ISSN: 0973-7510
E-ISSN: 2581-690X
This study assessed antimicrobial prescription at our institute to promote rational antimicrobial use and implement customized antimicrobial stewardship programs. This study is a cross sectional point-prevalence survey on antimicrobial utilisation conducted at HAH Centenary hospital, New Delhi in April, 2019, over a period of 3 days. All in-patients were included in the analysis. However, neonates, emergency room patients and palliative care patients, orders for anti-viral, anti-fungal, anti-tubercular and anti-parasitic medications were excluded. SPSS version 13.0 was planned to be used for analysis of the statistical data. The number of patients admitted at the point of time of our survey was 217, out of which 101 (46.54%) patients were receiving antimicrobial agents (AMA) with 160 (73.73%) AMAs prescribed. 50 patients (49.5%) were prescribed AMAs for infections, whereas, 49 patients (48.51%) were prescribed AMAs prophylactically and 2 (1.98%) patients received AMAs for reasons not defined. The intensive care units exhibited 93.33% patients receiving antimicrobials. The most common indication was intra-abdominal infections (32.40%). The most commonly used antibiotics were beta-lactam antibiotics (60.62%). The study suggests a high rate of antimicrobial use and highlights areas for intervention for rational antimicrobial use. We propose to sensitise the government on initiating a national antimicrobial stewardship program such as the Global Point Prevalence Survey and facilitate evidence-based antimicrobial practice.
Antimicrobial utilization, Antimicrobial stewardship, Antimicrobial resistance, Point prevalence survey
Antibiotic resistance is a matter of grave concern globally.1 Worldwide rise in the prevalence of antimicrobial resistance can be linked to higher numbers of patient morbidity and mortality.1,2 This pattern stands true especially in the case of developing countries like India which has one of the highest numbers in terms of antimicrobial resistance.3 It is well known that misuse of antimicrobials like prescribing them very frequently or for longer durations can play a major role in the development and spread of antimicrobial resistance.4 A report from 2010 shows that India is the largest consumer of antibiotics being utilised for human health, recording a large number of 12.9 x 10^9 units (10.7 units per person).5 It is also worrying to note that the study suggests nearly half of these prescriptions were advised unnecessarily.6 The major problem when it comes to antibiotic resistance includes a reduction in the ‘lifespan’ of available antimicrobials which ultimately makes treatment difficult due to limited options. This problem has been worsened further as very few new antimicrobial drugs have been developed for human use in the last three decades. Development of resistant organisms suggests that misuse of antimicrobials could result in damage to the health of patients who have not even been exposed to them. Therefore, it is absolutely necessary for healthcare systems to incorporate a conservative approach and preservation of currently available drugs should be a major priority. Rational and judicious use of antimicrobials has certainly been addressed by governments which also includes the Ministry of Health, India. Many recommendations have been made which also includes the development and implementation of antimicrobial stewardship along with monitoring of drug consumption in hospitals. It is very likely that such interventions will definitely decrease antimicrobial resistance, unwanted side-effects and medical costs.7, 8 A point prevalence survey can be defined as the prevalence calculated at a particular point in time. It offers cross-sectional quantitative information about patterns of drug utilisation.9
A major limitation when it comes to the drafting and implementation of successful antimicrobial stewardship programs is that there is very limited quantitative and qualitative data about antibiotic prescriptions. It is therefore necessary to develop competent surveillance methods that can monitor and record antimicrobial utilisation and emergence of any resistance.10 This should also be followed by audits and feedback must be obtained on prescribing practices which can ultimately improve key stewardship changes like prescription of empirical treatment as per protocols.11,12,13
Taking this background information into consideration, the survey was designed with the primary target to judge the trends involved in antimicrobial use at our institute to enable judicious utilisation of antimicrobials and reduce possible drug reactions. The survey was also devised to assess the adherence of prescription patterns to national guidelines and to recommend crucial points through the initiation of a customised antimicrobial stewardship program (ASP). To our knowledge, this is the first comprehensive comparative analysis of Point Prevalence Surveys conducted across India, reflecting the national antimicrobial utilisation status.
Study design and setting
This was a cross-sectional point prevalence survey of antimicrobial use conducted at HAH Centenary hospital, New Delhi. The hospital is a multi-speciality, tertiary-care, NABH accredited, teaching hospital; with 530 bed capacity across 11 clinical departments and 15 wards. The survey was conducted in April 2019, over 3 days after due approval and permission from the institutional ethics committee. The survey included all in-patients of both genders and all age groups receiving systemic antimicrobial agents on the day of the survey. Neonates, emergency room patients who were not admitted and patients on palliative care were excluded from the survey. Orders for anti-viral, anti-fungal, anti-tubercular and anti-malarial were excluded from the survey. The clinical departments were divided into 6 groups for the survey data analysis; Departments of Medicine (General Medicine and Respiratory Medicine), Departments of Surgery (General Surgery and Orthopaedics), Obstetrics and Gynaecology, Department of Paediatrics, Intensive Care Units (Medical and Surgical ICU) and Others (Psychiatry, ENT, Ophthalmology and Dermatology).
Study tool and validation of the study tool
A basic PPS tool was devised on the basis of point prevalence survey methodology on antibiotic use from WHO-version 1.1 for reference.14 The survey was conducted by faculty members from the Department of Pharmacology, clinicians and resident doctors stationed in the hospital wards. A survey skill exercise was organised before the study to train all the survey members regarding the methodology of the survey. The survey was conducted at one point in time in a particular ward, commencing at 10:00 AM, over 3 days. The data was collected using two forms, one for ward-level data; recording the denominators, such as the total number of patients in the ward, and the other for patient-level data. The patient-level data was collected in a well-structured case record form, which included patient characteristics, all information regarding the antimicrobials prescribed, the indication for treatment, the basis for diagnosis and the rationale behind the choice of antimicrobial. The survey was coordinated by the team leader from the Pharmacology department and monitored by the project monitor from the clinical team. The head of the Department of Pharmacology and hospital Medical Superintendent supervised the survey to maintain the quality of the data collection process and patient confidentiality.
Identification of study participants
The indications for the prescription of antimicrobials were classified into two classes. These classes were infection and prophylaxis. The infection class can further be divided into definitive or empiric. Definitive therapy is treatment which is started when the infection site or causative microorganism is identified using microbiological tests like culture and sensitivity testing. Empiric therapy is treatment that is started in patients with suspected infections prior to a site or causative organism being identified. Prophylaxis is the prevention of infection using an antimicrobial drug when the infection is not currently present. Another category ‘unknown’ was allotted to a patient when no possible reason behind the antimicrobial prescription could be identified from the patient’s records. The treatment duration was characterised as the number of days starting from the date of antimicrobial initiation up to the date of the survey. Only culture and sensitivity testing on specimens like blood, urine or sputum was used to classify a treatment as definitive.
Ethical considerations
Mentioned study proposal was approved by institutional ethics committee Jamia Hamdard, New Delhi-62. Study was conducted in adherence and compliance of Good Clinical Practice (GCP) and declaration of Helsinki.
Statistical analysis
SPSS version 13.0 was planned to be used for analysis of the statistical data. Two indicators, frequency and percentage were decided to be used for the description of the section of patients receiving antimicrobials and other parameters that were defined and observed during this survey.
The number of patients admitted at the point of time of our survey was 217, out of which 101 (46.54%) patients were receiving antimicrobial agents (AMA) with 160 (73.73%) AMAs prescribed. A total of 50 patients (49.5%) were prescribed AMAs for infection as indication, whereas, 49 patients (48.51%) were prescribed AMAs prophylactically and 2 (1.98%) patients received AMAs for reasons not defined. The most common indications for antibiotic prescription were intra-abdominal infections (32.40%), respiratory tract infections (23.14%) and bone & joint infections (8.33%). The most commonly used antibiotics were beta-lactam antibiotics (60.62%) followed by metronidazole (10.62%).
In India, very few point prevalence surveys have been conducted and this is the first study of its kind to characterize antimicrobial use practices at our hospital and compare them with other PPS conducted all over the nation. Even though antimicrobial resistance is a worldwide issue, the problem lies at the level of the hospitals as they are the centres for development of antimicrobial resistance.15 Such surveys help in enhancing antimicrobial use and allow qualitative improvement of antibiotic prescriptions. As a result, this data can be conveyed to regional and national antimicrobial stewardship programs.
The point prevalence of antimicrobial utilisation at our hospital was recorded as 46.54% (patients receiving AMA) with 73.73% of antimicrobial prescriptions (Table 1). Reassuringly, this number was slightly lower as compared to a multi-centric PPS conducted across India that demonstrated 57.4% of patients were on antimicrobials.16 Similarly, our study showed numbers lower than other studies like a study conducted in 2014 and 2017 in Eastern India which suggested an antimicrobial prevalence rate of 62% in 2014 and 69.1% in 2017; a Chinese study which showed 56% prevalence of antibioic use.17-19 In contrast, an Indian study conducted in Central India showed only 31.95% patients receiving at least one antimicrobial which was comparable to other studies like a Canadian study which suggested the use of at least one antimicrobial in 31% for acute care patients and a European survey which reported29% antimicrobial use.20-22 At a global level, the data which was collected in a 2015 PPS across 53 countries suggested 34.4% prevalence for antimicrobial use.23 Unfortunately, there are regional variations which have not been highlighted in the global PPS and studies from 2017 show antimicrobial use of 48.2% in South and East Asian hospitals compared to 29.6% in European hospitals.23
Table (1):
Patient demographic and general characteristics.
Characteristics |
N (%) |
|---|---|
Number of patients admitted |
217 |
Number of patients receiving antimicrobials |
101 (46.54%) |
Median age of treated patients in years (Interquartile range) |
32.5 (55-22.5) |
Gender |
|
Male |
59 (58.41%) |
Female |
42 (41.58%) |
Number of antimicrobials prescribed |
160 (73.73%) |
Number of antimicrobials per prescription |
|
1 |
55 (54.45%) |
2 |
34 (33.66%) |
≥3 |
12 (11.88%) |
Route of antimicrobial administration |
|
Oral |
38 (23.75%) |
Parenteral |
122 (76.25%) |
Culture & Sensitivity (C/S) testing ordered |
21 (20.79%) |
Diagnosis Definitive Provisional |
67 (66.33%) 34 (33.66%) |
AMA Indication Infection Definitive Empiric Prophylaxis Medical Surgical Unknown |
4 (3.96%) 46 (45.54%) 14 (13.86%) 35 (34.65%) 2 (1.98%) |
Co-morbidity Diabetes Mellitus Hypertension Received ATT |
19 (18.81%) 15 (14.85%) 12 (11.88%) |
It is vital to understand the factors behind the prevalence of antimicrobial use in patients. Firstly, doctors receive unfair compensations and hospital business models are focused on the sale of drugs which consequently leads to an increase in prescriptions. Secondly, quality improvement projects are poorly managed.24,25 Data from a study conducted in China suggested that pharmacist interventions influencing clinicians in the ward can play a role in aiding judicious antimicrobial use.26
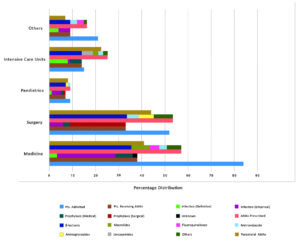
The data regarding the indications of antimicrobials in our study showed 49.5% prescribed for infections whereas 48.51% were prescribed for prophylaxis (Table 1). For infections, only 3.96% of prescriptions were definitive while 45.54% orders were empiric (Fig. 1). Furthermore, prophylaxis showed a trend of 13.86% orders for medical prophylaxis and 34.65% for surgical prophylaxis. Our data was comparable to some studies, like an Indian study which showed 51.4% use of antibiotics for community acquired infections and 30.2% for prophylaxis.18 Another Indian study showed 84.4% antimicrobials used for empirical treatment.16 Whereas, a European PPS data showed rates of only 15% for medical prophylaxis and 6.7% for surgical prophylaxis.27
One concern in our study was the use of surgical prophylaxis (including both pre- and post-surgical period in surgical patients) in 68.75% of patients for >24 hours. Our study revealed lower numbers in comparison to another Indian study whose data suggested that 77% of their patients received surgical prophylaxis for >24 hours whereas, only 14% of patients received single dose antibiotics as recommended as per protocols for surgical procedures.16 The ESAC PPS showed data similar to our study with 57.3% of patients receiving surgical prophylaxis for >24 hours and 25.2% of patients receiving single dose prophylaxis.27 It is daunting that many studies show that extended prophylaxis is not needed and unnecessarily results in the spread of antimicrobial resistance.28 Hence, this parameter acts as a quality check that needs to be monitored to tackle antimicrobial resistance.
The three most common indications for antibiotic prescription in our study were intra-abdominal infections (32.40%), respiratory tract infections (23.14%) and bone & joint infections (8.33%) (Table 2).In contrast to our data, an Indian study showed 19.9% of patients were being given antimicrobials for lower respiratory tract infections followed by skin and soft tissue infections as the most common indications.16 Also, a similar trend was observed in a teaching hospital in India.19 It is important to use these indications as targets for antimicrobial stewardship programs to influence patient care positively.
Table (2):
Antimicrobial utilisation pattern as a function of the system involved.
AMA Indication Organ System |
N=101 (%) |
|---|---|
Respiratory System |
25 (24.75%) |
Intra-abdominal/ GIT |
35 (34.65%) |
Bone and Joints |
9 (8.91%) |
Obstetrics/ Gynaecology |
3 (2.97) |
Renal System |
6 (5.94%) |
ENT |
5 (4.95%) |
Others (CNS, Ophthalmology, Dermatology, Blood, etc.) |
25 (24.75%) |
Similar to other studies, our results revealed that the intensive care units exhibited the highest proportion of antimicrobial treated patients (93.33%), who often were prescribed more than one AMA resulting in a huge antimicrobial consumption (166.6%) (Table 3, Fig. 1).16,20-22 One of the surprising observations of our study was that no patients were receiving any AMA in the Obstetrics and Gynaecology Department. This finding exhibits one of the limitations of the point prevalence survey.
Table (3):
Antimicrobial utilisation pattern across various medical specialities.
AMA Prescribed |
Medicine (%) |
Surgery (%) |
Paediatrics (%) |
Intensive Care (%) |
Others (%) |
Total (%) |
|---|---|---|---|---|---|---|
B-lactams |
35 (21.87%) |
32 (20%) |
7 (4.37%) |
14 (8.75%) |
9 (5.62%) |
97 (60.62%) |
Aminoglycosides |
2 (1.25%) |
6 (3.75%) |
1 (0.625%) |
1 (0.62%) |
—— |
10 (6.25%) |
Fluoroquinolones |
4 (2.5%) |
2 (1.25%) |
—— |
1 (0.625%) |
3 (1.87%) |
10 (6.25%) |
Macrolides |
8 (5%) |
——— |
——- |
2 (1.25%) |
——- |
10 (6.25%) |
Lincosamides (Clindamycin) |
——– |
1 (0.62%) |
——- |
5 (3.12%) |
——- |
6 (3.75%) |
Imidazole Derivatives (Metronidazole) |
4 (2.5%) |
7 (11.66%) |
1 (0.62%) |
2 (1.25%) |
3 (1.875%) |
17 (10.62%) |
Tetracyclines |
1 (0.62%) |
——— |
——- |
—— |
—— |
1 (0.62%) |
Others (Nitrofurantoin, Antiparasitic) |
3 (1.87%) |
5 (3.12%) |
——– |
—— |
1 (0.62%) |
9 (5.62%) |
Total (%) |
57 (35.62%) |
53 (33.12%) |
9 (5.62%) |
25 (15.62%) |
16 (10%) |
160 (100%) |
In the present study, the most commonly used antibiotics were beta-lactam antibiotics (60.62%) followed by metronidazole (10.62%) (Table 3). At the third position we had fluoroquinolones, aminoglycosides and macrolides each with 10% prescriptions. The most commonly prescribed antibiotics included the third-generation cephalosporin-ceftriaxone (28.12%), amoxicillin + clavulanic acid (19.37%) and metronidazole (10.62%). However, the positive finding in our study was that no Glycopeptides (vancomycin) were prescribed across any speciality. Similarly, observations from an Indian study showed the most common classes of antibiotics were third generation cephalosporins (44%), penicillins (14.4%) and metronidazole (12%).18 The ESAC-2011 and the global PPS 2015 which took 24.8% of the total antimicrobial prescriptions of the world into account suggested that penicillins along with b-lactamase inhibitors are the most commonly used antibiotics.23,27 Numerous studies conducted in countries from all over the world demonstrated very similar results to the global PPS 2015 for example, penicillins with b-lactamase inhibitors (24%) being the most commonly prescribed antibiotic followed by macrolides (15%) and fluoroquinolones (11%).20,21,29 A study conducted in USA in 2002-2003 showed levofloxacin, cefazolin, ceftriaxone, metronidazole and vancomycin as the most common antibiotics in prescriptions.30
It was observed that in our institute, ciprofloxacin (28.57%) and ceftriaxone (31.42%) were the most commonly used antibiotics for medical and surgical prophylaxis respectively. Our data differed from an Indian multicentric study which showed ceftriaxone (24%) as the most commonly used antibiotic for medical prophylaxis and cefuroxime (36%) for surgical prophylaxis.16 It is essential to note that the current protocols have suggested the use of third generation cephalosporins should only be used when first-line drugs are not effective.31 A recent investigation to study the sensitivity to antibiotics of agents causing community acquired pneumonia in India showed very high susceptibility to drugs like ampicillin and amoxicillin-clavulanic acid.32 The study emphasised that with excessive exposure of third generation cephalosporins in children there is a very high risk of colonisation early-on and the spread of Extended Spectrum Beta Lactamase to other members of the family. This information reconfirms that it is essential to stick to the national guidelines for prescribing antimicrobials.
It is also concerning that our data reveals a very high use of parenteral antibiotics (76.25%) (Table 1) which is relatively lesser than the Chinese study which demonstrated a use of 98% parenteral antibiotics.20 Surprisingly, the Indian teaching hospital study had only 30.55% antibiotics given parenterally.19 This data is crucial to understand as using oral therapy in cases when intravenous can be avoided would provide benefits such as reducing hospital admission durations and drug related side effects. It can also be used as a key parameter to monitor the antibiotic stewardship programs.19,34,35
Our study also analysed targeted antimicrobial treatment based on culture and sensitivity testing as one of the indicators of rational antimicrobial prescribing. Our observations showed that definitive treatment accounted for only 4% of antimicrobial prescriptions, even though culture and sensitivity testing was ordered in 20.79% of patients (documented) the remaining patients were continued on the empiric AMA therapy without reviewing the culture results (Table 1). This was a matter of serious concern which reflected underutilization of microbiological facilities and urgent need to implement a robust stewardship program. Other studies have revealed a targeted antimicrobial therapy administered in 11.12% patients.19 These indicators should be made into targets to achieve fewer cases of hospital acquired infections and antimicrobial resistance.
The comparative analysis of our study with other PPS surveys conducted across India highlighted some key aspects in terms of trends in antimicrobial utilisation (Table 4). Firstly, the number of patients receiving antimicrobials in our study (46.54%) was lesser than all other studies except the survey conducted by Najmi et al which suggested only 31.95% patients receiving antimicrobials. Secondly, the percentage of people receiving antimicrobials parenterally was higher in our study (76.25%) in comparison to
Najmi et al. (69%) and N. Shanmuga Vadivoo et al. (69%). Third key point analysed was that the targeted/definitive treatment was received by only 3.96% of our patients in comparison to 11.12% patients in the study conducted by Najmi et al. Fourth parameter that differed in our study was that the intra-abdominal infections (32.40%) were the most common indication as compared respiratory tract infections in studies conducted by Singh SK et al (26.9%) and Sumanth Gandara et al (32%).
Table (4):
Comparative analysis of various PPS Studies conducted across India.
Characteristics |
Current Study (2019) |
Singh SK et al16 |
Najmi et al20 |
Sumanth Gandra et al33 |
N. Shanmuga Vadivoo et al18 |
|---|---|---|---|---|---|
Number of patients admitted |
217 |
1750 |
241 |
681 |
502 |
Number of patients receiving antimicrobials |
101 (46.54%) |
1005 (57.42%) |
77 (31.95%) |
419 (61.52%) |
325 (70.11%) |
Median age of treated patients in years (interquartile range) |
32.5 (55-22.5) |
Not mentioned |
Not mentioned |
1-6 years |
39 |
Gender |
Not mentioned |
Not mentioned |
Not mentioned |
||
Male |
59 (58.41%) |
248 (59.18%) |
|||
Female |
42 (41.58%) |
171 (40.81%) |
|||
Number of antimicrobials prescribed |
160 |
1578 for 1005 patients |
100 |
Not mentioned |
Not mentioned |
Number of antimicrobials per prescription |
Not mentioned |
Not mentioned |
|||
1 |
55 (54.45%) |
291 (69.45%) |
55% |
||
2 |
34 (33.66%) |
85 (20.28%) |
45 % (more than one) |
||
>3 |
12 (11.88%) |
43(10.26%) |
|||
Route of antimicrobial administration |
Not mentioned |
||||
Oral |
38 (23.75%) |
31 (31%) |
31% |
||
Parenteral |
122 (76.25%) |
69 (69%) |
69% |
||
Targeted/ definitive AMA treatment |
4 (3.96%) |
8 (11.12%) |
Not mentioned |
Not mentioned |
|
AMA Indication |
Not mentioned |
||||
Infection |
50 (49.5%) |
727 (46.07%) |
273(65.1%) |
185 (56.6%) |
|
Prophylaxis |
49 (48.51%) |
725 (45.94%) |
79 (18.9%) |
117 (36%) |
|
Medical speciality with most AMA prescriptions |
Intensive Care 14 (93.33%) |
Not mentioned |
Not mentioned |
Only paediatrics study |
Gynaecology (88%) |
Most common indication organ system |
Not mentioned |
Not mentioned |
|||
1st |
Intra-abdominal infections 35 (32.40%) |
Respiratory tract infection (26.9%) |
Respiratory tract infections (32%) |
||
2nd |
Respiratory tract infection 25 (23.14%) |
Skin and soft tissue (7.6%) |
Sepsis (15.9%) |
||
3rd |
Bone and joint infections 9 (8.33%) |
CNS infection (6.6%) |
Surgery prophylaxis (11.9%) |
||
Most common AMA Class prescribed |
|||||
1st |
Beta lactam 97 (60.62%) |
Beta lactam (47.6%) |
Beta lactam (Number not mentioned) |
Beta lactams (53.2%) |
Beta lactams (58.4%) |
2nd |
Metronidazole 18 (10.62%) |
Aminoglycosides (10%) |
Fluroquinolones |
Aminoglycosides (10.4%) |
Metronidazole (12%) |
Most common AMA prescribed |
|||||
1st |
Ceftriaxone 45 (28.12%) |
Penicillin with beta-lactam inhibitor. (47.6%) |
Penicillin with a β-lactamase inhibitor |
Third-generation cephalosporins (38.9%), |
Third generation Cephalosporins ( 3GC)-44%, |
2nd |
Amoxicillin + clavulanic acid 31 (19.37%) |
Cefuroxime (36%). |
Ceftriaxone |
penicillin plus enzyme inhibitor combinations (14.3%) |
Penicillins (14.4%) |
3rd |
Metronidazole 18 (10.62%) |
Ceftriaxone (24%). |
Ciprofloxacin and levofloxacin. |
Aminoglycosides (10.4%) |
Metronidazole (12%) |
The variation in antimicrobial prescriptions between countries from all over the world exists due to some key factors. Studies conducted to understand these factors show that it can be due to different practices in terms of treating a disease, variations in antibiotic resistance across the globe and dissimilar adherence to guidelines.21,20 Another reason may be differences in the health care systems of countries which include the number of doctors per inhabitant or the time spent with each patient.36
There are some important limitations of a point prevalence survey. A PPS looks at a single point in time and the results can be influenced due to external factors such as day-to-day variations or seasonal use of certain antibiotics. Furthermore, this study could not analyse all quality indicators devised to assess antimicrobial prescribing due to limited resources. The strengths of this survey are the simplicity of the protocol, inclusion of every admitted patient receiving antimicrobials and expert involvement in data collection. In countries with low resources, PPS is possibly the only means to obtain a reliable antimicrobials utilisation status in hospitals.
The study suggests a high rate of antimicrobial use and highlights areas for intervention for rational antimicrobial use. We propose to sensitise the government on initiating a national antimicrobial stewardship program such as the Global Point Prevalence Survey and facilitate evidence-based antimicrobial practice.
ACKNOWLEDGMENTS
The authors would like to thank the team of interns and residents who helped in efficient and credible collection of patient data across all hospital wards.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made substantial direct and intellectual contribution to the work and approve it for publication.
FUNDING
None.
ETHICS STATEMENT
This study was approved by Jamia Hamdard Institutional Ethics Committee, New Delhi, India.
AVAILABILITY OF DATA
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
- Hulscher MEJL, van der Meer JWM, Grol RPTM. Antibiotic use: how to improve it? Int J Med Microbiol. 2010;300(6):351-356.
Crossref - Shorr AF. Review of studies of the impact on Gram-negative bacterial resistance on outcomes in the intensive care unit. Crit Care Med. 2009;37(4):1463-1469.
Crossref - Laxminarayan R, Matsoso P, Pant S, et al. Access to effective antimicrobials: a worldwide challenge. Lancet. 2016;387(10014):168-175.
Crossref - Bronzwaer SLAM, Cars O, Buchholz U, et al. A European study on the relationship between antimicrobial use and antimicrobial resistance. Emerg Infect Dis. 2002;8(3):278-282.
Crossref - Boeckel TPV, Gandra S, Ashok A, et al. Global antibiotic consumption 2000 to 2010: an analysis of national pharmaceutical sales data. Lancet Infect Dis. 2014;14(8):742-750.
Crossref - Hecker MT, Aron DC, Patel NP, Lehmann MK, Donskey CJ. Unnecessary use of antimicrobials in hospitalized patients: current patterns of misuse with an emphasis on the anti-anaerobic spectrum of activity. Arch Int Med. 2003;163(8):972-978.
Crossref - Cosgrove SE. The relationship between antimicrobial resistance and patient outcomes: mortality, length of hospital stay, and health care costs. Clin Infect Dis. 2006;42(Suppl. 2):S82-S89.
Crossref - Roberts RR, Hota B, Ahmad I, et al. Hospital and societal costs of antimicrobial-resistant infections in a Chicago teaching hospital: implications for antibiotic stewardship. Clin Infect Dis. 2009;49(8):1175-1184.
Crossref - Porta A, Hsia Y, Doerholt K, et al. Comparing neonatal and paediatric antibiotic prescribing between hospitals: A new algorithm to help international benchmarking. J Antimicrob Chemother. 2012;67:1278-1286.
Crossref - MacDougall C, Polk RE. Antimicrobial stewardship programs in health care systems. Clin Microbiol Rev. 2005;18(4):638-656.
Crossref - Barlam TF, Cosgrove SE, Abbo LM, et al. Implementing an antibiotic stewardship program: Guidelines by the Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America. Clin Infect Dis. 2016;62(10):e51-e77.
Crossref - Davey P, Marwick CA, Scott CL, et al. Interventions to improve antibiotic prescribing practices for hospital inpatients. Cochrane Database Syst Rev. 2017;2:CD003543.
Crossref - Schuts EC, Hulscher MEJL, Mouton JW, et al. Current evidence on hospital antimicrobial stewardship objectives: A systematic review and meta-analysis. Lancet Infect Dis 2016;16(7):847-856.
Crossref - World Health Organization. WHO methodology for point prevalence survey on antibiotic use in hospitals, version 1.1. http://www.who.int/iris/handle/10665/280063
- Kritsotakis EI, Dimitriadis I, Roumbelaki M, et al. Case-mix adjustment approach to benchmarking prevalence rates of nosocomial infection in hospitals in Cyprus and Greece. Infect Control Hosp Epi-demiol. 2008;29(8):685-692.
Crossref - Singh SK, Sengupta S, Antony R, et al., Variations in antibiotic use across India: multi-centre study through Global Point Prevalence survey. J Hosp Infect. 2019;103(3):280-283.
Crossref - Ravi N, Laha A, Hmar L, et al. Exploring the prescribing behaviours and the mind of antibiotic prescribers is critical for a successful antibiotic stewardship programme: results of a survey from Eastern India. Indian J Med Microbiol. 2017;35(2):299-301.
Crossref - Vadivoo NS, Usha B, Sudha K. Study to assess quality of antimicrobial use by point prevalence survey at a tertiary care centre. Indian J Microbiol Res. 2019;6(3):245-252.
Crossref - Xie D-s, Xiang L-l, Li R, Hu Q, Luo Q-q, Xiong W. A multicenter point-prevalence survey of antibiotic use in 13 Chinese hospitals. J Infect Public Health. 2015;8(1):55-61.
Crossref - Najmi A, Sadasivam B, Jhaj R, Atal S, Kumar S, Santenna C. A pilot point prevalence study of antimicrobial drugs in indoor patients of a teaching hospital in Central India. J Family Med Prim Care. 2019;8(7):2212-2217.
Crossref - Lee C, Walker SAN, Daneman N, et al. Point prevalence survey of antimicrobial utilization in a Canadian tertiary-care teaching hospital. J Epidemiol Glob Health. 2015;5(2):143-150.
Crossref - Zarb P, Amadeo B, Muller A, et al. Identification of targets for quality improvement in antimicrobial prescribing: the web-based ESAC Point Prevalence Survey 2009. J Antimicrob Chemother. 2011;66(2):443-449.
Crossref - Versporten A, Zarb P, Caniaux I, et al. Antimicrobial consumption and resistance in adult hospital in patients in 53 countries: results of an internet based global point prevalence survey. Lancet Global Health. 2018;6(6):e619-e629.
- Biomerieux. International experts joint forces against superbugs at the 4th World Forum on Health Care associated infections and antimicrobial resistance. https://www.biomerieux-nordic.com/international-experts-join-forces-against-superbugs-4th-world-forum-healthcare-associated-infections. Accessed November 10, 2021.
- Reynolds L, McKee M. Factors influencing antibiotic pre-scribing in China: an exploratory analysis. Health Policy. 2009;90(1):32-36.
Crossref - Shen J, Sun Q, Zhou X, et al. Pharmacist interventions on antibiotic use in inpatients with respira-tory tract infections in a Chinese hospital. Int J Clin Pharm. 2011;33(6):929-933.
Crossref - Zarb P, Goossens H. European Surveillance of Antimicrobial Consumption (ESAC): value of a point-prevalence survey of antimicrobial use across Europe. Drugs. 2011;71(6):745-755.
Crossref - Hagel S, Scheuerlein H. Perioperative antibiotic prophylaxis and antimicrobial therapy of intra-abdominal infections. Visceral Med. 2014;30(5):310-316.
Crossref - Ansari F, Erntell M, Goossens H, Davey P. The European Surveillance of Antimicrobial Consumption (ESAC) point-prevalence survey of antibacterial use in 20 European hospitals in 2006. Clin Infect Dis. 2009;49(10):1496-1504.
Crossref - Jacob JT, Gaynes RP. Emerging trends in antibiotic use in US hospitals: quality, quantification and stewardship.Expert Rev Anti-Infect Ther. 2010;8(8):893-902.
Crossref - National Treatment Guidelines for Antimicrobial Use in Infectious Diseases. National Centre for Disease Control. https://ncdc.gov.in
- Torumkuney D, Chaiwarith R, Reechaipichitkul W, et al. Results from the survey of antibiotic resistance (SOAR) 2012-14 in Thailand, India, South Korea and Singapore. J Antimicrob Chemother. 2016;71(suppl 1) i3-i19.
Crossref - Gandra S, Alvarez-Uria G, Murki S, et al. Point prevalence surveys of antimicrobial use among eight neonatal intensive care units in India: 2016. Int J Infect Dis. 2018;71:20-24
Crossref - Vogel F. Intravenous/oral sequential therapy in patients hospitalized with community-acquired pneumonia: which patients, when and what agents? Drugs. 2002;62(2):309-317.
Crossref - De luca M, Dona D, Montagnani C, et al. Antibiotic prescriptions and prophylaxis in Italian children. Is it time to change? Data from the ARPEC project. PLoS One. 2016;11(5):e0154662.
Crossref - Llor C, Bjerrum L. Background for different use of antibiotics in different countries. Clin Infect Dis. 2005;40(2):333.
Crossref
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.