787-1790.
Crossref - Darouiche RO, Berger DH, Khardori N, et al. Comparison of antimicrobial impregnation with tunneling of long-term central venous catheters: a randomized controlled trial. Ann Surg. 2005;242(2):193-200.
Crossref - Johnson JR, Kuskowski MA, Wilt TJ. Systematic review: antimicrobial urinary catheters to prevent catheter-associated urinary tract infection in hospitalized patients. Ann Intern Med. 2006;144(2):116-126.
Crossref - Darouiche RO, Mansouri M D, Gawande PV, Madhyastha S. Antimicrobial and antibiofilm efficacy of triclosan and Dispersin B® combination. J Antimicrob Chemother. 2009;64(1):88-93.
Crossref - Sampath LA, Tambe SM, Modak SM. In vitro and in vivo efficacy of catheters impregnated with antiseptics or antibiotics: evaluation of the risk of bacterial resistance to the antimicrobials in the catheters. ICHE. 2001;22(10):640-646.
Crossref
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.