ISSN: 0973-7510
E-ISSN: 2581-690X
The aim of this study was to choose the optimal PCR technique with specific designing primer design of cystic fluid of Echinococcus granulosus hydatid cyst in cattle have slaughtered in kerbala abattoirs / kerbala governorate, the techniques have allowed to reduce the time consumption ,Nucleotide sequences of Echinococcus granulosus in Iraq cattle was obtained from NCBI websites with other researchers concerned in this topic, numerous visceral organs Livers , lungs and liver with lung suspected have hydatid cysts were isolated from 35 cowºs slaughter, In PCR the highest number of positive results have 22(62.8%) were detected as Echinococcus granulosus hydatid cyst, on the other organs, the parasite isolation ratio was calculated from internal organs the percentage of hydatid cysts were significant increases as 12 (63.6%), 6 (27.2%)and 2(18.1%) for Liver, lung and liver with lung, respectively.
Echinococcus granulosus, Hydatid cyst in cow and PCR
Echino coccosis is a zoonotic disease of man and demoestic animals with world wide importance1 caused by larved stage of Echinocossus granulosus , the common site for infections are liver , lung and other organs2.
The life cycle of this parasite is indirect , it has dogs and other canids as a final host and Omnivorus with harbivorus as intermediate host3.
The disease transmission , when food or water that contains the eggs of parasite is eaten or close contact with infected.
Animals4 , in the human and intermediate host via infected handling soil or dirt animal hair with eggs5, but there are not biological or mechanical vectors for larval stage except birds and arthropods can act a mechanical vectors for eggs6. The disease occure without symptoms and signs but these appear deponton the cyst’s size and location7, the disease has in both human and animals health considerable impact with economic loss in animal production8.
There are ten genotypes of E. granulosns (G1-G10), (G1) in sheep , (G2 – G3) in buffalo , (G4) in aquine , (G5) in cattle , (G6) in camel , (G7) in pigs and (G8 – G10) are cervid strains9,10 , the strains have a biological and molecular aspects11,12 with different in regions distribution and the pathological identification is difficult in the case of aberrant forms13 also different in host specificity , pathogenicity life cycle pattern , transmission and rates of development finally response to the chemotherapeutic drugs14.
Polymerase chain reaction (PCR) based other methods has been vastly used for genotyping of E. granulous15,16 , now the clinical sample are taken biopsy to PCR for amplified fragments of mitochondrial and nucleur DNA are subsequently sequenced17
The aim of this study
The aim of this study were to choose the optimal PCR technique with specific designing primer for cystic fluid to detect Echinococcus granulosus hydatid cyst in internal visceral organs of cow abattoirs-kerbala governorate.
An internal organ of cow which includes Liver, lung and liver with lung were sent to parasitological laboratory in the kerbala Veterinary medicine for parasite testing. For this, all cysts with organs were cut into one centimeter pieces and left in warm water for 20 minutes. Then, removed by compression, and the sediments were obtained9. DNA extraction were used for all cystic fluid and pieces by Tissue/Blood DNA Mini Kit (Geneaid, Korea), by following of manufacturer’s instructions, The primer pair was used to amplify the partial mitochondrial NADH dehydrogenase subunit 1 (ND1) gene of Echinococcus granulosus , The sequence used in this study was from the National Center for Biotechnology ( http://www.18.nlm.nih.gov/) websites have accession numbers as (AY386216.1, KY766890.1, AF297617.1 and KX039956.1) the primer was designed from Primer3 plus ( http://primer3.wi.19.edu/ ) website, The sequence of the primers are designed for the following. PCR amplification was completed using conventional PCR conditions in a final volume of twenty five microne : five microne of DNAtemplate (20 ng/µl), 12¼lMilliQ water and 1.5µl for both primer forward and reverse primer (10 µM),all mixer were collected on each PCR tubes containing 5µlof AccuPower® ProFiTaq PCR PreMix (Bioneer, korea). The PCR reactivity was hold out in a Techne TC-512 thermocycler with the following cycling conditions: initial denaturation(95 °C, 15 min), succeed by 35 cycles of denaturation (95 °C, for thirty seconds), annealing (56 °C, 40 s) and sixty second for extension have (72 °C), and a last extension step (72 °C, 10 min). All the amplification of single fragment of the expected size was establish by gel electrophoresison a 1.5 % agarose gel (TBE, 1 %) stained with ethidium bromide. followed by 30 cycles using 1 min denaturation at 94°C, I min annealing at 55°C and 2 min extension at 72°C, PCR reactions were assayed on 1.5% agarose gels in the presence of 5 g/ml ethidium bromide.(B).
Echinocossosis in Iraqi is major endom disease20 and enzootic21 , a total number of 35 slaughtered cattle were inspected in this study. The overall prevalence of hydatidosis in slaughtered livestock was 22 (62.8%). In total, 12 (63.6%), 6 (27.2%) and 2(18.1%) Liver, lung and liver with lung, were condemned in slaughterhouses due to E. granulosus hydatid cysts in cattle, respectively. Additionally, PCR technique were used to detect these parasites in hydatid cysts in both organs. Figure 1.
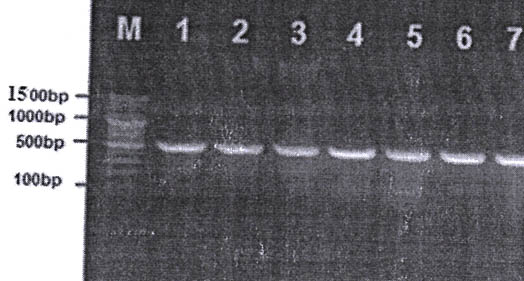
Fig .1.
1 to 74.4 % in dogs and sheep, respectively, 3.5 to 38.3 % in cattle 1.7 to 20 Vorin. L goats, (Estaim—and Hosseint1998; Ahmacti 2005; AnsarirLari_2 05; gsakiaL20_091 Shahb-azi—eral72171:4).
The application of polymerase chain reaction (PCR) is based on studies involved large number of Echinococcus granulosus were limited due to cumbersome methods access to the genomes of these parasities (@) lytic Enzymes and protein Extraction to purified DNA parasities in particular Echinococcus granulosus involve weakening of the membrance of hydatied cyst by the prsesence of followed by enzymatic treatment and ionic detergents.
As detergents such as (sodium dodecyl sulfate )SDS have a deleterious effect on the protein denatyuration of membrance of hydtid cysts (@) it was necessary to modify a designing primer such procedures to use in PCR, the selection of appropriate primers for maximal specificity and efficiency. Primer specificity was affected by a number f factors, including sequence , primer location and the PCR system used. General primar – designing rules for polymerase chain reaction were also applicable in PCR to avoid primaer – dimer mispriming formation.
Table (1):
Showed the primer instruments of this study
Name of Primers |
Sequences |
Base pair |
Melting Temperature |
G-C contens |
|---|---|---|---|---|
Cox1F |
5’-CGAACACGTGGTAATGTCGC |
20 bp |
59.83°C |
50.0 % |
Cox1R |
5’-TCCGTGTTGTGGTTTTTGGC-3’ |
20 bp |
59.91 °C |
50.0 % |
There are many studies reviewed the importance in of echinoccosis but the program control against hydiat cyst cannot be successful unless the farmers education , charged people about this disease and controlled slaughters of livestock must be effective.
- A.R.Satoskar . G.L Simon ,P.J Hotoz and M.Tusji “ medical parasitology lardes Bioscience “ , Taxas , 2009.
- M.Jawetz and adelberg’s “medical microbiology “. 24th Edn. The McGraw – Hill companies , Inc, New York , 2007.
- Latif ,A.A, Yanveer , A , maqbool,A. Siddigmi ,N, Kyaw – Tanner ,M and Traub, R.J – Morphological and melecualr – characterization of E. grunulosus in livestock & human in Punjab, Piskistan, Vet. Parasitol, 2010; 170: 44 – 49.
- World health Organization “Echinococcosis fact sheet No. 377 “ March , 2014)
- Eekert , J. and Deplazes ,p. “ Biological , epidemiological and clinical aspects of echinocossosis , azoonosis of increasing concern “ clin.microbile rev. 2004; 17(1) : 107 – 135.
- “Echinococcus granulosus “ material safety Data sheets (MSDs) public healthy agency of Canada, 2001.
- Connolly stephaine . “ Echinococcosis “ , 2006.
- Torgerson , P.R “ Economic effects of echinocossosis “. Acta Trop. 2003; 85: 113 – 118.
- Nakao , m, Lavikainen , A , Yanagida , T and Akira , I “ phylogenetic systematics of the genus Echincoccus (Gestoda Taeniidae ) Int. J paraditol. 2013; 43“ 117 – 1029.
- Romig , T , Ebi , D. and Wassermannk, M “ Toxonomy and molecular epidemiology of E. granulosus , sensu lato “ veto parasitol. 2015; 213: 76 – 84.
- E. Sanchez , G, Caceres , C. Naguiret D. , Garcia , G. patino , H. silvia , A.C, voloteno and O. Fernandez . “ Melecular characterization of E. grannlosis from peru by Eqmenencing of the mitochondriol Cytochromec oxidase subunit 1 gene “ memories do instituto Oswalado cruz, 2010; 105(6) : pp. 806 – 801.
- McManns , Dp “ Current status of the genetics and molecular taxonomy ofechinococcus spp. Parasitology., 2013; 140: 1617 – 1623.
- Nakao ,m , Tetsuya , Y ,A , Munehiro , O,R , Jenny , k ,A , Agathe , N .A yashinto , S.A and Akira , I .A “ . state of – the Art echinococcus and taenia : phylogenetic tape morms and its application to melecualr diagnosis infection , Genetics and evolaution, 2010; 10(4): Pp: 444 – 452.
- Sharafi , S.M , Rstami – Nejad , M. Mozaeni , m. Yousefi , M , saneie , B., Hosseini – Safe , A . “ echinocosus granulosus genotypes in Iran , Gastroenterol hepatol the etiological diagnosis of suspected hydatid cyst” J.Parasit Dis, 2004; 28(2) : 8 – 64.
- McManus , Dp. Zhang , W , Li , J and Bartley , P.B “ Echinococcosos Lancet, 2003; 362: 1295 – 1304.
- Kagendo , D. Magambo , J., Agola E.L et.al “ A survey for Echinococcus spp. Of carnivores in six wild life conservation area in Kenya parasitol Int. 2014; 63(4) : 604 – 11.
- Moghaddas , E., Borji , H., Naghibi , A , shayan , P and Razmi , G.R “ Moolecualr gentyping of Echinoococcus gramulousus from dromedrairs in eastern Iran J. Helminthol: 2013; 1-5.
- Untergasser , A. cutuulache , I Koressaar , T , J ., faircolthes , Bc remn , M and Rozan m Sc primer , new capabilities and interfeaces Nucleic acids Res 2012, 140(15) : e 115.
- Koressaar , T abd Romm , M Enhancements and modification of primer design program primer 3 , bioinformatics 23(10) : 1290 – 91.
- Al-Mounasi , Ms. “ Epidemiological and Serommunological studies on Hudatid disease in Basrah ( southern Iraq ) PhD thesis biology department education college university of Basrah Iraq. 1998.
- Babero , B.B and Al-Dabbagh , M.A the zoonosis of animal parasite in Iraqi J.Fac med Baghdad 1963; 5: 79 – 84.
- N,Ahmadi and A m Dalimi “ moleculer characterization of E. granulosus isolate from sheep , human and female in Iran , B.v . 2006; 6: 85 – 90 .
- Rokni , M. “ Echinococcosis / Hydatidosis in Iran parasitol 4 : 1-16 . 2009. Eslami , A and Hosseini , S.H .” Echincocco granulosus infection of farm dogs of Iran. Parasitology research, 1998; 84: 205-207.
- Hakialilo , E , Fasihi , Harandi , M ., sharbatkhori , M . Mirhendi , H and Rostami , S , “ Genetic characterization of Egranulosus in camels , cattle and sheep from the south east of Iran indicates the G3genotype. T Helminthe, 2012; 86 – 263 – 270.
- Fasihi Haranmdi , M, Budke , Cm. and Rostami , S. “ The monetary burden of aystic echincoccosis in Iran plosnegl trop. Dis, 2012; 6: 38.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


