ISSN: 0973-7510
E-ISSN: 2581-690X
The aim of this present work is to isolate and identify bacterial strains from sweet lime juice sample for bacteriocin production. The cell-free supernatant of these isolates was screened for antimicrobial activity against four test organisms i.e. Bacillus subtilis, Staphylococcus aureus, Escherichia coli and Pseudomonas aeruginosa. Out of 10, three isolates (30%) showed positive inhibition against test organism. The influence of different external parameters on bacteriocin activity as well as on its production was examined. It was found that strain BP03 out of three strains exhibited antimicrobial activity against test pathogens at high temperature (121!), high salt concentration (10% w/v) and over the wide range of pH (3, 6, 7, 9). Further, the morphological and physiological identification was done through Gram staining and biochemical test.
Bacteriocin, Antimicrobial, Juice isolates, biochemical identification, Enterobacter.
Food safety has become an important matter of concern in developing as well as developed nations1. The conventional practices of food preservation include- heat treatment, drying, cold room storage, pasteurization, canning and extensive use of chemical preservatives for preserving the indigenous nutrients, the flavour of food while simultaneously reducing spoilage and extending the shelf life of food. Although they have played the significant role in food preservations, the consistent use of chemical preservatives led to many allergic and detrimental effects on consumers. The intensive physical and chemical treatment has degraded the nutrients and quality of food2. Also, the emergence of antibiotic-resistant bacteria has posed the serious threat to public health3. These concerns have led the researchers around the globe to identify alternative naturally produced antimicrobial compounds for food preservation. There are a variety of antimicrobial products produced by the microorganism such as organic acids, lactic acid, acetic acid, diacetyl, hydrogen peroxide, ethanol which have the potential for food fermentation and biopreservation in the food industry4. Apart from these metabolic end products, some strains also secrete antimicrobial peptides termed as bacteriocins described as the proteinaceous compounds that kill closely related bacteria5. The researchers are increasingly recognizing the potential of bacteriocin-producing bacterial strains as an alternative to chemical preservatives in preventing the growth of foodborne and food spoilage microorganisms, thereby ensuring food safety without altering the gut microbiota as well as improving the shelf life of food1,2.
Bacteriocins are ribosomally synthesized, extracellularly released low molecular mass peptides or proteins (usually 30-60 amino acids) which have a bactericidal or bacteriostatic effect on other bacteria6,7, either in the same species (narrow spectrum) or across genera (broad spectra) 8,9. The bacteriocins have been classified into three classes as- Class I known as the lantibiotics; class II comprised the small heat-stable non-lantibiotics and class III include large heat labile bacteriocins. There is also a fourth class of bacteriocins composed of the mixture of proteins, lipids and carbohydrates10. These peptides are cationic and hydrophobic in nature exhibiting antimicrobial activity by deploying mainly two mechanisms- inhibition of cell wall synthesis and pore formation in the membrane which imbalance the potential of the cell, ultimately induce cell death.
Since the discovery of bacteriocin by Gratia in 1925 as colicine which has the potential to kill Escherichia coli, numerous bacterial species have been identified with the potential to produce proteinaceous compound i.e. bacteriocin against the broad range of foodborne pathogens including Bacillus, Clostridium, Staphylococcus and Listeria. The bacteriocin Nisin produced by Lactococcus lactis was the first bacteriocin approved by FDA as well as enlisted in the European food additive list for its use in the preservation of pasteurized processed cheese11.
The objective of this study was to isolate and screen the bacteriocin-producing bacterial strains from Citrus limetta commonly known as sweet lime, sweet lemon, and sweet limetta. Sweet lime juice is the most commonly available vitamin C rich citrus juice. It has been reported that the presence of limonoids in Citrus species was responsible for their antibacterial activity against many bacterial strains12. The antimicrobial spectra of bacteriocin-producing strains were done against a wide range of Gram-positive and Gram-negative bacteria in vitro by agar well diffusion assay. This study also evaluated their potential under adverse conditions such as high temperature, salinity and pH. Based on antimicrobial spectra and functional properties further morphological and biochemical characterization of all strains was carried out.
Sample Collection
The juice of Citrus limetta (sweet lime) was collected from fruit juice shop in the local market, in the month of July and allowed it to ferment overnight. It was used for isolation of bacteriocin-producing bacteria.
Isolation and Screening of Bacteriocin Producing Microbes
The fermented juice sample was serially diluted (10-1 to 10-7) in 0.85% sterile physiological saline solution. Then 100 µl of appropriate dilutions are plated on MRS (de Mann Rogosa Sharpe) agar (containing peptone 10 g/l, yeast extract 5 g/l, beef extract 10 g/l, dextrose 20 g/l, ammonium citrate 2g/l, sodium acetate 5g/l, MnSO4 0.05 g/l, MgSO4 0.01g/l, dipotassium phosphate 2g/l, tween-80 1ml/l, pH 6.5) and incubated for 2-3 days at 37 degrees Celsius. The colonies were randomly selected and streaked on MRS agar to check the purity of the isolates. The purified strains were inoculated in MRS broth with 25% glycerol as a cryoprotectant and stored at -70 degrees Celsius.
Microbial Indicator Strains
The Gram positive and Gram negative bacterial indicator strains – Bacillus subtilis, Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa were used to evaluate the bacteriocin activity of isolates. They were revived and maintained in laboratory by repeated sub-culturing in growth media.
Assay for Bacteriocin Activity-Agar Well Diffusion Method
The frozen stock of isolates was first propagated thrice in MRS broth as well as in MRS agar and then used for bacteriocin assay. The aliquots of 1mL of culture broth were prepared and centrifuged at 5000xg for 10 minutes. The cells were separated out and cell-free supernatant was collected in another sterile microcentrifuge tube. This cell-free supernatant was used as crude bacteriocin. The agar diffusion method was used to screen for bacteriocin producing microbes among the five isolates against Bacillus subtilis, Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa. A lawn of an indicator strain was made by spreading the cell suspension over the surface of the LB Agar plates with a spreader. The plates were allowed to dry and a sterile cork borer of diameter 8mm was used to cut uniform wells in the agar plates. Each well was filled with 50ul of cell-free supernatant from each isolate. Plates were incubated at 37o C for 24 hours. The bacteriocin activity of isolates was examined by measuring the diameter of the inhibition zone around the wells. Screening for bacteriocins producing microbes was repeated thrice for each isolate.
Effect of pH, Temperature and Salinity on Activity of Bacteriocin
High temperature Tolerance
The thermostability of bacteriocin was examined at different temperatures. The cell-free supernatant was isolated from all three bacteriocin producing isolates and incubated for 15 minutes at different temperature 60, 100 and also autoclaved (121oC). The respective samples were first cooled to room temperature (37°C ) and then assayed for antimicrobial activity against test organisms using method described above.
Effect of pH
To test stability at different pH, the aliquots of cell free supernatant of all three isolates was adjusted to pH values 3, 6, 7, 9 using 1M NaOH or 1M HCl and final pH of treated crude bacteriocin was confirmed with the help of pH meter prior to test against indicator microorganisms. Then 50 µl from each treated crude extract were placed in the wells of test organism seeded plates.
Effect of NaCl
The effect of salt on production of bacteriocin was determined by inoculating the isolates in the MRS broth supplemented with 5 and 10 %( w/v) NaCl. Then bacteriocin activity was assayed by agar well diffusion method described above.
Biochemical Characterization of Bacteriocin Producing Microbes
Bacteriocin producing strains were Gram stained in order to identify the cellular morphology and staining pattern. Taxonomical characterization of strains is done through various biochemical test such as indole test (using Kovac’s reagent), methyl red-Voges-Proskauer test, citrate test, oxidase (using p-phenylenediamine dihydrochloride), catalase (with 3% hydrogen peroxide), urease test, Gelatin hydrolysis, starch hydrolysis and carbohydrate fermentation test.
Isolation and screening for bacteriocin producing trait
In this present study, total 10 strains of bacteria were isolated from the collected sweet lime juice sample by spread plate technique on MRS agar and examined for potential of bacteriocin production against 4 indicator microorganisms. The antimicrobial activity of cell free supernatant (referred as crude bacteriocin) of isolates was tested by agar diffusion method against Gram- positive bacteria, viz., Bacillus subtilis, Staphylococcus aureus as well as Gram-negative bacteria Escherichia coli, Pseudomonas aeruginosa. From total 10, 3 isolates were recorded as positive, showing inhibition against pathogenic strains.
Table (1):
Showing the zone of inhibition of all isolates against all tested strains
| Strains | Zone of inhibition (in mm) | |||
|---|---|---|---|---|
| Escherichia coli | Staphylococcus aureus | Bacillus subtilis | Pseudomonas aeruginosa | |
| BP01 | 12 | 11 | 9 | 7 |
| BP02 | 10 | 9 | 8 | 8 |
| BP03 | 11 | 9 | 8 | 6 |
Effects of pH, Temperature and Salinity on the bacteriocin activity
The bacteriocin activity was tested with different temperatures (68, 100, and 121) to evaluate thermal stability of bacteriocin. Maximum activity of bacteriocin produced by isolate BP01 and BP02 were found to be at 121oC against Escherichia coli while that of isolate BP03 was found to be at 68oC against Bacillus subtilis (Table 2). Regarding pH, the maximum inhibhitory activity of all the isolates were found to be at pH 3 (Figure 1). The effect of salt concentration on the production of three bacteriocin were studied. It was found that activity of bacteriocin produced by isolate BP01 and BP03 has been increased while that of strain BP02 has decreased with increasing NaCl concentration (5% to 10%) in the growth media (Table 3).
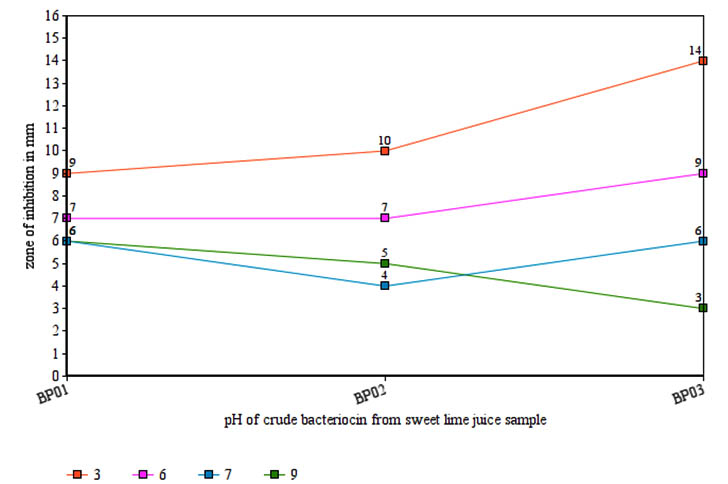 Fig. 1. Effect of pH on antimicrobial activity of bacteriocin
Fig. 1. Effect of pH on antimicrobial activity of bacteriocinTable (2):
Thermal stability of bacteriocin produced by the selected isolates
| Sample | Temperature | Incubation Time | Inhibition Zone Diameter (in mm) | |||
|---|---|---|---|---|---|---|
| Escherichia coli | Staphylococcus aureus | Bacillus subtilis | Pseudomonas aeruginosa | |||
| BP01 | 68°C | 15 min | 7 | 7 | 8 | 6 |
| 100°C | 15 min | 6 | 9 | 7 | 6 | |
| 121°C | 15 min | 10 | 9 | 11 | 8 | |
| Control (without heat treatment) | 12 | 11 | 10 | 8 | ||
| BP02 | 68°C | 15 min | 7 | – | 8 | 6 |
| 100°C | 15 min | – | 9 | 5 | 7 | |
| 121°C | 15 min | 9 | 8 | 6 | 8 | |
| Control (without heat treatment) | 10 | 7 | 8 | 7 | ||
| BP03 | 68°C | 15 min | 8 | 6 | 12 | 7 |
| 100°C | 15 min | 7 | 8 | 4 | 6 | |
| 121°C | 15 min | 12 | 13 | 12 | 10 | |
| Control (without heat treatment) | 9 | 10 | 8 | 10 | ||
Table (3):
Salt tolerance of the selected bacteriocin producing isolates
| NaCl concentration | Samples | Inhibition zone diameter in mm | |||
|---|---|---|---|---|---|
| Escherichia coli | Staphylococcus aureus | Bacillus subtilis | Pseudomonas aeruginosa | ||
| 5% | BP01 | 9 | 10 | 8 | 13 |
| BP02 | 8 | 12 | 13 | 11 | |
| BP03 | 11 | 13 | 9 | 8 | |
| 10% | BP01 | 17 | 15 | 12 | 16 |
| BP02 | 15 | 9 | 5 | 6 | |
| BP03 | 14 | 17 | 13 | 7 | |
Physiological and Biochemical Characterization of Bacteriocin Producing Isolates
These isolates were then studied for Gram staining profile. All three strains showed pink colour under microscope therefore, tested Gram negative. Two strains exhibited coccoid morphology while one strain was rod shaped bacilli. Biochemical properties of these strains are shown in (Table 4).

Fig. 2. All strains were showed negative indole test

Fig. 3. All strains were unable utilize citrate as carbon source

Fig. 4. All strains were showed urease negative
Table (4):
Biochemical Characterization of Bacterial Isolates
| Name of the Test | Bacterial isolates | ||
|---|---|---|---|
| BP01 | BP02 | BP03 | |
| Gram staining | Negative | Negative | Negative |
| Morphology | Cocci | Cocci | Bacilli |
| Indole | – | – | – |
| Methyl Red (MR) | + | – | + |
| Voges Proskauer (VP) | – | + | – |
| Citrate | – | – | – |
| Starch | – | – | – |
| Gelatin | – | – | – |
| Sucrose | + | + | + |
| Lactose | + | + | + |
| Dextrose | + | + | + |
| Urease | – | – | – |
| Catalase | + | + | + |
| Oxidase | – | – | – |
Growth (+), no growth (-)

Fig. 5. MR-VP test of all three strains
Fig. 6. Carbohydrate fermentation test with no production of gas in all three strains

Fig. 7. Catalase positive and oxidase negative test of all strains
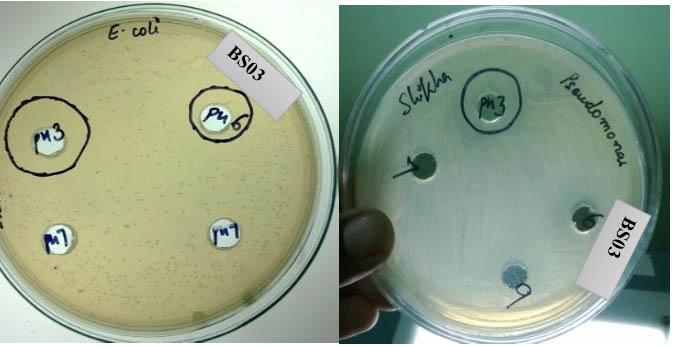
Fig. 8. Bacteriocin activity of isolate BS03 at pH 3 against Gram negative bacteria
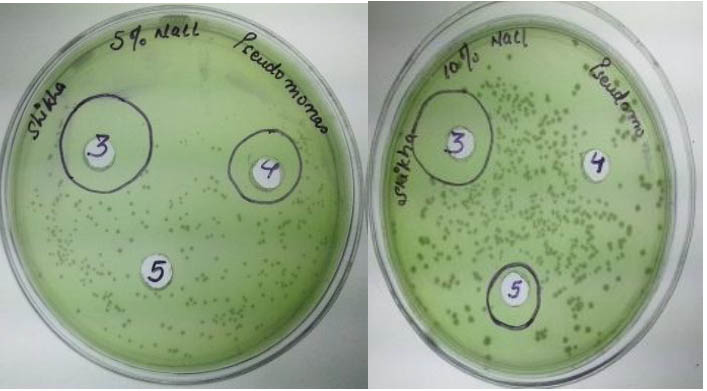
Fig. 9. Bacteriocin activity of isolate BS03 at 10% (w/v) NaCl against Pseudomonas aeruginosa
In general, bacteriocins are extracellularly secreted proteinaceous compounds produced by bacterial strains with potent antimicrobial activity against bacteria either in the same species (narrow spectrum) or across genera (broad spectra). Furthermore, they are active against food-borne pathogens including Listeria monocytogenes, Staphylococcus aureus, and Clostridium botulinum, Clostridium perfringens, Bacillus subtilis, therefore they are used as natural or so-called bio preservative in food industries to overcome the problems associated with chemical preservatives. As reported by Cotter et al., 2005 “bacteriocins can be used to confer a rudimentary form of innate immunity to food stuffs”.
The isolation and screening of microorganisms from natural sources has always proved to be effective for obtaining antimicrobial compounds with medical and industrial applications. The aim of this study was to isolate and characterize bacterial strains from sweet lime juice for bacteriocin production. The bacteriocin production from such bioactive metabolites rich sources will be beneficial for food industries to prevent the growth of food borne pathogens and enhancing the shelf life of food.
The agar well diffusion assay was employed in vitro to examined the inhibitory role of bacteriocin produced by these isolates against pathogenic microbes. The cell free supernatant (crude bacteriocin) of three out of 10 randomly picked colonies exhibited antimicrobial potential against Gram negative and Gram-positive bacteria such as Bacillus subtilis, Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa. These organisms have been reported as causative agent for food borne diseases capable for inducing food poisoning.
They were screened as bacteriocin producers by measuring clear zone of inhibition. The results so obtained by this preliminary screening showed that these three isolates exhibit maximum inhibition against test pathogens as compared to other strains.
In addition to this, antibacterial activity of crude bacteriocin from these three isolates was subjected to different external environmental factors like pH, temperature and salinity.
The bacteriocins from all the isolates were stable in acidic pH 3 with strain BP03 showed maximum inhibitory activity against all four test organisms. The cell free supernatant of isolate BP03 exhibited potent inhibition against Staphylococcus aureus at autoclaved temperature 121 while other strain BP01 and BP02 showed moderate inhibition at 121 against Escherichia coli and Bacillus subtilis. This also suggests that bacteriocin produced by strain BP03 could be categorized as class II bacteriocin which are heat stable inhibitory peptides. This thermostability as well as potential to be in active state in acidic pH has suggested the applications of strain BP03 in food and biopharmaceuticals industries.
Further more it was also revealed that on increasing the concentration of NaCl (from 5% to 10%) in the growth medium, the inhibitory activity of bacteriocin so produced from isolates BP01 and BP03 was also increased. This showed that these strains were able to tolerate high (10%) salt concentration.
After determining the antimicrobial activity of strains from preliminary bacteriocin assay and depicting their functional characteristic by subjecting to different stress condition further, the morphological and biochemical tests were carried out. All the three strains were gram negative with two strains BP01 and BP02 in coccoid morphology while third strain BP03 was rod shaped bacilli. The strains were catalase positive producing bubbles with 3% hydrogen peroxide while unable to oxidise tetra-methyl-p-phenylenediamine dihydrochloride reagent (oxidase test) indicating cytochrome c oxidase deficient strains. The strains BP01 and BP03 gave methyl test positive and negative Vogues- Proskauer test while opposite is true for strain BP02. These strains were unable to breakdown high molecular weight biomolecules such as Gelatin, starch, citrate, tryptophan (indole test) and urea. However, these strains were capable to assimilate carbohydrates sources such as Sucrose, Lactose, Dextrose while production of gas was not detected by Durham tubes. This test is mainly useful to identify Gram-negative enteric bacteria. On the basis of outcomes of biochemical tests, it was found that all the strains were classified as Enterobacter species.
In recent years, bacteriocin producing isolates from the natural source has found a significant role in food and pharmaceutical industry as an alternative to chemical preservatives in food to prevent the growth of foodborne pathogens. Moreover, they have therapeutic applications which substituted the antibiotic-based therapy. To conclude, the antimicrobial activity of cell-free extract (crude bacteriocin) isolated from sweet lime juice was studied by agar well diffusion method against Gram-negative and Gram-positive pathogenic test organisms responsible for food spoilage and poisoning. The bacteriocin of these isolates showed antibacterial stability at various external factors such as elevated temperature, acidic pH and increased salinity. The strains were further described as Gram-negative bacteria and characterized as Enterobacter species with the help of various biochemical tests.
ACKNOWLEDGMENTS
The authors thank DST-SERB for providing financial support with research grant ECR/2017/000080 to carry out this research work. The authors are also thankful to the Amity University Uttar Pradesh for providing infrastructural support. There is no conflict of interest.
- Galvez, A., Abriouel, H., Lopez, R.L., Omar, N.B. Bacteriocin-based strategies for food biopreservation. International Journal of Food Microbiology., 2007; 120(1–2): 51-70.
- Johnson, E.M., Jung, Y.G., Jin, Y.Y., Jayabalan, R., Yang, S.H., Suh, J.W. Bacteriocins as food preservatives: Challenges and emerging horizons. Critical Reviews in Food Science and Nutrition., 2017.
- Grosu-Tudor, S.S., Stancu, M.M., Pelinescu, D., Zamfir, M. Characterization of some bacteriocins produced by lactic acid bacteria isolated from fermented foods. World Journal of Microbiology and Biotechnology., 2014; 30(9): 2459-2469.
- Lindgren, S. E., Dobrogosz, W. J. Antagonistic activities of lactic acid bacteria in food and feed fermentations. FEMS Microbiol. Rev., 1990; 7: 149-163.
- De, V. L., Leroy, F. Bacteriocins from lactic acid bacteria: production, purification, and food applications. J Mol Microbiol Biotechnol., 2007; 13: 194–199.
- Klaenhammer, T.R. Genetics of bacteriocins produced by lactic acid bacteria. FEMS Microbiology Reviews., 1993; 12: 39–85.
- Tagg, J. R., Dajani, A. S., Wannamaker, L.W. Bacteriocins of gram-positive bacteria. Bacteriol. Rev., 1976; 40: 722-756.
- Cotter, P.D., Hill, C., Ross, R.P. Bacteriocins: developing innate immunity for food. Nature Review Microbiology., 2005; 3: 777–788.
- Cleveland, J., Montville, T.J., Nes, I.F., Chikindas, M.L. Bacteriocins: safe, natural antimicrobials for food preservation. International Journal of Food Microbiology., 2001; 71: 1–20.
- Nes, I.F., Diep, D.B., Haverstein, I.S., Brurberg, M.B., Eijsink, V., Holo, H. Biosynthesis of bacteriocins in lactic acid bacteria. Antonie Van Leeuw., 1996; 70: 113–128.
- Forouhandeh, H., Zununi, V.S., Hejazi, M.S., Nahaei, M.R., Akbari, D.M. Isolation and phenotypic characterization of lactobacillus species from various dairy products. Current Research in Bacteriology., 2010; 3(2): 84-88.
- Hindi, N.K.K., Chabuck, Z.A.G. Antimicrobial Activity of Different Aqueous Lemon Extracts. Journal of Applied Pharmaceutical Science., 2013; 3(06): 074-078.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


