ISSN: 0973-7510
E-ISSN: 2581-690X
This study evaluates the effects of supplementing whiteleg shrimp (Penaeus vannamei) feed with Streptomyces strains (TM1, TM2, TM7, TM21, TM22), Bacillus strains (AQ1, BIO2, BAL3), and their combinations at different densities (106, 107, and 108 CFU/g) on growth performance, immune response, water quality, and Vibrio control. Two experiments were conducted over 36 days: the first tested individual Bacillus and Streptomyces consortia at varying doses, and the second assessed combined treatments. Survival was monitored after shrimp were fed probiotics and subsequently challenged with Vibrio parahaemolyticus. Results showed significant improvements in final body weight (FBW), daily weight gain (DWG), feed conversion ratio (FCR), survival rate (SR), and total hemocyte count (THC) in all probiotic treatments, with greater benefits at higher probiotic doses. Combined probiotic treatments consistently produced greater numerical enhancements in growth performance than single-strain applications, indicating synergistic effects between Streptomyces and Bacillus consortia. Water quality responses differed between probiotic types, with Streptomyces treatments providing rapid early reductions in total ammonia nitrogen (TAN) and nitrite, whereas Bacillus-supplemented treatments achieved delayed but more stable and higher removal efficiencies toward the end of the experiment. The combination treatment B8S8 (Bacillus and Streptomyces at 108 CFU/g each) yielded the greatest growth enhancement (approximately 143% FBW and 149% DWG increase relative to the control) and effectively reduced Vibrio counts, achieving 2.9 and 3.1 log reductions in water (B6S8 and B8S8, respectively) and 3.5 and 3.6 log reductions in the shrimp gastrointestinal tract. In combined treatments, TAN and nitrite removal efficiencies exceeded 95% by Day 36, with no significant differences among probiotic combinations. This probiotic supplementation strategy enhances shrimp health and growth in recirculating aquaculture systems at 100 shrimp/100 L, suggesting that combined Streptomyces and Bacillus consortia are a promising approach for improving P. vannamei aquaculture productivity.
Streptomyces, Bacillus, Penaeus Vannamei, Whiteleg Shrimp, RAS, Vibrio parahaemolyticus, Total Ammonia Nitrogen, Nitrite, Total Hemocyte Count, Growth Performance
The role of aquaculture in food security also indirectly impacts GDP by ensuring the stability of food prices and availability, particularly in countries where seafood is a major part of the diet. Therefore, aquaculture can be considered a stabilizing force in the national economy, helping to balance trade and contributing to the overall economy.1 One innovative solution to achieving this sustainability is the use of Recirculating Aquaculture Systems (RAS), which offer advantages over traditional open systems by efficiently managing water resources and improving environmental control.2 RAS can reuse more than 90% of the water in a closed-loop system, minimizing water use and reducing environmental impacts while maintaining optimal conditions for aquaculture.3,4 This system not only supports higher stocking densities of cultured species but also allows for better management of water quality parameters, making it an increasingly popular choice for intensive aquaculture.5 In the 1990s period, various research projects were carried out in the USA aimed at cultivating Litopenaeus vannamei with the use of recirculating raceway systems.6 For the last three decades, researchers have continually explored different RAS techniques for rearing Litopenaeus vannamei. The technologies cover a variety of RAS types, such as raceway RAS,7 elevated pond systems,8 fully enclosed land-based setups,9,10 constructed wetland systems,11 and small experimental RAS facilities.12-14 These systems use industrial methods to regulate water conditions in culture tanks, improving the environment for shrimp growth and survival.14
Bacterial diseases remain one of the most significant challenges in aquaculture, especially those caused by Vibrio species.15 These bacteria are commonly associated with infectious diseases in marine organisms, particularly shrimp. The first 30 days of shrimp culture are a particularly vulnerable period, as postlarvae are in the early stages of development and more prone to infection.16 Various species of Vibrio, including V. harveyi, V. vulnificus, V. parahaemolyticus, V. alginolyticus, and V. penaeicida, play a significant role in the occurrence of vibriosis in aquatic organisms, especially under certain environmental conditions.17,18 Shrimp infected with AHPND often exhibit visible symptoms, including a distended stomach and a reduced size of the hepatopancreas (HP). The mortality rate tends to rise significantly within the first three days of infection, and disease outbreaks generally occur within 8 to 45 days post-stocking.19 The rise and spread of diseases in shrimp have restricted the growth of shrimp farming areas and global productivity, leading to excessive antibiotic use by farmers to reduce disease-related losses. However, in recent years, concerns over the long-term environmental effects and potential risks to human health have led to a backlash against antibiotic use. In 2020, global antimicrobial usage reached approximately 99,500 tons, and it is projected to grow by 8.0%, reaching about 107,500 tons by 2030. The bulk of this usage is concentrated in Asia, accounting for nearly 67% of the total, while Africa uses less than 1%. These trends point to an increase in global antimicrobial consumption by 2030, surpassing earlier 2017 forecasts, with significant growth expected in Asia/Oceania and the Americas.20 The widespread use of antibiotics exerts selection pressure, driving the development of resistance in bacteria. These bacteria adapt to this pressure, primarily through horizontal gene transfer, which can lead to the unclear flow of resistance genes. Resistance mechanisms can emerge through the acquisition of chromosomal material, although these genetic alterations are not transferable to other bacterial cells. Furthermore, some bacterial pathogens are capable of acquiring resistance via plasmid-based mechanisms.20,21
The 2001 FAO/WHO report defines probiotics as beneficial live microbes that, when ingested in sufficient quantities, contribute to the health of the host by modulating the microbial balance. Studies have shown that probiotics can improve nutrition, enhance feed efficiency, strengthen immunity, and increase disease resistance.20 In their 2014 study, Lazado and Caipang emphasized the practical benefits of using probiotics in aquaculture, with their initial demonstration of this potential dating back to 1986.22 Probiotics operate through multiple mechanisms and are essential for promoting the well-being of aquatic species.23
Among the many probiotics, Bacillus and Streptomyces have attracted particular attention due to their beneficial effects in aquaculture. Bacillus species are recognized for producing a range of bioactive compounds, including antimicrobial peptides and enzymes, which help control harmful bacteria, enhance water quality, and support the growth of aquatic species. Similarly, Streptomyces, well-known for its antibiotic production, has demonstrated potential in controlling various marine pathogens, including Vibrio. Bacillus licheniformis DAHB1, isolated from shrimp ponds, is capable of producing an AHL-lactonase enzyme that destroys the communication signal molecule involved in the biofilm formation of Vibrio parahaemolyticus DAHP1.24 Bacillus thuringiensis QQ1 and Bacillus cereus QQ2 have high potential to degrade both chemically synthesized and naturally biosynthesized AHL signal molecules from Vibrio harveyi and Vibrio alginolyticus.25 The bacteriocin CAMT2, which is synthesized by Bacillus amyloliquefaciens isolated from Epinephelus areolatus, demonstrates antibacterial effects against Vibrio parahaemolyticus.26 Bacillus sp. Lts40, obtained from shrimp ponds, exhibits antibacterial properties against Vibrio harveyi, the pathogen responsible for shrimp diseases, by producing bacteriocins.27 Approximately 70% of antibiotics used today are derived from actinomycetes.28,29 These Streptomyces strains produce siderophores that bind iron in the aquatic environment, inhibiting the growth of pathogenic Vibrio strains.30 Streptomyces griseus and Streptomyces nigrescens demonstrate bacteriocin-mediated inhibition of pathogenic bacteria, including Vibrio parahaemolyticus.31 Although both Bacillus and Streptomyces have been studied individually in aquaculture, limited research has explored the combined application of these two genera in recirculating aquaculture systems (RAS) to control Vibrio infections and improve shrimp health. This study aimed to assess the effects of integrating Bacillus and Streptomyces strains in a recirculating aquaculture system for controlling Vibrio levels, improving water quality, and supporting the development of Penaeus vannamei over a 36 day culture period.
Tank preparation and shrimp
The study was carried out at Lien Hiep Phat Science and Technology Co. Ltd., which is located in Ho Chi Minh City, Vietnam. Twelve-day-old whiteleg shrimp postlarvae (PL12) were purchased from Viet Uc Seafood Corporation. The shrimp were acclimatized for 3 days in a 400L HDPE plastic tank. Water parameters in the acclimation tank were regulated to match hatchery standards, with temperature ranging from 28 to 30 °C, salinity at 10‰, dissolved oxygen between 4 and 6 ppm, pH from 7.8 to 8.2, and alkalinity between 140 and 160 ppm. Shrimp were given Tomboy Aquafeed JSC commercial feed, with a daily feeding amount equivalent to 3% of their body weight. After acclimation, the shrimp were moved to the designated RAS, with a stocking density of 1 shrimp per liter.
A RAS unit consists of a 100 L glass rearing tank designed for shrimp farming and connected to a biological filtration system with a working volume of 36 L, resulting in a total operational water volume of 136 L per system. Solid waste is removed using a polyester matting sponge (Biopro, Japan), followed by a moving-bed biofilter containing Kaldnes media (Small Boss, China) and a fixed-bed biofilter with ceramic noodles and volcanic stones as carrier materials (Biopro, Japan). RAS units are illuminated by sunlight through solar panels on the roof. Oxygen is supplied to the system through continuous aeration in both the rearing tanks and the biological filtration units.
Culture water circulates continuously through the system using a 28 W pump (Atman, China) with a nominal flow rate of 2000 L h-1, corresponding to a hydraulic retention time (HRT) of approximately 4 minutes per circulation cycle. Water parameters in the RAS units were adjusted similarly to those during the acclimation period. Prior to shrimp stocking, all RAS units were operated continuously for 14 days to allow biofilter maturation. No external microbial inoculum was added during this period, as the experiment aimed to evaluate the effects of diets supplemented with Bacillus spp. and Streptomyces spp. on nitrogen compound dynamics in the culture water.
Biofilter performance during the experimental period was assessed based on total ammonia nitrogen (TAN) and nitrite (NO2–-N) concentrations measured from the day of shrimp stocking onward. No routine water exchange was applied during the experiment, except for the addition of freshwater to compensate for evaporative losses.
Bacterial strains
The microorganism strains utilized in this project include Streptomyces sp. TM1, Streptomyces sp. TM2, Streptomyces sp. TM7, Streptomyces sp. TM21, and Streptomyces sp. TM22, which were isolated and selected from the water and bottom mud of whiteleg shrimp ponds in previous research.32 Additionally, Bacillus sp. AQ1, Bacillus sp. BIO2, and Bacillus sp. BAL3 were isolated and selected from the mud of Can Gio mangrove forest, Vietnam.33 Vibrio parahaemolyticus was isolated from diseased shrimp provided by Lien Hiep Phat Sci-Tech Company, Ltd.
Vibrio parahaemolyticus and Bacillus strains were cultivated in Tryptone Soy Broth (TSB) medium with 1.5% NaCl at 37 °C for 24-48 hours, while Streptomyces strains were cultured on Gause I medium with 1.5% NaCl at 37 °C for a duration of 7 days. Following cultivation, the centrifugation process was carried out for 20 minutes at a speed of 6000 rpm for the cultures, and the resulting pellet was washed three times with saline (1.5% w/v). After washing, the pellet was resuspended in 1.5% saline to prepare a bacterial stock, with concentrations of 5.0 x 108 CFU/mL for Bacillus strains34,35 and 1.0 x 109 CFU/mL for Streptomyces strains,36,37 determined by measuring the optical density using a spectrophotometer. The bacterial suspensions, with optical density measured at 600 nm, were quantified by counting the colonies formed after plating on solid media, using the spread plate method. Streptomyces strains were cultured on Gause I, while Bacillus strains and Vibrio parahaemolyticus were cultivated on Tryptone Soy Agar (TSA).35,38
Probiotic formulation
Commercial shrimp feed (Tomboy, Skretng Vietnam) was mixed with bacterial stocks prepared as described in the Bacterial strains section, so that the CFU/g feed density reached approximately three levels: 1 x 106, 1 x 107, and 1 x 108 of Bacillus spp.36,37 and Streptomyces spp.39-41 The bacterial cultures were evenly applied to the surface of the feed pellets, which were then left to dry at 40 °C for 24 hours. In the control group, an equal volume of sterile seawater was used in place of the bacterial suspension. To confirm the presence of bacteria in the feed, quantitative analysis was performed using NA with 1.5% NaCl (Nutrient Agar) for Bacillus and Gause I agar for Streptomyces.
Experimental design
Impact of Bacillus spp. and Streptomyces spp. in Feed on Water Quality, Intestinal Vibrio Levels, and Growth Performance of Shrimp
The experiment was conducted over 5 weeks with 21 Recirculating Aquaculture System (RAS) units, divided into 7 treatments, each replicated 3 times. The treatments included B6, B7, B8 (Bacillus spp. AQ1, BIO2, BAL3 in a 1:1:1 ratio), and S6, S7, S8 (Streptomyces spp. TM1, TM2, TM7, TM21, and TM22 in a 1:1:1:1:1 ratio), corresponding to feed formulas containing 1 × 106, 1 × 107, and 1 × 108 CFU/g feed. The control treatment (C) consisted of feed without probiotics.
The shrimp were provided food four times a day, at intervals of 4 hours, with a feeding rate of 5%-10% of their body weight.35,42,43 Water was not replaced during the experiment, except for water lost through evaporation, which was replenished as necessary.
After 5 weeks, the shrimp from the experimental tanks were collected, measured, and tallied to assess SR (Survival rate, %), biomass, FBW (Final body weight, g), DWG (Daily weight gain, g/day), and FCR (feed conversion ratio). These parameters were calculated based on previously described methods.44-46
The number of strains and their internal ratios within each probiotic group were kept constant across treatments, while the total bacterial density (CFU/g feed) was used as the primary experimental variable. Equal ratios among Bacillus strains (1:1:1) and Streptomyces strains (1:1:1:1:1) were applied to ensure uniform representation of each strain and to minimize additional sources of variation. This experimental design allowed the evaluation of dose-dependent effects of Bacillus spp. and Streptomyces spp. supplementation rather than effects related to strain composition or ratio.
Effect of Combining Bacillus spp. and Streptomyces spp. in feed at screened densities on water quality, intestinal Vibrio density, and shrimp growth parameters
Following the results of the initial experiment, the optimal Bacillus spp. and Streptomyces spp. densities for improving water quality, reducing Vibrio densities in the gut, and enhancing shrimp growth performance were determined. The optimal feed formulas for each bacterium were selected for further combination in the next phase of the study.
The feed formulas will be prepared as described in the Probiotic formulation section, ensuring that both Bacillus spp. and Streptomyces spp. are present simultaneously in the feed at the selected densities and inoculation ratios, similar to those used in the initial experiment. This experiment will be conducted with another batch of shrimp, of similar age to the initial batch, purchased from Viet Uc Seafood Corporation. Feeding procedures, experimental duration, and evaluation criteria will remain the same as in the initial experiment.
In the combined treatments, the internal strain ratios of Bacillus spp. and Streptomyces spp. were maintained as defined in the initial experiment. This approach was applied to preserve the previously screened strain composition and to avoid introducing additional experimental variables, thereby allowing the evaluation of the combined effects of Bacillus spp. and Streptomyces spp. supplementation.
Water quality analysis
Fifteen milliliters of water were collected from just beneath the surface, approximately 20 cm deep, in each tank using a transparent, aseptic borosilicate vial with a tight-sealing lid (manufactured by Pyrex, USA). This was accomplished with a plastic Pasteur pipette to analyze ammonia and nitrite levels, and to assess the density of Bacillus, actinomycetes, and Vibrio. The quantification of TAN and Nitrite was performed using APHA 4500 NH3 F – Phenate method and APHA 9245 B – Multiple tube method, respectively.47 Bacterial counts were performed using the spread plate method. The water samples were diluted tenfold using saline (0.9% NaCl), and 100 µL aliquots of these dilutions were spread on different types of agar: nutrient agar (NA) with 1.5% NaCl for total heterotrophic count, Gause I agar for actinomycetes detection, and thiosulfate-citrate-bile-salt-sucrose (TCBS) agar for enumerating Vibrio counts. Bacillus enumeration was carried out using the heat-cold shock method,48 where the sample was heated to 85 °C for 15 minutes to eliminate vegetative cells and promote spore formation, followed by rapid cooling on ice for 1-2 minutes. The total Bacillus count was determined by plating on NA medium with 1.5% NaCl.
Determination of bacterial counts in shrimp gut
After completing the experiment, the digestive systems of the shrimp, encompassing the stomach, hepatopancreas, and intestines, were extracted from three shrimp chosen at random from each experimental tank. After weighing and homogenizing the samples in saline, a tenfold dilution was performed. The diluted samples were analyzed for microbial populations, such as Vibrio, Bacillus, heterotrophic bacteria, and actinomycetes, employing techniques outlined in the Water Quality Analysis section.
Total hemocyte count
Three shrimp from each experimental tank were randomly selected for blood collection to assess the total hemocyte count at the end of the experiment. A 27 gauge needle was used to collect 50 µL of hemolymph from the first abdominal segment’s ventral side. The sample was then mixed with 450 µL of an anticoagulant solution (EDTA 1 × 10-2 M, trisodium citrate 3 × 10-2 M, citric acid 2.6 × 10-2 M, NaCl 45 × 10-2 M, and glucose 10 × 10-2 M), and the solution’s pH was adjusted to 4.6 using HCl (1 M).49,50 This mixture was stored on ice and fixed with a 6% formaldehyde solution in a 1:3 ratio. The post-fixed blood mixture was then combined with 20 µL of Rose Bengal (1.2% in 50% ethanol solution) and allowed to incubate for 20 minutes.49 The total hemocyte count was assessed with a Neubauer counting chamber under a transmission microscope, with the results expressed in cells/mL.51,52
Challenge with Vibrio parahaemolyticus
The challenge was conducted through immersion using a suspension of Vibrio parahaemolyticus (1.0 x 106 CFU/mL and 1.0 x 108 CFU/mL).53 This challenge was administered following a 5 week period of probiotic administration. There were two experiments conducted. In Experiment 1, a total of seven treatments, including the control group, were tested at each challenge density, with each group consisting of ten animals and three replicates. After obtaining the results from Experiment 1, selected treatments were chosen for further testing in Experiment 2, which followed a similar setup with the same challenge densities and animal groups. Survival rates were recorded daily over a four-day period.
The Vibrio infection experiments were conducted under strictly controlled conditions. All equipment, including tanks, water containers, and sampling tools, was sterilized with a 70% ethanol solution before and after each use. To prevent cross-contamination, personnel handling the shrimp wore gloves, lab coats, safety glasses, and masks. Access to the experimental area was restricted to minimize the risk of introducing external pathogens.
The culture water was initially treated with sodium hypochlorite (5% – Xilong, China) at a concentration of 30 ppm. After a 12 hour incubation period, the residual chlorine concentration was measured using a Sera Chlorine Test Kit to ensure complete neutralization of chlorine before stocking the shrimp. These biosecurity measures were implemented to maintain the integrity of the study and ensure the safety of the experimental environment.
Statistical methods
One-way ANOVA was applied to assess the differences between groups, with statistical significance determined at a significance level of P < 0.05. Following this, pairwise comparisons were conducted using Fisher’s test (Statgraphics Centurion XV) to identify specific treatment effects.
Impact of Bacillus spp. and Streptomyces spp. in feed on water quality, intestinal Vibrio levels, and growth performance of shrimp
Bacterial parameters and total hemocyte count
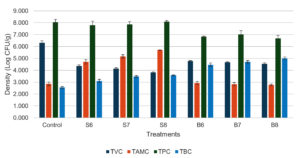
Supplementary Figure S1 shows the progression of the total Vibrio count (TVC) during the experiment. At the start of the experiment, TVC varied from 1.059-1.905 log CFU/mL. TVC increased over the first two weeks (D15), with values ranging from 2.626-3.395 log CFU/mL across treatments, while the control had a value of 3.950 log CFU/mL. From week 4 (D22) onward, TVC in the Streptomyces–fed treatments decreased and stabilized, with log CFU/mL values of 3.094 ± 0.056 (S6), 3.011 ± 0.061 (S7), and 2.948 ± 0.021 (S8), showing no significant differences among them. In the Bacillus–added treatments, TVC remained stable from week 4 onwards, with log CFU/mL values of 3.827 ± 0.065 (B6), 3.825 ± 0.046 (B7), and 3.576 ± 0.035 (B8). Treatment B8 showed a significantly lower count compared to B7 and B6, but significantly higher than S6, S7, and S8. The TVC in all treatments was notably lower than that in the control treatment (5.303 ± 0.125 log CFU/mL). For total actinomycetes count (TAMC), actinomycetes appeared in the culture water after 1 week of feeding with Streptomyces–containing feed, but not in the control or Bacillus treatments. After 4 weeks, actinomycetes were present in all treatments, with higher densities in those supplemented with probiotics (Figure S2). By the end of the experiment, the log CFU/mL values were 1.651 ± 0.090 (Control), 3.946 ± 0.095 (S6), 4.122 ± 0.037 (S7), 4.310 ± 0.087 (S8), 2.822 ± 0.144 (B6), 2.680 ± 0.208 (B7), and 2.504 ± 0.115 (B8), with S6, S7, and S8 having significantly higher actinomycete density than the other treatments. Regarding the total heterotrophic plate count (TPC), no considerable variations were observed among treatments throughout the experimental period (Figure S3). In terms of total Bacillus count (TBC), Bacillus density tended to increase over time in all treatments (Figure S4). At the end of the experiment, Bacillus-fed treatments (B6-B8) showed significantly higher TBC values compared to the other treatments, with log CFU/mL values of 2.881 ± 0.076 (Control), 3.098 ± 0.024 (S6), 3.088 ± 0.047 (S7), 3.144 ± 0.068 (S8), 3.477 ± 0.106 (B6), 3.400 ± 0.072 (B7), and 3.608 ± 0.133 (B8). Figure 1 illustrates the overall levels of TVC, TPC, TBC, and TAMC in the shrimp gastrointestinal tract. TVC in the probiotic treatments was markedly lower than the control group (6.304 ± 0.179 log CFU/g). No notable variations in TVC were observed with values of log CFU/g across B6 (4.774 ± 0.045), B7 (4.689 ± 0.045), and B8 (4.454 ± 0.086), while S6 (4.368 ± 0.094) showed similar values to B8 and S7 (4.134 ± 0.083). TVC in S8 (3.823 ± 0.043 log CFU/g) was considerably lower than that in all other treatments. TAMC (log CFU/g) was significantly higher in the Streptomyces treatments, particularly in S7 (5.172 ± 0.142) and S8 (5.698 ± 0.026). Bacillus supplemented treatments (B6, B7, B8) showed no significant difference from the control, with TAMC values of 2.922-2.855 log CFU/g. TBC was significantly higher in B6 (4.467 ± 0.145), B7 (4.711 ± 0.111), and B8 (4.997 ± 0.116) compared to the other treatments, while the control group showed the lowest count (2.554 ± 0.075 log CFU/g). The addition of Bacillus reduced TPC in the gastrointestinal tract, with B6 (6.834 ± 0.059), B7 (7.023 ± 0.313), and B8 (6.687 ± 0.256) showing significantly lower values compared to S6 (7.797 ± 0.336), S7 (7.864 ± 0.227), S8 (8.109 ± 0.095), and the control (8.032 ± 0.239).
Figure 1. Bacterial counts in shrimp gastrointestinal tract after 36 days of feeding different densities of Bacillus spp. and Streptomyces spp.
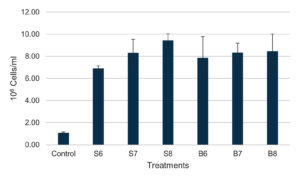
After 36 days of the experiment, both Streptomyces spp. and Bacillus spp. notably elevated the total hemocyte count (THC) in shrimp, with observed values (106 cells/ml) of 6.91 ± 0.23 (S6), 8.31 ± 1.22 (S7), 9.44 ± 0.60 (S8), 7.88 ± 1.90 (B6), 8.34 ± 0.86 (B7), and 8.45 ± 1.56 (B8), all of which were considerably higher than those of the control group (1.08 ± 0.08) (Figure 2). Nonetheless, no notable variations in THC were found among the treatments.
Figure 2. Total hemocyte count of shrimp after 36 days of feeding different densities of Bacillus spp. and Streptomyces spp.
Concentrations of Total Ammonia Nitrogen (TAN) and Nitrite in the Rearing Water
The TAN concentration in the rearing water of the control group increased throughout the experiment, reaching 5.770 ± 0.397 ppm by D36 (Figure S5A). In treatments with Streptomyces, TAN remained stable until D26 (0.085-0.089 ppm) and gradually increased by D36, reaching 0.865 ± 0.233 ppm (S6), 0.958 ± 0.113 ppm (S7), and 0.973 ± 0.154 ppm (S8). In contrast, TAN levels in Bacillus supplemented treatments spiked on D8 (1.085 ± 0.039 ppm for B6, 0.971 ± 0.113 ppm for B7, and 0.942 ± 0.027 ppm for B8), then decreased after D12 and stabilized with values ranging from 0.037 to 0.115 ppm by D36. When TAN removal efficiency (RE%) for probiotic treatments was calculated using the accumulated TAN trend of the control as the baseline, clear time-dependent differences were observed between probiotic groups (Figure S5B). The Streptomyces treatments (S6-S8) showed high RE% early in the trial (D4-D8; approximately 55.00%-88.86%) and generally maintained high performance through the mid-phase (D12-D26; approximately 75.16%-96.48%); however, RE% declined markedly from D29 onward, reaching approximately 35.64%-42.32% at D36. In contrast, the Bacillus treatments (B6-B8) exhibited low to negative RE% at the beginning (D4-D8; -31.51% to -0.09%), but RE% increased substantially from D12 and remained consistently high thereafter, exceeding approximately 89.34% from D15-D36 and reaching approximately 95.74%-97.07% at D36. Overall, Streptomyces produced a rapid but less persistent reduction pattern, whereas Bacillus achieved a delayed yet more stable and superior TAN removal toward the end of the experiment.
Similarly, the nitrite concentration in the control group increased over time, reaching 5.770 ± 0.397 ppm on D36 (Figure S6A). In Streptomyces fed treatments, nitrite levels began increasing from D8 and peaked at 0.917 ± 0.244 ppm on D36 in S6, while S7 and S8 showed stable levels from D22 to D36. In Bacillus spp. fed treatments, nitrite levels peaked on D12 (2.515 ± 0.267 ppm for B6, 2.049 ± 0.200 ppm for B7, and 1.834 ± 0.084 ppm for B8), before declining by D19 and stabilizing for the remainder of the study, with concentrations ranging from 0.036-0.180 ppm. Nitrite removal efficiency (RE%) increased over time in all probiotic treatments, but the magnitude and stability differed by probiotic type (Figure S6B). In the Streptomyces treatments (S6-S8), RE% was modest and more variable at the beginning (D4-D8), then rose sharply by D12 and remained consistently high through the end of the trial, ranging from 67.23%-88.73% at later sampling points. The Bacillus treatments (B6-B8) started with scattered responses, including negative RE% in some cases, yet demonstrated a pronounced improvement after D12. From D19 onward, Bacillus treatments maintained near-complete nitrite removal (RE% between 92.77% and 99.33%), staying above 96% at D36. Starting from Day 22, Bacillus treatments had significantly higher nitrite removal efficiency than the Streptomyces treatments.
Shrimp growth performance
The growth performance of shrimp fed diets supplemented with single strains of Bacillus spp. and Streptomyces spp. at different densities is presented in Table 1. All probiotic treatments significantly enhanced shrimp growth compared with the control group (P < 0.05). Final body weight (FBW) increased progressively with increasing probiotic density, with the highest value observed in the S8 treatment (0.53 ± 0.02 g), which was significantly higher than all other treatments. FBW in S6 was comparable to B6, while S7 showed similar values to B7 and B8.
Table (1):
Growth performance of shrimp after 36 days of feeding different densities of Bacillus spp. and Streptomyces spp.
| Treatment | Growth Performance | |||
|---|---|---|---|---|
| FBW (g) | DWG (g/day) | FCR | SR (%) | |
| Control | 0.27 ± 0.04a | 0.0071 ± 0.0010a | 1.82 ± 0.25b | 64.67 ± 2.19a |
| S6 | 0.35 ± 0.02b | 0.0095 ± 0.0005b | 1.24 ± 0.05a | 94.67 ± 0.88b |
| S7 | 0.43 ± 0.02d | 0.0118 ± 0.0005cd | 1.10 ± 0.03a | 96.67 ± 1.45b |
| S8 | 0.53 ± 0.02e | 0.0144 ± 0.0004e | 1.06 ± 0.03a | 97.00 ± 0.58b |
| B6 | 0.36 ± 0.02bc | 0.0097 ± 0.0005bc | 1.28 ± 0.06a | 94.33 ± 0.33b |
| B7 | 0.41 ± 0.00cd | 0.0113 ± 0.0001bcd | 1.19 ± 0.02a | 95.67 ± 1.45b |
| B8 | 0.44 ± 0.02d | 0.0119 ± 0.0005d | 1.09 ± 0.02a | 96.00 ± 0.58b |
The data are shown as mean ± standard error, with different letters after the standard error representing statistically significant differences (P < 0.05)
Daily weight gain (DWG) followed a similar pattern, increasing with probiotic supplementation. The S8 group exhibited the highest DWG (0.0144 ± 0.0004 g/day), which was significantly greater than those of all other treatments. DWG in S6 did not differ significantly from B6 and B7, while S7 showed comparable performance to Bacillus-supplemented diets. In contrast, no significant differences in feed conversion ratio (FCR) and survival rate (SR) were observed among probiotic treatments, although all probiotic-fed shrimp exhibited significantly lower FCR and higher SR than the control (P < 0.05).
To further quantify the magnitude of growth enhancement, relative improvement percentages compared with the control are summarized in Table 2. Probiotic supplementation resulted in substantial increases in FBW and DWG, with FBW improvement ranging from 38.99%-107.21% and DWG improvement from 42.54%-111.36%. The greatest numerical improvements were consistently observed in the S8 treatment, indicating that higher densities of Streptomyces spp. exerted the strongest growth-promoting effect among single-strain applications.
Table (2):
Relative improvement (%) in growth performance of shrimp compared with the control after 36 days of feeding different densities of Bacillus spp. and Streptomyces spp.
| Treatment | Relative Improvement (%) In Growth Performance | |||
|---|---|---|---|---|
| FBW improvement (%) | DWG improvement (%) | FCR reduction (%) | SR improvement (%) | |
| S6 | 38.99 ± 25.31a | 40.67 ± 26.43a | 28.33 ± 13.57a | 49.11 ± 6.18a |
| S7 | 70.65 ± 29.83a | 73.49 ± 31.29a | 36.64 ± 10.91a | 52.33 ± 7.25a |
| S8 | 107.21 ± 34.45a | 111.36 ± 36.26a | 38.83 ± 9.81a | 52.75 ± 5.79a |
| B6 | 40.87 ± 25.93a | 42.54 ± 27.12a | 26.59 ± 11.89a | 48.53 ± 5.28a |
| B7 | 62.09 ± 22.80a | 64.48 ± 23.94a | 31.87 ± 9.90a | 50.61 ± 5.63a |
| B8 | 69.03 ± 16.82a | 71.60 ± 17.76a | 37.78 ± 8.19a | 51.08 ± 4.39a |
Data are expressed as mean ± standard error. Percentage improvement was calculated relative to the control. Different letters indicate significant differences among treatments (P < 0.05)
Feed utilization efficiency was markedly improved in all probiotic treatments, as reflected by FCR reductions of 26.59%-38.83% relative to the control. Survival rate also increased considerably, showing improvements of approximately 48%-53%. However, no statistically significant differences were detected among probiotic treatments for any relative improvement parameter (P > 0.05), suggesting that both Bacillus spp. and Streptomyces spp. effectively enhanced shrimp growth and survival across the tested densities.
Challenge with Vibrio parahaemolyticus
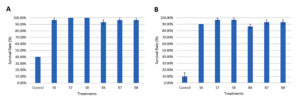
Following 36 days of feeding shrimp with diets containing Streptomyces spp. and Bacillus spp., the shrimp in the treatments were exposed to a challenge with Vibrio parahaemolyticus at 106 CFU/mL and 108 CFU/mL. After a 4 day challenge, the survival rates of shrimp in the probiotic-supplemented groups at 106 CFU/mL were 96.67% (S6), 100.00% (S7), 100.00% (S8), 93.33% (B6), 96.67% (B7), and 96.67% (B8), which were notably greater than the control group, which had a survival rate of 40.00%. For the 108 CFU/mL challenge, the survival rates were 90.00% (S6), 96.67% (S7), 96.67% (S8), 86.67% (B6), 93.33% (B7), and 93.33% (B8), with the control group showing a survival rate of 10.00% (Figure 3). No notable variations in survival rates were found across the treatments supplemented with probiotics for either concentration.
Figure 3. Survival rate of shrimp after 4 days of exposure to Vibrio parahaemolyticus at 106 CFU/mL (A) and 108 CFU/mL (B) following 36 days of feeding with different densities of Bacillus spp. and Streptomyces spp.
Effect of Combining Bacillus spp. and Streptomyces spp. in Feed at Screened Densities on Water Quality, Intestinal Vibrio Density, and Shrimp Growth Parameters
Based on the findings from the first experiment, the densities of Streptomyces spp. and Bacillus spp. were chosen for inclusion in the shrimp diet at concentrations of 106 CFU/g and 108 CFU/g, respectively. The experiment was designed with five treatments, including a control group with no probiotic supplementation. The probiotic treatments were added to the feed as follows: B6S6 (Streptomyces consortium with 106 CFU/g and Bacillus consortium with 106 CFU/g), B6S8 (Streptomyces consortium with 106 CFU/g and Bacillus consortium with 108 CFU/g), and B8S8 (Streptomyces consortium with 108 CFU/g and Bacillus consortium with 108 CFU/g). The experimental procedure followed the protocol established in the previous experiment. Water quality parameters, shrimp growth performance, survival rates after infection with Vibrio parahaemolyticus, and total hemocyte counts were measured upon completion of the 5 week experimental period.
Bacterial parameters and Total hemocyte count
The TVC (log CFU/mL) in the control treatment increased from 1.661 ± 0.276 at D0 to 4.991 ± 0.003 at D36 (Figure S7). Treatments with higher levels of Streptomyces spp. (B6S8 and B8S8) showed significantly lower TVC than those with lower levels (B6S6 and B8S6) by D36, with log CFU/mL values of 2.039 ± 0.029, 1.872 ± 0.075, 3.275 ± 0.010, and 3.275 ± 0.068 for B6S8, B8S8, B6S6, and B8S6. No significant differences in TPC were observed across treatments, with values ranging from 5.345-5.676 log CFU/mL (Figure S8). In treatments supplemented with high levels of Bacillus spp. and Streptomyces spp.
(108 CFU/g), both TBC and TAMC were notably elevated compared to the control (Figures S9 and S10). At D36, TBC (log CFU/mL) was highest in B8S6 (3.570 ± 0.016) and B8S8 (3.591 ± 0.050), significantly higher than B6S6 (3.357 ± 0.081) and B6S8 (3.331 ± 0.040). TAMC (log CFU/mL) was highest in B6S8 (4.069 ± 0.075) and B8S8 (4.057 ± 0.062), significantly different from B6S6 (3.741 ± 0.060) and B8S6 (3.597 ± 0.144).
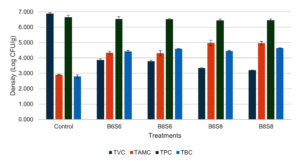
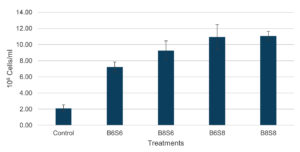
The microbial density in the shrimp digestive tract showed that probiotic-supplemented diets reduced TVC by approximately 3 log compared to the control (Figure 4). High-level Streptomyces spp. treatments (B6S8 and B8S8) had significantly lower TVC values (3.346 ± 0.046 and 3.205 ± 0.015 log CFU/g) compared to low-level treatments (3.867 ± 0.076 and 3.791 ± 0.065 log CFU/g). TPC showed consistent values, ranging from 6.445-6.641 log CFU/g, with no significant changes observed. TAMC and TBC in probiotic-supplemented treatments showed significantly higher values than the control group. B6S8 and B8S8 had significantly higher TAMC values (4.974 ± 0.179 and 4.958 ± 0.110 log CFU/g) than B6S6 and B8S6 (4.338 ± 0.088 and 4.306 ± 0.167 log CFU/g), while TBC in B8S8 was highest (4.645 ± 0.015 log CFU/g), significantly higher than B6S6 (4.423 ± 0.074 log CFU/g). The combination of Streptomyces spp. and Bacillus spp. significantly increased total hemocyte count (THC), with values of 2.09 ± 0.44 (Control), 7.22 ± 0.63 (B6S6), 9.27 ± 1.19 (B8S6), 10.94 ± 1.55 (B6S8), and 11.08 ± 0.56 (B8S8). THC in B6S8 and B8S8 was significantly higher than in B6S6 but not different from B8S6 (Figure 5).
Figure 4. Bacterial counts in shrimp gastrointestinal tract after 36 days of feeding with combined Bacillus spp. and Streptomyces spp.
Figure 5. Total hemocyte count of shrimp after 36 days of feeding with combined Bacillus spp. and Streptomyces spp.
Concentrations of Total Ammonia Nitrogen (TAN) and Nitrite in the Rearing Water
Figures S11A and S12A show that TAN and nitrite levels in the control treatment gradually increased throughout the experiment, with TAN values reaching up to 1.605 ppm and nitrite values up to 2.694 ppm by the end of the experiment. In the probiotic-supplemented treatments, TAN and nitrite also increased gradually, peaking on Day 15, before decreasing gradually from Day 19 until the end of the experiment. No significant differences were observed among these treatments. By Day 36, the TAN and nitrite values in the probiotic treatments ranged from 0.055-0.070 ppm and 0.107-0.124 ppm, respectively.
In terms of TAN removal efficiency (RE%) (Figure S11B), all probiotic treatments (B6S6, B8S6, B6S8, and B8S8) showed substantial improvements in TAN removal, particularly on Days 12, 15, and 19. B8S6 and B8S8 demonstrated a peak TAN removal efficiency of 98.61 ± 0.35% and 98.36 ± 0.37% on Day 22, respectively. At Day 36, the TAN removal efficiencies in the probiotic treatments ranged from 95.63%-96.58%. Notably, there were no significant statistical differences among the four probiotic treatments (B6S6, B8S6, B6S8, and B8S8), with all treatments demonstrating similar removal efficiencies. Overall, B8S6 and B8S8 exhibited better and more stable TAN removal efficiency compared to B6S6 and B6S8 throughout the experiment. Similarly, for nitrite removal efficiency (RE%) (Figure S12B), the probiotic treatments also exhibited increased removal efficiency over time, with B8S6 showing the highest removal efficiency at 99.09 ± 0.02% on Day 29. However, this was not significantly different from B8S8, which had a RE% of 98.53 ± 0.35%. By Day 36, nitrite removal efficiencies in the probiotic treatments ranged from 95.35% to 96.05%, with no significant statistical differences between the treatments.
In summary, the supplementation of Bacillus spp. and Streptomyces spp. (at concentrations of 106 and 108 CFU/g) significantly improved both TAN and nitrite removal efficiencies in the rearing water.
Shrimp growth performance
After 36 days, diets supplemented with combined Bacillus spp. and Streptomyces spp. significantly enhanced shrimp growth performance compared with the control (Table 3). Treatments containing higher probiotic densities (B8S6, B6S8, and B8S8) resulted in significantly greater FBW and DWG than the low-density treatment (B6S6) (P < 0.05). Among all combinations, B8S8 exhibited the highest FBW (0.64 ± 0.05 g) and DWG (0.0174 ± 0.0014 g/day), indicating superior growth stimulation when both probiotics were applied at high density. Feed utilization efficiency was also improved in probiotic-fed groups, as reflected by significantly lower FCR values compared with the control, while survival rate was significantly increased across all combined treatments (P < 0.05).
Table (3):
Growth performance of shrimp after 36 days of feeding with combined Bacillus spp. and Streptomyces spp.
| Treatment | Growth Performance | |||
|---|---|---|---|---|
| FBW (g) | DWG (g/day) | FCR | SR (%) | |
| Control | 0.27 ± 0.02a | 0.0072 ± 0.0006a | 1.40 ± 0.07c | 82.00 ± 1.73a |
| B6S6 | 0.39 ± 0.03b | 0.0106 ± 0.0009b | 1.17 ± 0.03b | 92.33 ± 0.88b |
| B8S6 | 0.51 ± 0.01c | 0.0139 ± 0.0004c | 1.02 ± 0.01a | 96.67 ± 0.88c |
| B6S8 | 0.52 ± 0.02c | 0.0143 ± 0.0004c | 1.00 ± 0.01a | 97.33 ± 0.33c |
| B8S8 | 0.64 ± 0.05d | 0.0174 ± 0.0014d | 1.02 ± 0.01a | 96.67 ± 1.45c |
The data are shown as mean ± standard error, with different letters after the standard error representing statistically significant differences (P < 0.05)
Relative improvement analysis further confirmed the growth-promoting effects of probiotic combinations (Table 4). FBW and DWG increased by 49.69%-142.67% and 51.79%-148.50%, respectively, compared with the control. The greatest numerical improvements were consistently observed in B8S8, which showed significantly higher FBW and DWG enhancement than B6S6 (P < 0.05), while exhibiting comparable performance to B8S6 and B6S8. Feed conversion ratio was reduced by 16.41%-28.20%, and survival rate improved by 12.72%-19.17% across treatments, with no significant differences among combined probiotic groups (P > 0.05).
Table (4):
Relative improvement (%) in growth performance of shrimp compared with the control after 36 days of feeding with combined Bacillus spp. and Streptomyces spp.
| Treatment | Relative Improvement (%) In Growth Performance | |||
|---|---|---|---|---|
| FBW (g) | DWG (g/day) | FCR | SR (%) | |
| B6S6 | 49.69 ± 23.38a | 51.79 ± 24.49a | 16.41 ± 5.22a | 12.72 ± 2.88a |
| B8S6 | 92.54 ± 17.04ab | 96.27 ± 17.97ab | 26.78 ± 3.03a | 18.04 ± 3.55a |
| B6S8 | 96.48 ± 9.42%ab | 100.33 ± 10.09ab | 28.20 ± 2.87a | 18.80 ± 2.53a |
| B8S8 | 142.67 ± 37.12b | 148.50 ± 39.07b | 27.01 ± 3.98a | 19.17 ± 2.00a |
Data are expressed as mean ± standard error. Percentage improvement was calculated relative to the control. Different letters indicate significant differences among treatments (P < 0.05)
Challenge with Vibrio parahaemolyticus
Following 36 days of feeding with combined Streptomyces spp. and Bacillus spp., shrimp were exposed to Vibrio parahaemolyticus at 106 CFU/mL and 108 CFU/mL. After 4 days of exposure, survival rates in the probiotic-supplemented treatments at 106 CFU/mL were 96.67% (B6S6), 100.00% (B8S6), 100.00% (B6S8), and 100.00% (B8S8), all of which were notably higher than the control group, which had a survival rate of 43.33%. For the 108 CFU/mL challenge, survival rates were 93.33% (B6S6), 96.67% (B8S6), 96.67% (B6S8), and 96.67% (B8S8), significantly higher than the control group, which had a survival rate of 10.00% (Figure 6). No significant variation in survival rates was observed between the probiotic treatments for either concentration.
Probiotics are among the most widely proven solutions for controlling and antagonizing pathogens in various host species, including Vibrio spp.54 Several probiotic bacterial strains have been identified that can control pathogenic Vibrio in aquaculture, including Bacillus spp.55,56 and Streptomyces spp.57 Bacillus and Streptomyces strains are capable of controlling Vibrio through various mechanisms, such as quorum quenching,24,58 bacteriocins,26,31 and other secondary compounds, such as siderophores,30 quinone antibiotics,59 peptide antibiotics,60 aminoglycoside antibiotics,61 and macrolide antibiotics.62 In the present study, we did not conduct direct assays (e.g., AHL degradation, gene expression, enzyme activity, or metabolite profiling) to verify these mechanisms; therefore, they should be considered as plausible explanations based on prior literature rather than confirmed pathways in our experimental system.
This study showed that shrimp diets enriched with Bacillus spp., Streptomyces spp., or a mix of both effectively reduced TVC in the culture water and TVC in the gastrointestinal tract. Additionally, incorporating a mix of Streptomyces spp. and Bacillus spp. into the feed at three varying concentrations (106, 107, and 108 CFU/g feed) proved effective in lowering TVC in the culture water after 2 weeks of the experiment. This reduction was maintained consistently throughout the remainder of the study, suggesting a sustained effect of the microbial supplementation. The Streptomyces consortium reduced the TVC in both rearing water (by 2.2-2.3 log CFU/mL) and the shrimp digestive tract (by 1.9-2.4 log CFU/g) compared to the control group. In comparison, the Bacillus consortium led to reductions of 1.4 to 1.7 log units in rearing water and 1.5-1.7 log units in the shrimp digestive tract. These results are consistent with earlier research, which reported that the addition of Bacillus megaterium and Streptomyces fradiae to the diet of Penaeus monodon postlarvae led to a reduction in Vibrio density, with decreases of approximately 0.87 and 0.89 log units in the rearing water and 1.36 and 1.40 log units in the shrimp postlarvae, respectively.63 When combining two consortia of Streptomyces spp. and Bacillus spp. at densities of 106 and 108 CFU/g of feed, the higher-density consortia (B6S8 and B8S8) showed greater effectiveness in controlling total Vibrio in water after just 1 week, reducing the maximum TVC by 2.9 to 3.1 log units in water and 3.5 to 3.6 log units in the shrimp digestive tract. These reductions were higher compared to the lower-density consortia (B6S6 and B8S6), which resulted in a reduction of TVC in water by 1.7-2.5 log CFU/mL and in the shrimp digestive tract by 1.6-1.7 log CFU/g. Previous studies have also demonstrated that adding a combination of Bacillus consortium and Streptomyces consortium to the diet of whiteleg shrimp resulted in a reduction of TVC in the rearing water (by 4.1 log units) and in the hepatopancreas (by 3.6 log units), relative to the control group.52 These findings suggest that, in terms of reducing TVC, this combination in shrimp diets effectively controls TVC in both the rearing water and the gastrointestinal tract of the shrimp, outperforming the use of individual probiotics. Furthermore, these probiotics did not alter the density of total heterotrophic bacteria in the culture water or shrimp digestive tract, suggesting that they do not negatively impact water quality and help maintain a stable bacterial population in the shrimp digestive tract. The presence of total heterotrophic bacteria is essential in the process of decomposing and mineralizing organic matter.64 The counts of total actinomycetes and Bacillus in both the water and the shrimp digestive tract were higher than those in the control group, with the density increasing in correlation to the probiotic levels in the feed. Therefore, the addition of a Bacillus consortium combined with a Streptomyces consortium to the shrimp diet had a stable effect in controlling TVC in both the digestive tract and the rearing water, contributing to the maintenance of shrimp health and preventing diseases caused by Vibrio infections. Taken together, our results demonstrate a consistent reduction in TVC; however, the specific inhibitory mechanisms remain to be elucidated in future work using targeted functional assays.
The increased TAMC and TBC in the culture water may explain the effective reduction of TAN and nitrite levels. Based on the observed trends, the Bacillus spp. treatment provided strong and stable nitrogen removal, achieving approximately 98% TAN removal efficiency (TAN RE%) and ~99% nitrite removal efficiency (Nitrite RE%). In contrast, the Streptomyces spp. treatment reached approximately 96% TAN RE% and ~89% Nitrite RE%. Notably, despite its relatively high peak performance, the Streptomyces spp. treatment showed weaker TAN control toward the end of the experiment, as evidenced by the decline in TAN RE% to the lowest range (~36%-43%). These findings align with earlier research on Bacillus sp. strains, including BIO2, BAL3, and AQ1, which have shown their capacity to reduce TAN and nitrite levels in both in vitro and in vivo environments.33,35 Several studies have shown that Streptomyces strains are capable of metabolizing various nitrogen sources. For instance, He et al. reported that the S. mediolani EM-B2 strain achieved maximum removal rates of ammonia, nitrite, and nitrate of 3.46, 1.71, and 1.73 ppm·h-1, respectively. Furthermore, enzymes associated with nitrogen metabolism were identified in this strain, such as ammonia monooxygenase-AMO, hydroxylamine oxidoreductase-HAO, nitrate reductase-NR, and nitrite reductase-NiR.65 Thus, the decrease in the TAN control rate towards the end of the experiment in treatments supplemented with only Streptomyces spp. may be due to the accumulation of TAN in the water, surpassing the metabolic capacity of these strains. Alternatively, it could result from the assimilation process, where these strains use nitrite and nitrate as nitrogen sources, converting them into ammonia through enzymes such as nasA (nitrate reductase assimilatory, NO3– ® NO2–) and nirBD (nitrite reductase assimilatory, NO2– ® NH4+), with the support of the nitrate transport protein (Nark-type NO3– transporter).66-68 These genus-specific patterns provide a clear rationale for co-supplementation, as Bacillus spp. ensured robust baseline TAN/nitrite control, whereas Streptomyces spp. contributed broader nitrogen metabolic functions but appeared more vulnerable to late-stage constraints. Accordingly, the combined Bacillus spp. and Streptomyces spp. treatment was expected to improve functional complementarity and stability. Consistent with this expectation, all combined formulations (B6S6, B8S6, B6S8, and B8S8) achieved near-complete peak removal and maintained high end-point performance for both TAN and nitrite (Figures S11 and S12). The combined treatments also showed similar removal efficiencies across formulations, with no significant differences detected. Notably, unlike the Streptomyces spp. treatment, co-supplementation did not exhibit late-stage deterioration in TAN control, supporting improved robustness under prolonged rearing conditions. Overall, co-supplementation at 106 -108 CFU/g achieved near-ceiling peak TAN/nitrite removal while maintaining stable end-point efficiencies, supporting its advantage over genus-only treatments for sustained nitrogen management in rearing water.
Numerous studies have demonstrated that probiotics can enhance the immune response of shrimp to Vibrio infection. The total hemocyte count (THC) serves as a key indicator of the shrimp’s immune response to pathogens, such as Vibrio.69,70 In this study, both the Streptomyces consortium and the Bacillus consortium significantly increased THC of shrimp, with the combination of both consortia showing the greatest effect in comparison to the control group. THC showed an increasing trend with higher probiotic content in the feed, reaching its peak when Streptomyces spp. was combined with Bacillus spp. These results align with earlier research, which showed that a 0.4% concentration of Bacillus cereus significantly increased total hemocyte numbers (259 × 105 cells/mL), 47% higher than the control in P. monodon. Furthermore, the THC increased with higher Bacillus cereus density in the shrimp diet.71 The Bacillus subtilis S12 strain significantly increased THC to 4.71 × 106 cells/mL, 27% higher than the control in whiteleg shrimp.72 Additionally, Bacillus licheniformis has demonstrated its ability to boost the immune response in shrimp, raising THC to 45.8 × 105 cells/mL, a 145% increase compared to the control group.73 Streptomyces sp. (RL8 or N7), and combinations of N7 and RL8, or Bacillus and Streptomyces strains, increased THC in shrimp by 43% (4.0 million cells/mL), 111% (5.9 million cells/mL), 279% (10.6 million cells/mL), and 200% (8.4 million cells/mL), respectively, relative to the control group.52
An increase in THC improves the health of whiteleg shrimp by enhancing phagocytic capacity, which is vital for defending the shrimp against microbial threats.74,75 All treatments supplemented with probiotics at varying concentrations, either alone or in combination, provided strong protection against Vibrio parahaemolyticus challenge in whiteleg shrimp. The increased TBC and TAMC in the shrimp digestive tract, with their ability to control Vibrio through antibacterial compounds and stimulate immune response via increased THC, contributed to this enhanced protection. This finding is consistent with the research by Bernal et al., which reported that treatments containing Streptomyces or a Streptomyces–Bacillus combination enhanced the survival of whiteleg shrimp following infection with Vibrio parahaemolyticus.52 Moreover, it has been demonstrated that the application of Streptomyces sp. CLS-28 notably enhanced the survival rate of black tiger shrimp after infection with Vibrio harveyi.76
It has been demonstrated that Streptomyces and Bacillus improve the growth performance of different livestock species.77,78 In this study, varying dietary supplementation concentrations of the Bacillus consortium, Streptomyces consortium, or particularly their combination, significantly enhanced the growth parameters (FBW, FCR, DWG) and survival of whiteleg shrimp relative to the control group. A clear dose-dependent response was observed, with higher probiotic concentrations resulting in greater growth improvement. Notably, combined supplementation consistently produced the highest numerical enhancements in growth performance, indicating a synergistic interaction between the two probiotic consortia rather than a merely additive effect. This is consistent with previous studies, where the addition of Streptomyces strains at 1% concentration to the feed improved growth parameters in Penaeus monodon after 15 days.76 Additionally, Streptomyces fradiae and Bacillus megaterium at 109 CFU/g of feed improved the weight of P. monodon after 60 days.63 Supplementing the feed with Streptomyces strains (1.0 × 108 CFU/g) alone or together with Bacillus strains (1.0 × 106 CFU/g) improved the growth and survival of Litopenaeus vannamei.52 The observed growth enhancement is likely associated with extracellular digestive enzymes produced by probiotic strains in the shrimp gastrointestinal tract, including protease, amylase, and lipase, which facilitate nutrient breakdown and absorption.79 Both Streptomyces and Bacillus species synthesize a broad range of exoenzymes such as protease, amylase, cellulase, and lipase, thereby improving feed utilization efficiency and digestive capacity.77,78,80 Furthermore, Bacillus strains act as a source of amino acids, vitamins, and macro- and micronutrients, contributing further to shrimp health.77 The synergistic growth-promoting effect observed in combined treatments may arise from complementary enzyme production profiles and expanded substrate degradation capacity, leading to more efficient nutrient utilization than that achieved by individual probiotic strains. In a previous study, Streptomyces sp. strains used in the present research demonstrated strong exoenzyme production, including strains TM1, TM2, TM7, TM21, and TM22, along with pronounced antagonistic activity against several Vibrio species and the production of protease, amylase, and cellulase.33 The multifunctional enzyme production and antimicrobial potential of these strains likely contributed to the enhanced growth performance of whiteleg shrimp observed in this study.
In summary, the results of this research demonstrated that, regardless of the varying densities of individual Bacillus and Streptomyces consortia in the feed, they effectively controlled TVC, enhanced growth, and increased THC in response to pathogenic Vibrio infection. Nonetheless, the pairing of Streptomyces and Bacillus proved to be one of the most promising alternatives. The Streptomyces consortium outperformed the Bacillus consortium in reducing TVC in both the rearing water and shrimp digestive tract, with higher supplementation densities leading to a more significant decrease in TVC. In contrast, the Bacillus consortium was more effective at lowering TAN and nitrite concentrations in the rearing water. Furthermore, when Streptomyces and Bacillus were combined at high densities (108 CFU/g feed), the best shrimp growth, as well as the most effective control of Vibrio in the rearing water and digestive tract, along with the best control of TAN and nitrite, were observed. Therefore, the combination of Streptomyces and Bacillus offers functional complementarity, providing comprehensive water quality control and promoting more effective growth of whiteleg shrimp. The combined effect of Streptomyces and Bacillus strains aligns with previous research, which suggests that probiotics containing multiple strains or species can be more effective than those with a single strain.35,52,81
This study showed that the combination of Streptomyces strains (TM1, TM2, TM7, TM21, and TM22) and Bacillus strains (AQ1, BIO2, and BAL3) supplemented into feed at a density of 108 CFU/g can effectively improve growth parameters (FCR, FBW, DWG), survival rate, and total hemocyte count (THC), helping shrimp resist Vibrio parahaemolyticus infection. It also controls TVC, TAN, and nitrite concentrations in the culture water, as well as TVC in the shrimp digestive system. This combination can be considered as a supplementary feed for P. vannamei postlarvae at a density of 100 shrimp/100 L in a recirculating aquaculture system.
Additional file: Figure S1-S12.
ACKNOWLEDGMENTS
The authors would like to thank Lien Hiep Phat Science Technology Company Limited for providing necessary facilities to carry out the research work. The authors also acknowledge the support of time and facilities from Ho Chi Minh City University of Technology (HCMUT), VNU-HCM, for this study.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
TDP conceived and designed the experiments. TDP, BTHL, PTTD, HTTL, and NTNH carried out the experiments. TDP analyzed the data. PMT performed data validation. TDP wrote the manuscript. NTH guided the study. PMT and NTH supervised the study. All authors read and approved the final manuscript for publication.
FUNDING
This research was funded by Ho Chi Minh City University of Technology, Vietnam National University–Ho Chi Minh City (VNU-HCM).
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
Not applicable.
- Martinell DP, Vergara-Solana FJ, Padilla MEA, Garza FA. An Introduction to Sustainable Aquaculture. 2024.
- Cressey D. Aquaculture: Future fish. Nature. 2009;458(7237):398-400.
Crossref - Colt J, Plesha P, Huguenin J. Impact of net positive suction head on the design and operation of seawater pumping systems for use in aquaculture. Aquac Eng. 2006;35(3):239-257.
Crossref - Ebeling JM, Timmons MB. Recirculating Aquaculture Systems. Aquaculture Production Systems. 2012:245-277.
- Du Y, Xu J, Zhou L, Chen F, Qiu T, Sun J. Retrofitting Sea Cucumber Nursery Tanks to Recirculating Aquaculture Systems for Highly Intensive Litopenaeus vannamei Aquaculture. Appl Sci. 2021;11(20):9478.
Crossref - Reid B, Arnold CR. The intensive culture of the penaeid shrimp Penaeus vannamei Boone in a recirculating raceway system. J World Aquac Soc. 1992;23(2):146-153.
Crossref - Davis DA, Arnold CR. The design, management and production of a recirculating raceway system for the production of marine shrimp. Aquac Eng. 1998;17(3):193-211.
Crossref - Yang JG, C, Song H, Liu X, Gu Z, Guo Y. Design and test of mass balance-based recirculating aquaculture system for higher place shrimp pond. Trans Chin Soc Agric Eng. 2017;33:217-222.
Crossref - Yang JN, Q, Zhang, Y, Xu, B. Construction technology on RAS for shrimp culture. Trans Chin Soc Agric Eng. 2010;26(8):136-140.
Crossref - Suantika GS, ML, Kurniawan JB, Pratiwi SA, et al. Development of a zero water discharge (ZWD)-Recirculating aquaculture system (RAS) hybrid system for super intensive white shrimp (Litopenaeus vannamei) culture under low salinity conditions and its industrial trial in commercial shrimp urban farming in Gresik, East Java, Indonesia. Aquac Eng 2018;82:12-24.
Crossref - Shi YHZ GY, Liu JZ, Zhu YZ, Xu JB. Performance of a constructed wetland in treating brackish wastewater from commercial recirculating and super-intensive shrimp growout systems. Bioresour Technol. 2011;102(20):9416-9424.
Crossref - Fleckenstein LJ, Tierney TW, Fisk JC, Ray AJ. Effects of supplemental LED lighting on water quality and Pacific white shrimp (Litopenaeus vannamei) performance in intensive recirculating systems. Aquaculture. 2019;504:219-226.
Crossref - Fleckenstein LJ, Tierney TW, Ray AJ. Comparing biofloc, clear-water, and hybrid recirculating nursery systems (Part II): Tilapia (Oreochromis niloticus) production and water quality dynamics. Aquacultural Engineering. 2018;82:80-85.
Crossref - Chen Z, Chang Z, Zhang L, et al. Effects of water recirculation rate on the microbial community and water quality in relation to the growth and survival of white shrimp (Litopenaeus vannamei). BMC Microbiol. 2019;19(1):192.
Crossref - Yazid SHM, Daud HM, Azmai MNA, Mohamad N, Mohd Nor N. Estimating the Economic Loss Due to Vibriosis in Net-Cage Cultured Asian Seabass (Lates calcarifer): Evidence From the East Coast of Peninsular Malaysia. Original Research. Front Vet Sci. 2021;8;644009.
Crossref - Morales V, Cuéllar-Anjel J. Technical Guide: Pathology and Immunology of Penaeid Shrimp [Guía Técnica: Patología e Inmunología de Camarones Penaeidos; in Spanish]. OIRSA; 2014:382
- Brock JA, Lightner DV. Chapter 3: Diseases of Crustacea. Diseases of Marine Animals. Biologische Anstalt Helgoland. 1990:245-424.
- Ishimaru KA-M, M, Muroga, K. Vibrio penaeicida sp. nov, a Pathogen of Kuruma Prawns (Penaeus japonicus). Int J Syst Bacteriol. 1995;45(1):134-138. doi: 10.1099/00207713-45-1-134
- Hoa TTT, Fagnon MS, Thy DTM, Chabrillat T, Trung NB, Kerros S. Growth Performance and Disease Resistance against Vibrio parahaemolyticus of Whiteleg Shrimp (Litopenaeus vannamei) Fed Essential Oil Blend (Phyto AquaBiotic). Animals (Basel). 2023;13(21):3320.
Crossref - Rahayu S, Amoah K, Huang Y, et al. Probiotics application in aquaculture: its potential effects, current status in China and future prospects. Front Marine Sci. 2024;11:1455905.
Crossref - Romano N, Koh C-B, Ng W-K. Dietary micro-encapsulated organic acids blend enhances growth, phosphorus utilization, immune response, hepatopancreatic integrity and resistance against Vibrio harveyi in white shrimp, Litopenaeus vannamei. Aquaculture. 2015;435:228-236.
Crossref - Lazado CC, Caipang CMA. Mucosal immunity and probiotics in fish. Fish Shellfish Immunol. 2014;39(1):78-89.
Crossref - Schar D, Klein EY, Laxminarayan R, Gilbert M, Van Boeckel TP. Global trends in antimicrobial use in aquaculture. Sci Rep. 2020;10(1):21878.
Crossref - Vinoj G, Vaseeharan B, Thomas S, Spiers AJ, Shanthi S. Quorum-quenching activity of the AHL-lactonase from Bacillus licheniformis DAHB1 inhibits Vibrio biofilm formation in vitro and reduces shrimp intestinal colonisation and mortality. Mar Biotechnol (NY). 2014;16(6):707-15.
Crossref - Ghanei-Motlagh R, Mohammadian T, Gharibi D, et al. Quorum Quenching Properties and Probiotic Potentials of Intestinal Associated Bacteria in Asian Sea Bass Lates calcarifer. Mar Drugs. 2019;18(1):23.
Crossref - An J, Zhu W, Liu Y, et al. Purification and characterization of a novel bacteriocin CAMT2 produced by Bacillus amyloliquefaciens isolated from marine fish Epinephelus areolatus. Food Control. 2015;51:278-282.
Crossref - Rusmana I, Isramilda I, Akhdiya A. Characteristics of anti-Vibrio harveyi compounds produced by Bacillus spp. isolated from shrimp ponds. Biodiversitas Journal of Biological Diversity. 2021;22(11):221120.
Crossref - Genilloud O. Actinomycetes: still a source of novel antibiotics. Nat Prod Rep. 2017;34(10):1203-1232.
Crossref - Behie SW, Bonet B, Zacharia VM, McClung DJ, Traxler MF. Molecules to Ecosystems: Actinomycete Natural Products In situ. Front Microbiol. 2016;7:2149.
Crossref - Hariharan S, Dharmaraj S. Selection of New Probiotics: The Case of Streptomyces. Therapeutic, Probiotic, and Unconventional Foods. 2018:27-54.
- Hernandez-Saldana OF, Barboza-Corona JE, Bideshi DK, Casados-Vazquez LE. New bacteriocin-like substances produced by Streptomyces species with activity against pathogens. Folia Microbiol (Praha). Aug 2020;65(4):669-678.
Crossref - Phuc TD, Huong NTT, Dan PTT, Phuong TTM. Isolation and Screening of Actinomycetes Against Vibrio spp. and Producing Extracellular Enzymes. Ho Chi Minh City University of Education Journal of Science. 2021;18(6):1016-1027.
Crossref - Phuc TD, Huong NT, Thong DH, Tung CV, Dan PTT, Phuong TTM. Investigation of Ability to Remove NH3 and NO2 By Combination of Yucca Schidigera Extract and Bacillus Strains. IOSR J Biotechnol Biochem. 2021;7(4):01-17.
Crossref - Kewcharoen W, Srisapoome P. Probiotic effects of Bacillus spp. from Pacific white shrimp (Litopenaeus vannamei) on water quality and shrimp growth, immune responses, and resistance to Vibrio parahaemolyticus (AHPND strains). Fish Shellfish Immunol. 2019;94:175-189.
Crossref - Phuc TD, Huong NT, Dan PTT, Linh BTH, Mui TV. Effect from Combinations of Yucca schidigera extract with Bacillus Strains on the Growth of White Leg Shrimp (Penaeus vannamei), Density of Vibrio sp, and on the Ammonia and Nitrite Content of Culture Water. J Pure Appl Microbiol. 2023;17(3):1444-1457.
Crossref - Luis-Villasenor IE, Macias-Rodriguez ME, Gomez-Gil B, Ascencio-Valle F, Campa-Cordova A. Beneficial effects of four Bacillus strains on the larval cultivation of Litopenaeus vannamei. Aquaculture. 2011;321(1):136-144.
Crossref - Abasolo-Pacheco F, Saucedo PE, Mazon-Suastegui JM, et al. Isolation and use of beneficial microbiota from the digestive tract of lions-paw scallop Nodipecten subnodosus and winged pearl oyster Pteria sterna in oyster aquaculture. Aquac Res. 2016;47(10):3042-3051.
Crossref - Gopalakrishnan S, Vadlamudi S, Bandikinda P, et al. Evaluation of Streptomyces strains isolated from herbal vermicompost for their plant growth-promotion traits in rice. Microbiol Res. 2014;169(1):40-48.
Crossref - Li L-m, Zheng T, Chen Y, et al. The antagonistic mechanisms of Streptomyces sioyaensis on the growth and metabolism of poplar canker pathogen Valsa sordida. Biological Control. 2020;151:104392.
Crossref - Phuc TD, Huong NT, Dan PTT, Linh BTH, Loan VTT, Hang NTB. Dual Role Of Streptomyces Strains: Reduction of Aeromonas Counts In Both Culture Water And The Intestine, And Growth Enhancement of Discus Fish (Symphysodon sp.). IOP Conf Ser Earth Environ Sci. 2024;1340(1):012016.
Crossref - Newaj-Fyzul A, Austin B. Probiotics, immunostimulants, plant products and oral vaccines, and their role as feed supplements in the control of bacterial fish diseases. J Fish Dis. 2015;38(11):937-955.
Crossref - Sha Y, Wang L, Liu M, Jiang K, Xin F, Wang B. Effects of lactic acid bacteria and the corresponding supernatant on the survival, growth performance, immune response and disease resistance of Litopenaeus vannamei. Aquaculture. 2016;452:28-36.
Crossref - Yang Q-h, Tan B-p, Dong X-h, Chi S-y, Liu H-y. Effects of different levels of Yucca schidigera extract on the growth and nonspecific immunity of Pacific white shrimp (Litopenaeus vannamei) and on culture water quality. Aquaculture. 2015;439:39-44.
Crossref - Abdel-Tawwab M, Khalil RH, Nour AM, Elkhayat BK, Khalifa E, Abdel-Latif HMR. Effects of Bacillus subtilis-fermented rice bran on water quality, performance, antioxidants/oxidants, and immunity biomarkers of White leg shrimp (Litopenaeus vannamei) reared at different salinities with zero water exchange. Journal of Applied Aquaculture. 2020;34(2):332-357.
Crossref - Novriadi R, Albasri H, Wahyudi AE, Fadhilah R, Ali A, Trullas C. Effects of the addition of oak (Quercus robur L.) and yucca (Yucca schidigera) on the water quality and growth performance of pacific white shrimp (Litopenaeus vannamei) cultured intensively in concrete tanks. J World Aquac Soc. 2022;53(5):984-994.
Crossref - Felix N, Sudharsan M. Effect of glycine betaine, a feed attractant affecting growth and feed conversion of juvenile freshwater prawn Macrobrachium rosenbergii. Aquac Nutr. 2004;10(3):193-197.
Crossref - APHA. Standard Methods for the Examination of Water and Wastewater (23rd ed.). Washington DC: American Public Health Association; 2017.
- Sookchaiyaporn N, Srisapoome P, Unajak S, Areechon N. Efficacy of Bacillus spp. isolated from Nile tilapia Oreochromis niloticus Linn. on its growth and immunity, and control of pathogenic bacteria. Fisheries Science. 2020;86(2):353-365.
Crossref - Chotikachinda R, Lapjatupon W, Chaisilapasung S, Sangsue D, Tantikitti C. Effect of inactive yeast cell wall on growth performance, survival rate and immune parameters in Pacific White Shrimp (Litopenaeus vannamei). Songklanakarin J Sci Technol. 2008;30(6):687-692.
- Owens L, O’Neill A. Use of a clinical cell flow cytometer for differential counts of prawn Penaeus monodon haemocytes. Dis Aquat Organ. 1997;31:147-153.
Crossref - Muralisankar T, Kalaivani P, Thangal SH, Santhanam P. Growth, biochemical, antioxidants, metabolic enzymes and hemocytes population of the shrimp Litopenaeus vannamei exposed to acidified seawater. Comp Biochem Physiol C Toxicol Pharmacol. Jan 2021;239:108843.
Crossref - Bernal MG, Marrero RM, Campa-Cordova AI, Mazon-Suastegui JM. Probiotic effect of Streptomyces strains alone or in combination with Bacillus and Lactobacillus in juveniles of the white shrimp Litopenaeus vannamei. Aquaculture International. 2016;25(2):927-939.
Crossref - Hamsah H, Widanarni W, Alimuddin A, Yuhana M, Junior MZ, Hidayatullah D. Immune response and resistance of Pacific white shrimp larvae administered probiotic, prebiotic, and synbiotic through the bio-encapsulation of Artemia sp. Aquac Int. 2019;27(2):567-580.
Crossref - Mustafa MF, Bunga M, Achmad M. Use of probiotics to fight bacterial populations of Vibrio sp. on vaname shrimp cultivation (Litopenaeus vannamei). Torani: JFMarSci. 2019;2(2):69-76.
Crossref - Nimrat S, Boonthai T, Vuthiphandchai V. Effects of probiotic forms, compositions of and mode of probiotic administration on rearing of Pacific white shrimp (Litopenaeus vannamei) larvae and postlarvae. Anim Feed Sci Technol. 2011;169(3):244-258.
Crossref - Proespraiwong P, Mavichak R, Imaizumi K, Hirono I, Unajak S. Evaluation of Bacillus spp. as Potent Probiotics with Reduction in AHPND-Related Mortality and Facilitating Growth Performance of Pacific White Shrimp (Litopenaeus vannamei) Farms. Microorganisms. 2023;11(9).
Crossref - Tan LT-H, Chan K-G, Lee L-H, Goh B-H. Streptomyces Bacteria as Potential Probiotics in Aquaculture. Mini Review. Front Microbiol. 2016;7L79.
Crossref - Miao L, Xu J, Yao Z, et al. The anti-quorum sensing activity and bioactive substance of a marine derived Streptomyces. Biotechnol Biotechnol Equip. 2017;31(5):1007-1015.
Crossref - Hayashi M, Unemoto T, Minami-Kakinuma S, Tanaka H, Omura S. The mode of action of nanaomycins D and A on a gram-negative marine bacterium Vibrio alginolyticus. J Antibiot (Tokyo). 1982;35(8):1078-85.
Crossref - Castillo UF, Strobel GA, Ford EJ, et al. Munumbicins, wide-spectrum antibiotics produced by Streptomyces NRRL 30562, endophytic on Kennedia nigriscans. Microbiology (Reading). 2002;148(Pt 9):2675-2685.
Crossref - Yang N, Sun C. The Inhibition and Resistance Mechanisms of Actinonin, Isolated from Marine Streptomyces sp. NHF165, against Vibrio anguillarum. Front Microbiol. 2016;7:1467.
Crossref - Rateb ME, Houssen WE, Harrison WT, et al. Diverse metabolic profiles of a Streptomyces strain isolated from a hyper-arid environment. J Nat Prod. 2011;74(9):1965-71.
Crossref - Aftabuddin SK, M Abul; Kader, M Abdul; Sikder, M Nurul Azim; Hakim, M Abdul. Use of Streptomyces fradiae and Bacillus megaterium as probiotics in the experimental culture of tiger shrimp Penaeus monodon (Crustacea, Penaeidae). AACL BIOFLUX. 2013;6(3):253-267.
- Thompson KD, Rodkhum C, Bunnoy A, et al. Addressing Nanovaccine Strategies for Tilapia. Vaccines. 2023;11(8):1356.
Crossref - He T, Zhang M, Ding C, et al. New insight into the nitrogen removal capacity and mechanism of Streptomyces mediolani EM-B2. Bioresour Technol. 2022;348:126819.
Crossref - Tiffert Y, Supra P, Wurm R, Wohlleben W, Wagner R, Reuther J. The Streptomyces coelicolor GlnR regulon: identification of new GlnR targets and evidence for a central role of GlnR in nitrogen metabolism in actinomycetes. Mol Microbiol. 2008;67(4):861-80.
Crossref - Amin R, Reuther J, Bera A, Wohlleben W, Mast Y. A novel GlnR target gene, nnaR, is involved in nitrate/nitrite assimilation in Streptomyces coelicolor. Microbiology (Reading). 2012;158(Pt 5):1172-1182.
Crossref - Fischer M, Alderson J, van Keulen G, White J, Sawers RG. The obligate aerobe Streptomyces coelicolor A3(2) synthesizes three active respiratory nitrate reductases. Microbiology. 2010;156(Pt 10):3166-3179.
Crossref - Ji PF, Yao CL, Wang ZY. Immune response and gene expression in shrimp (Litopenaeus vannamei) hemocytes and hepatopancreas against some pathogen-associated molecular patterns. Fish Shellfish Immunol. 2009;27(4):563-570.
Crossref - Zubaidah A, Yuhana M, Widanarni. Encapsulated Synbiotic Dietary Supplementation at Different Dosages to Prevent Vibriosis in White Shrimp, Litopenaeus vannamei. HAYATI J Biosci. 2015;22(4):163-168.
Crossref - NavinChandran M, Iyapparaj P, Moovendhan S, et al. Influence of probiotic bacterium Bacillus cereus isolated from the gut of wild shrimp Penaeus monodon in turn as a potent growth promoter and immune enhancer in P. monodon. Fish Shellfish Immunol. Jan 2014;36(1):38-45.
Crossref - Liu H, Li Z, Tan B, et al. Isolation of a putative probiotic strain S12 and its effect on growth performance, non-specific immunity and disease-resistance of white shrimp, Litopenaeus vannamei. Fish Shellfish Immunol. 2014;41(2):300-307.
Crossref - Li K, Zheng T, Tian Y, et al. Beneficial effects of Bacillus licheniformis on the intestinal microflora and immunity of the white shrimp, Litopenaeus vannamei. Biotechnol Lett. 2007;29(4):525-530.
Crossref - Daniel K, & Sari, P. D. W. Feeding management of vannamei shrimp (Litopenaeus vannamei) with immunostimulant addition. IOP Conf. Series: Earth and Environmental Science. 2023:
- Chiu CH, Guu YK, Liu CH, Pan TM, Cheng W. Immune responses and gene expression in white shrimp, Litopenaeus vannamei, induced by Lactobacillus plantarum. Fish Shellfish Immunol. 2007;23(2):364-77.
Crossref - Das S, Ward LR, Burke C. Screening of marine Streptomyces spp. for potential use as probiotics in aquaculture. Aquaculture. 2010;305(1-4):32-41.
Crossref - James G, Das BC, Jose S, Vattringal Jayadradhan RK. Bacillus as an aquaculture friendly microbe. Aquac Int. 2021;29(1):323-353.
Crossref - James G, Prasannan Geetha P, Thavarool Puthiyedathu S, Vattringal Jayadradhan RK. Applications of Actinobacteria in aquaculture: prospects and challenges. 3 Biotech. Feb 2023;13(2):42.
Crossref - Reda RM, Selim KM. Evaluation of Bacillus amyloliquefaciens on the growth performance, intestinal morphology, hematology and body composition of Nile tilapia, Oreochromis niloticus. Aquac Int. 2015;23(1):203-217.
Crossref - Jagannathan SV, Manemann EM, Rowe SE, Callender MC, Soto W. Marine Actinomycetes, New Sources of Biotechnological Products. Mar Drugs. 2021;19(7):365.
Crossref - Timmerman HM, Koning CJM, Mulder L, Rombouts FM, Beynen AC. Monostrain, multistrain and multispecies probiotics—A comparison of functionality and efficacy. Int J Food Microbiol. 2004;96(3):219-233.
Crossref
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.