ISSN: 0973-7510
E-ISSN: 2581-690X
Antimicrobial resistance (AMR) is an increasing global concern, with Acinetobacter spp. being among the most common causes of difficult-to-treat hospital-acquired infections. Catheter-associated urinary tract infections (CAUTI) are common among hospitalized patients, and they worsen when caused by multidrug-resistant Acinetobacter spp. Such infections lead to extended sickness, increased death, and higher healthcare expenses. According to recent WHO and ICMR publications (2023-2024), carbapenem resistance in Acinetobacter is more than 70% in India, demonstrating the severity of the situation. The goal of this study was to determine the prevalence of MDR Acinetobacter spp. infections in CAUTI patients, examine their drug resistance patterns, and identify associated risk factors. The study comprised 250 CAUTI patients hospitalized to Sree Balaji Medical College and Hospital in Chennai between November 2024 and June 2025. Patient demographic and clinical information, as well as admission location and catheterization details, were obtained. To examine resistance profiles, isolates were selected and evaluated for antimicrobial susceptibility. Out of 250 CAUTI patients, 95 (38%) had urine cultures positive for multidrug-resistant Acinetobacter spp. Infections were most prevalent in the intensive care unit (50%), followed by medical wards (35%), and surgical wards (15%). Imipenem and meropenem showed the strongest resistance (80% and 78%, respectively), although colistin resistance was very low (10%). Prolonged catheterization (75%), ICU stay (60%), and poor hand hygiene compliance (50%), were the most significant risk factors for infection. MDR Acinetobacter spp. poses a severe difficulty in CAUTI treatment. Effective infection prevention strategies, antibiotic stewardship, and early detection are critical. Alternative medicines, such as phage therapy and combination regimens, should be investigated to lessen the load on healthcare systems.
Acinetobacter, Infections, Resistance, CAUTI, Carbapenem Resistance, Antimicrobial, Intensive Care, Isolates
Antimicrobial resistance (AMR) is one of the most pressing global health issues of the twenty-first century, with the potential to render common diseases incurable and strangle advances in modern medicine. According to the World Health Organization (WHO), if not treated effectively, AMR might kill 10 million people per year by 2050, more than cancer’s current mortality rate.1 According to the WHO Surveillance (2023), Acinetobacter baumannii remains one of the most serious drug-resistant bacteria. One of the most critical issues with AMR is the rapid spread of drug-resistant bacteria in hospitals. Species, particularly Acinetobacter baumannii, have emerged as major health risks in hospitals. They have been linked to dangerous infections such as ventilator-associated pneumonia, bloodstream infections, urinary tract infections, and wound infections, especially in ICU patients, those with recent surgery, catheters, or compromised immune systems.2
This pathogenic bacterium is well-adapted to hospital environments, surviving on dry surfaces for long periods of time and accumulating resistance genes via horizontal gene transfer, making routine antibiotic therapy difficult and sometimes ineffective.3 In 2023, researchers discovered that many Indian hospital strains include OXA-23 and NDM carbapenemase genes. According to WHO data from 2023, catheter-associated urinary tract infections (CAUTIs) are among the most common infections observed in hospitals, accounting for 40%-45% of all hospital-acquired infections worldwide. The risk of acquiring a CAUTI increases dramatically with catheterization duration because prolonged use creates an excellent entry point and surface for multidrug-resistant (MDR) pathogen colonization, notably by Acinetobacter spp.4 This bacterium has caused widespread worry in medical institutions because to its amazing capacity to survive in harsh circumstances, adhere to catheter surfaces, and escape both immune system reactions and antibiotic therapy.
One of Acinetobacter spp. most important virulence traits is their ability to build biofilms on catheter materials, which shield the bacterial community from drugs and immunological responses, resulting in persistent and recurring infections.5 The emergence of carbapenemases, which inhibit carbapenem antibiotics,6 the use of efflux pumps, which remove an extensive variety of antibiotics from the bacterial cell, lowering intracellular concentrations of drugs,7 site changes that prevent antibiotics from adhering effectively,8 and strong biofilm formation, resulting in greater survival and resistance.9 According to recent study (2022-2024), novel genes such as adeJ and bap also assist bacteria break free from biofilms and fight antibiotics.
MDR Acinetobacter spp. infections have serious clinical effects, with mortality rates ranging from 20%-60%, particularly in patients who require long-term hospitalization, have several medical problems, or are in critical care.10 These infections also cause longer hospital admissions, greater use of healthcare resources, and higher overall treatment costs.11 Compounding the problem is the growing resistance to last-resort antibiotics such polymyxins, leaving very few therapeutic alternatives.12 Early recognition of high-risk patients may also help reduce complications, length of hospital stay, and overall healthcare costs.13 Given these challenges, the goal of this study is to look into MDR resistance trends, identify risk factors for bacterial colonization and infection in catheterized patients, and develop specific approaches for preventing, detecting, and treating CAUTIs caused by this life-threatening pathogen.
This study was carried out from November 2024 to June 2025 at Sree Balaji Medical College and Hospital, Chennai. The Institutional Ethics Committee (Approval No: 002/SBMCH/IHEC/2024/2190) has approved the study. Written informed consent was obtained from all participants, and patient confidentiality was maintained throughout the study. Adult patients aged 18 and up with catheter-associated urinary tract infections (CAUTI) and positive urine cultures fulfilling CDC criteria for CAUTI were included in the study. This study only included people who had been catheterized for more than 48 hours. Patients were excluded if their samples revealed mixed conditions, their medical histories were inadequate, or they had begun antibiotics before the urine culture was verified.
Catheter-associated urinary tract infection (CAUTI) was defined as a urinary tract infection occurring in a patient with an indwelling urinary catheter in situ for ≥48 hours, with clinical features of UTI and a positive urine culture, as per CDC criteria. Multidrug-resistant (MDR) was defined as resistance to at least one agent in three or more antimicrobial classes, in accordance with international consensus definitions.
A total of 250 urine samples from catheterised patients which showed growth in culture were included in the study, of which 120 samples were from ICU patients, 85 from medical wards, and 45 from surgical wards. All urine samples underwent routine physical and microscopic examination, assessing appearance, leukocytes, and bacterial presence to support CAUTI diagnosis.
Colistin susceptibility results were interpreted based on MIC values obtained from the automated VITEK-2 system, in accordance with CLSI recommendations.
Sample collection and microbiological analysis
Urine samples were obtained aseptically from patients with indwelling catheters to ensure accuracy and avoid contamination. Bacterial species were detected using both conventional microbiological methods and the automated VITEK-2 (bioMerieux) technology. The Kirby-Bauer disk diffusion method was used to test microorganisms for antibiotic resistance in strict adherence to the Clinical and Laboratory Standards Institute (CLSI) standards for 2024. The antibiotic susceptibility of the isolated Acinetobacter species was tested against a panel of routinely used antibiotics. Imipenem and meropenem were employed as carbapenems, colistin as a polymyxin, and antibiotics such as ciprofloxacin, amikacin, and gentamicin. This option contributed to determining the resistance pattern and recommending relevant treatment options. To ensure that the test results were correct, quality control was performed with standard bacterial strains, Escherichia coli ATCC 25922 and Pseudomonas aeruginosa ATCC 27853.
Data collection and statistical analysis
The patient’s age, gender, catheterization time, length of stay in the ICU, and adherence to hand hygiene guidelines were all recorded. Resistance was assessed by determining the proportion of CAUTI cases caused by multidrug-resistant Acinetobacter spp. The data was analysed using SPSS version 26.0. Statistical significance was determined using the Chi-square test (p-values < 0.05).
Out of 250 positive culture samples from CAUTI patients, 95 samples (38%) yielded culture-positive growth of multidrug-resistant Acinetobacter spp. Poor hand hygiene was assessed through infection control audits, direct observations, and documented non-compliance with WHO guidelines.
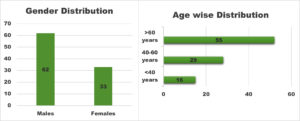
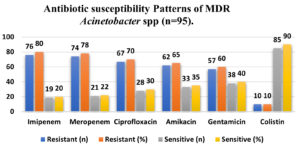
On MacConkey agar, the isolates formed non-lactose-fermenting colonies that were smooth, convex, and transparent. The organism tested positive for citrate, negative for indole, and negative for urease. The reaction on triple sugar iron (TSI) agar had an alkaline slope with an alkaline butt (K/K), and no gas or hydrogen sulfide were formed. The isolates did not move and did not ferment mannitol. Final species identification was confirmed using the automated VITEK-2 (bioMerieux) system. Antimicrobial susceptibility testing with the Kirby-Bauer disc diffusion method revealed resistance to multiple antibiotic classes. The findings were interpreted in accordance with Clinical and Laboratory Standards Institute (CLSI) guidelines. Among the 95 multidrug-resistant Acinetobacter spp. isolates, the highest proportion was recovered from the ICU (50%), followed by medical wards (35%) and surgical wards (15%). Figure 1 demonstrates that males accounted for the vast majority of affected individuals (65%). The age distribution demonstrated that 55% of instances occurred in persons over the age of 60, showing that elderly men were more vulnerable. The antimicrobial resistance profile (Figure 2) revealed considerable carbapenem resistance, with 80% of isolates resisting imipenem and 78% resisting meropenem. Ciprofloxacin (70%), amikacin (65%), and gentamicin (60%) all demonstrated moderate resistance. Colistin was the most effective medication, with resistance observed in only 10% of samples.
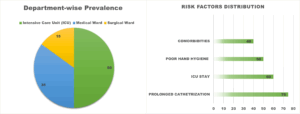
Figure 3. Department-wise Prevalence of MDR Acinetobacter spp.: (ICU: 50%, Medical Ward: 35%, Surgical Ward: 15%) & Distribution of risk factors: Prolonged Catheterization (75%), ICU stay (60%), Poor Hand Hygiene (50%), and Comorbidities (40%)
MDR Acinetobacter infections were most common in the intensive care unit (50%), followed by the medical ward (35%) and the surgical ward (15%), as illustrated in Figure 3, and also depicts an analysis of associated risk factors that identified prolonged catheterization lasting more than seven days as the leading contributor (75%), followed by ICU admission (60%), poor hand hygiene compliance (50%), and comorbidities such as diabetes mellitus (40%). Table 1 displays broad antibiotic susceptibility profiles, revealing significant resistance across most antibiotic classes, with colistin maintaining relative efficacy. Table 2 summarizes demographic and clinical risk factors for MDR infections, emphasizing substantial associations with age, gender, ward distribution, and comorbidities. All comparisons were statistically significant (p < 0.05). These findings emphasize the need of identifying high-risk patients early on, as well as the effectiveness of strict infection control strategies in lowering the prevalence of MDR infections among catheterized patients. These findings are consistent with national AMR surveillance findings in India (ICMR 2024).
Table (1):
Antibiotic susceptibility Patterns of MDR Acinetobacter spp. (n = 95)
Antibiotic |
Resistant (n) |
Resistant (%) |
Sensitive (n) |
Sensitive (%) |
|---|---|---|---|---|
Imipenem |
76 |
80 |
19 |
20 |
Meropenem |
74 |
78 |
21 |
22 |
Ciprofloxacin |
67 |
70 |
28 |
30 |
Amikacin |
62 |
65 |
33 |
35 |
Gentamicin |
57 |
60 |
38 |
40 |
Colistin |
10 |
10 |
85 |
90 |
Table (2):
Distribution and Statistical Association of MDR isolates based on Gender, Age Group, Ward Distribution, and Risk Factors
| Variable | Category (within MDR) | Frequency (n) | Percentage (%) | P-value |
|---|---|---|---|---|
| Gender | Male | 62 | 65% | 0.012 |
| Female | 33 | 35% | ||
| Age Group | >60 years | 52 | 55% | <0.001 |
| 40-60 years | 28 | 29% | ||
| <40 years | 15 | 16% | ||
| Ward Distribution | ICU | 48 | 50% | <0.0001 |
| Medical Ward | 33 | 35% | ||
| Surgical Ward | 14 | 15% | ||
| Prolonged Catheterization | Yes (>7days) | 71 | 75% | <0.0001 |
| No (<7days) | 24 | 25% | ||
| ICU Stay | Yes | 57 | 60% | <0.0001 |
| No | 38 | 40% | ||
| Poor Hand Hygiene | Yes | 48 | 51% | <0.0001 |
| No | 47 | 49% | ||
| Comorbidities | Present | 38 | 40% | <0.001 |
The present study highlights the significant burden of multidrug-resistant (MDR) Acinetobacter spp., among catheter-associated urinary tract infections (CAUTIs in hospitalized patients. MDR Acinetobacter accounted for 38% of culture-confirmed CAUTI cases, underscoring its growing importance as a nosocomial pathogen in urinary tract infections. This finding reflects the increasing prevalence of difficult-to-treat infections in tertiary care settings, particularly in patients requiring prolonged hospitalization and invasive devices.
A higher proportion of MDR Acinetobacter infections was observed in the intensive care unit (50%), compared to medical (35%) and surgical wards (15%). ICU patients are particularly vulnerable due to severe underlying illness, prolonged catheterization, frequent invasive procedures, and greater exposure to broad-spectrum antibiotics. These factors collectively promote colonization and infection by resistant organisms, explaining the higher ICU prevalence noted in this study.14
The antimicrobial susceptibility profile revealed marked resistance to carbapenems, with resistance rates of 80% to imipenem and 78% to meropenem, indicating widespread carbapenem resistance among the isolates. This severely restricts available treatment options for CAUTI caused by Acinetobacter spp. In contrast, colistin retained relatively good activity, with resistance observed in only 10% of isolates, suggesting that it remains one of the few effective therapeutic options in this setting. However, the presence of even low-level colistin resistance is concerning and highlights the need for cautious use of last-resort antibiotics.
Risk factor analysis demonstrated that prolonged catheterization (>7 days) was the most significant contributor, identified in 75% of MDR cases. Extended catheter use facilitates bacterial adherence and biofilm formation, which protects organisms from host immune responses and antimicrobial agents. ICU stay (60%) further increased the risk, reflecting the cumulative impact of critical illness and intensive medical interventions. Additionally, poor hand hygiene compliance (50%), identified through infection control audit records and observational assessments, emphasizes the continued role of suboptimal infection control practices in the transmission of MDR pathogens. The presence of comorbidities such as diabetes mellitus (40%) also contributed to increased susceptibility due to impaired host defenses.3
Overall, the findings of this study emphasize the need for early identification of high-risk patients, strict adherence to infection prevention measures, and rational antimicrobial use. Targeted surveillance and reinforcement of catheter care protocols may significantly reduce the incidence of MDR Acinetobacter-associated CAUTIs in hospital settings.
Strategies for managing MDR Acinetobacter spp. in CAUTIs
Biofilm control
Anti-biofilm therapies including EDTA, silver-coated catheters, and catheters treated with antimicrobial chemicals can help keep germs from adhering to the surface and forming protective coats (biofilms). To reduce the risk of infection, catheters should be changed on a regular basis, evaluated daily to see if they are still required, and always inserted and managed using sterile techniques.15,16
Alternative therapeutics
Phage therapy, which uses bacteriophages to target specific MDR bacteria, is a promising and highly targeted approach, especially when traditional antibiotics fail. Combination antibiotic therapy, such as colistin plus rifampicin, tigecycline, or sulbactam, has shown synergistic advantages in vitro and in clinical settings, offering an additional option for treating severe infections.17,18 Recent (2024) research show that combining bacteriophage therapy with antibiotics can effectively treat MDR Acinetobacter infections.
Infection control measures
It is vital to organize and execute hospital-wide hand hygiene campaigns, conduct regular training sessions for healthcare staff, and adopt environmental cleaning procedures. Antimicrobial stewardship programs should include multidisciplinary teams that monitor prescribing trends, train staff, and develop facility-specific treatment guidelines.19,20
Implementing CAUTI prevention packages, such as catheter insertion checklists, proper catheter care, early catheter removal protocols, and routine infection monitoring, can assist to lower infection rates. Continuous microbiological surveillance and antibiotic resistance tracking are also required to update empirical therapy procedures and detect resistance trends in the hospital setting.21-23
Challenges and future scope
Because this study was conducted at a single institution, the findings may not be applicable to other settings with different patient demographics or antibiotic use. Long-term multi-hospital research is recommended to gain a better understanding of how Acinetobacter spreads and develops resistance. Furthermore, clinical trials to assess the efficacy and safety of phage therapy, novel antimicrobial combinations, and anti-biofilm techniques are required. Molecular research such as PCR, whole genome sequencing, and transcriptome analysis can disclose more about resistance gene expression, plasmid transfer, and biofilm-associated gene regulation, all of which are important for creating future diagnostics and therapies.24-26 AI-based antibiotic stewardship and rapid molecular tests, introduced in 2023-2025, are assisting hospitals in detecting resistance genes more quickly and reducing antibiotic abuse.27
Limitations of the study
This study has certain limitations. Being a single-centre study, the findings may not be generalizable to other healthcare settings. Molecular characterization of resistance mechanisms was not performed, which could have provided deeper insight into carbapenem and colistin resistance. Additionally, patient outcomes such as mortality and treatment response were not analysed.
Multidrug-resistant Acinetobacter spp. accounted for 38% of catheter-associated urinary tract infection cases, with the highest prevalence observed among patients admitted to intensive care units. The high level of carbapenem resistance observed in this study substantially limits available therapeutic options; however, colistin retained comparatively good in vitro activity. Prolonged duration of catheterization, ICU stay, and poor hand hygiene compliance were identified as significant risk factors. These findings highlight the critical need to strengthen infection prevention measures and antimicrobial stewardship programs to reduce the burden of MDR CAUTIs in hospital settings.
ACKNOWLEDGMENTS
The authors would like to thank the Department of Microbiology and the Central Laboratory of Sree Balaji Medical College and Hospital for their assistance with the research.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
LK designed the study. RP and NN performed sample collection and laboratory analysis. CS supervised the study. RP and NN wrote the manuscript. CS and LK reviewed the manuscript. LK revised the manuscript. All authors read and approved the final manuscript for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This study was approved by the Institutional Human Ethics Committee, Sree Balaji Medical College and Hospital, vide reference number 002/SBMCH/IHEC/2024/2190.
INFORMED CONSENT
Written informed consent was obtained from the participants before enrolling in the study.
- Willemsen A, Reid S, Assefa Y. A review of national action plans on antimicrobial resistance: strengths and weaknesses. Antimicrob Resist Infect Control.2022;11(1):90.
Crossref - Bertagnolio S, Suthar AB, Tosas O, Van Weezenbeek K. Antimicrobial resistance: Strengthening surveillance for public health action. PLoS Med. 2023;20(7):e1004265.
Crossref - Al Fadhli AH, Jamal WY, Rotimi VO. Prevalence of carbapenem-resistant Enterobacteriaceae and emergence of high rectal colonization rates of blaOXA-181-positive isolates in patients admitted to two major hospital intensive care units in Kuwait. PLoS One. 2020;15(11):e0241971.
Crossref - Kubin CJ, Garzia C, Uhlemann A-C. Acinetobacter baumannii treatment strategies: a review of therapeutic challenges and considerations. Antimicrob Agents Chemother. 2025;69(8):e0106324.
Crossref - Xu P, Xu L, Ji H, et al. Analysis and comparison of adverse events of colistin administered by different routes based on the FAERS database. Sci Rep. 2025;15(1):10384.
Crossref - Ahuatzin-Flores OE, Torres E, Chavez-Bravo E. Acinetobacter baumannii, a Multidrug-Resistant Opportunistic Pathogen in New Habitats: A Systematic Review. Microorganisms. 2024;12(4):644.
Crossref - Rangel K, De-Simone SG. Treatment and Management of AcinetobacterPneumonia: Lessons Learned from Recent World Event. Infect Drug Resist. 2024;17:507-529.
Crossref - De Oliveira DMP, Forde BM, Kidd TJ, et al. Antimicrobial Resistance in ESKAPE Pathogens. Clin Microbiol Rev. 2020;33(3):e00181-19.
Crossref - Tristancho-Baro A, Franco-Fobe LE, Ariza MP, et al. Genomic Characterization of Carbapenemase-Producing Enterobacteriaceae from Clinical and Epidemiological Human Samples. Antibiotics (Basel). 2025;14(1):42.
Crossref - Alwazzeh MJ, Algazaq J, Al-Salem FA, et al. Mortality and clinical outcomes of colistin versus colistin-based combination therapy for infections caused by Multidrug-resistant Acinetobacter baumannii in critically ill patients. BMC Infect Dis. 2025;25(1):416.
Crossref - Kyriakidis I, Vasileiou E, Pana ZD, Tragiannidis A. Acinetobacter baumanniiAntibiotic Resistance Mechanisms. Pathogens. 2021;10(3):373.
Crossref - Wu HJ, Xiao ZG, Lv XJ, et al. Drug resistant Acinetobacter baumannii: From molecular mechanisms to potential therapeutics (Review). Exp Ther Med. 2023;25(5):209.
Crossref - Motbainor H, Bereded F, Mulu W. Multi-drug resistance of blood stream, urinary tract and surgical site nosocomial infections of Acinetobacter baumannii and Pseudomonas aeruginosa among patients hospitalized at Felegehiwot referral hospital, Northwest Ethiopia: a cross-sectional study. BMC Infect Dis. 2020;20(1):92.
Crossref - GBD 2021 Antimicrobial Resistance Collaborators. Global burden of bacterial antimicrobial resistance 1990-2021: a systematic analysis with forecasts to 2050. Lancet. 2024;404(10459):1199-1226.
Crossref - Roy S, Chowdhury G, Mukhopadhyay AK, Dutta S, Basu S. Convergence of Biofilm Formation and Antibiotic Resistance in Acinetobacter baumanniiInfection. Front Med (Lausanne). 2022;9:793615.
Crossref - Mishra SK, Baidya S, Bhattarai A, et al. Bacteriology of endotracheal tube biofilms and antibiotic resistance: a systematic review. J Hosp Infect. 2024;147:146-157.
Crossref - Qu J, Zou J, Zhang J, Qu J, Lu H. Phage therapy for extensively drug resistant Acinetobacter baumanniiinfection: case report and in vivo evaluation of the distribution of phage and the impact on gut microbiome. Front Med (Lausanne). 2024;11:1432703.
Crossref - Yehya A, Ezzeddine Z, Chakkour M, et al. The intricacies of Acinetobacter baumannii: a multifaceted comprehensive review of a multidrug-resistant pathogen and its clinical significance and implications. Front Microbiol. 2025;16:1565965.
Crossref - Li Y, Zhang J, Gu Y, Wang L, Hu J. Nosocomial, Healthcare-Associated, and Community-Acquired Acinetobacter baumanniiin China: Clinical Characteristics, Antimicrobial Resistance Patterns and Risk Factors Associated with Carbapenem Resistance. Infect Drug Resist. 2024;17:4089-4099.
Crossref - Asmare, Z, Awoke, T, Genet, C. et al.Incidence of catheter-associated urinary tract infections by Gram-negative bacilli and their ESBL and carbapenemase production in specialized hospitals of Bahir Dar, northwest Ethiopia. Antimicrob Resist Infect Control. 2024;13(1):10.
Crossref - Bouhrour N, Nibbering PH, Bendali F. Medical Device-Associated Biofilm Infections and Multidrug-Resistant Pathogens. Pathogens. 2024;13(5):393.
Crossref - Na SH, Eom JS, Seo YB, et al. Impact of Infection Prevention Programs on Catheter-Associated Urinary Tract Infections Analyzed in Multicenter Study. J Korean Med Sci. 2024;39(18):e151.
Crossref - Huang H, Huang L, Yan S, et al. A bundle-based approach on catheter-associated urinary tract infection: a multi-center study in Chinese tertiary hospitals. BMC Infect Dis. 2025;25(1):248.
Crossref - Rastegar S, Skurnik M, Tadjrobehkar O, et al. Synergistic effects of bacteriophage cocktail and antibiotics combinations against extensively drug-resistant Acinetobacter baumannii. BMC Infect Dis. 2024;24(1):1208.
Crossref - Yu L, Zhao Y, Zhang S, et al. Antimicrobial resistance and virulence factors analysis of a multidrug-resistant Acinetobacter baumannii isolated from chickens using whole-genome sequencing. BMC Microbiol. 2024;24(1):526.
Crossref - Luchian N, Salim C, Condratovici AP, et al. Episode- and Hospital-Level Modeling of Pan-Resistant Healthcare-Associated Infections (2020-2024) Using TabTransformer and Attention-Based LSTM Forecasting. Diagnostics (Basel). 2025;15(17):2138.
Crossref - Harandi H, Shafaati M, Salehi M, et al. Artificial intelligence-driven approaches in antibiotic stewardship programs and optimizing prescription practices: A systematic review. Artif Intell Med. 2025;162:103089.
Crossref
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.