ISSN: 0973-7510
E-ISSN: 2581-690X
Recovery of bioactive compounds from microalgal waste represents a sustainable approach for producing value-added nutraceuticals. In this study, natural anti-inflammatory and antioxidant compounds were extracted from the defatted biomass (DB) of ultrasonic-pretreated, municipal-wastewater-grown double mutant Neodesmus pupukensis DMA5 through submerged fermentation (SmF) using immobilized Bacillus stearothermophilus assisted by zinc oxide nanoparticles (ZnO-NPs). The extract was characterized by Fourier-transform infrared (FTIR) and evaluated for its in vitro anti-inflammatory and antioxidant activities, with corresponding IC50 values determined. Maximum total phenolic content of 61.37 ± 0.09 mgGAE/g DW and total flavonoid content of 57.77 ± 0.11 mgQE/g DW were obtained from the DB using immobilized B. stearothermophilus, surpassing those from conventional methanolic extraction. This enhancement correlated with elevated cellulase activity (82.43 ± 0.11 U/mL) observed during SmF. The extract exhibited potent anti-inflammatory and antioxidant activities, with IC50 values of 52.6 µg/mL (albumin denaturation), 57.5 µg/mL (proteinase inhibition), 50.0 µg/mL (lipoxygenase inhibition), 41.6 µg/mL (DPPH), and 46.8 µg/mL (ABTS). FTIR analysis confirmed the presence of hydroxyl, carbonyl, aromatic, and ether functional groups, indicating the enrichment of phenolic, flavonoid, and polysaccharide derivatives responsible for the observed bioactivities. Overall, SmF with immobilized B. stearothermophilus and ZnO-NPs supplementation offers an efficient, eco-friendly, and solvent-free strategy for recovering anti-inflammatory and antioxidant compounds from microalgal DB, highlighting their potential applications in nutraceutical and functional food formulations.
Anti-inflammatory, Antioxidants, Bacillus stearothermophilus, Defatted Biomass, Neodesmus pupukensis, Submerged Fermentation, Zinc Oxide Nanoparticles
In recent years, the rising awareness of the antagonistic effects related with synthetic anti-inflammatory drug and antioxidants has intensified the global demand for safe and effective natural alternatives.1,2 The broad utilization of bioactive compounds (BACs) is largely attributed to their diverse biological effects, such as enhancing immune defense, exerting anti-inflammatory activity, minimizing oxidative cell impairment, and dropping the danger of chronic non-communicable ailments.3,4 Traditionally, plants have served as a primary source of BACs including polyphenols, carotenes, xanthophylls, sterols, and vitamins as an anti-inflammatory drug and antioxidants.5 However, given the increasing global population and escalating demand for nutraceuticals and functional foods, higher plants-derived BACs alone may not be sufficient to meet future needs.6 This concern underscores the urgent need to explore new, natural anti-inflammatory drug and antioxidant sources that are both sustainable and bioactive.
Microalgae have arisen as highly capable candidates in this context because of their rich and diverse biochemical composition. They exhibit exceptional photosynthetic efficiency, rapid biomass accumulation, high biodiversity, and metabolic flexibility under optimized culture conditions.7 Despite their immense potential, many microalgal species remain unexplored, especially in terms of their antioxidant content and biological applications. Exploring new and underutilized microalgal strains could reveal novel antioxidants with enhanced functionality, paving the way for their integration into pharmaceutical and nutraceutical formulations.8 Therefore, there is a pressing need to investigate and optimize the usage of DB from novel microalgae for the sustainable production of natural anti-inflammatory and antioxidants. Certain microalgae, especially members of Chlorophyta and Eustigmatophyceae, have demonstrated antioxidant capacities that are comparable to or even surpass those of terrestrial plants, fruits and vegetables. For instance, antioxidant activity of Chlorella vulgaris was 621.83 ± 36.41 µmol/g DW, exceeding the 464.83 ± 25.96 µmol/g DW observed in lemon balm-Melissa officinalis.9,10
An emerging area of interest is the utilization of defatted microalgal biomass (DMB), which is typically considered a byproduct of biodiesel production. DMB offers an economically and environmentally sustainable substrate for the extraction of proteins, polysaccharides, and phenolic compounds.11 Valorizing this biomass not only enhances the overall sustainability of microalgal biorefineries but also facilitates the production of value-added BACs, thereby supporting the values of a biobased economy.11,12 However, comprehensive investigations into the utilization of DMB are still limited. Existing research has primarily focused on its application in animal and fish feed, biofertilizers, bioethanol production, and bioremediation.13-16 Further exploration of its potential, particularly in the context of antioxidant production, remains an underexplored yet promising research direction.
BACs from DMB can be extracted through both conventional and advanced extraction techniques. Traditional approaches rely on solvent extraction combined with heat or mechanical agitation. Polar solvents such as water are less effective in extracting non-polar compounds like lipids and carotenoids, resulting in lower yields.17 Despite their widespread use, these methods have notable limitations, including long extraction times, low selectivity and specificity, degradation of heat-sensitive compounds, and reduced product purity following purification. Additionally, they often require high pressure, significant energy input, large volumes of high-purity solvents, pose environmental and toxicity concerns, which hinder their scalability and suitability for food and nutraceutical applications.18-20
Hence, there is a growing need to efficiently extract BACs from plants via sustainable and eco-friendly techniques. In this regard, biological processes have emerged as promising alternatives that improve the extraction efficiency and functional applicability of BACs while maintaining environmental compatibility. Owing to their high selectivity, biological approaches provide several advantages, such as producing extracts of superior quality with enhanced bioactivity and reduced toxicity.21 Among these approaches, enzyme aided extraction facilitates the release of BACs from complex matrices under optimized conditions, improving overall process efficiency.22 Additionally, microbial fermentation serves as a versatile strategy wherein microorganisms convert organic substrates present in by-products into proteins, bioethanol, and phenolic compounds.23-25 Considering the escalating costs and technical challenges associated with conventional extraction methods, fermentation represents a cost-effective, scalable, and sustainable alternative.26
In recent years, the incorporation of nanoparticles into fermentation processes has attracted significant attention owing to their distinctive physicochemical and electrical properties at the nanoscale. Nanoparticles such as magnetic materials, carbon nanotubes, and metal oxides have shown great potential in enhancing microbial stability and enzymatic activity, thereby improving the efficiency of bioenergy generation and algal biomass bioconversion.27,28 However, to date, limited research has explored their application in the recovery of BACs from microalgal biomass through microbial fermentation. This gap highlights a promising direction for future research aimed at enhancing the extraction of BACs from sustainable algal resources.
Generally, extracts contain a diverse mixture of BACs or phytochemicals with diverse polarities, which makes their purification, identification, and characterization highly challenging. To obtain pure compounds, several separation techniques are often applied in combination. The purified compounds are then subjected to structural elucidation and biological activity assessment. Fourier-transform infrared (FTIR) spectroscopy is widely recognized as an effective analytical technique for detecting and identifying BACs within complex plant extracts, even when their exact composition is unknown. Each compound exhibits a unique FTIR spectrum that acts as a molecular “fingerprint”, allowing for precise identification by comparison with spectral libraries of known reference compounds. Sample preparation methods for FTIR vary according to the physical state of the sample sodium chloride plates are typically used for liquid samples, whereas potassium bromide (KBr) pellets are preferred for solid samples.29
To the best of our knowledge, this is the first report demonstrating the anti-inflammatory and antioxidant potential of extract obtained from DB of ultrasonically pretreated municipal-wastewater-grown double mutant Neodesmus pupukensis DMA5 through microbial fermentation. The main objectives of the present study were: (i) to evaluate the effect of ZnO-NP s on the extraction efficiency of total phenolic content (TPC) and total flavonoid content (TFC) by immobilized B. stearothermophilus from DB using batch SmF; (ii) to assess the in vitro anti-inflammatory and antioxidant potential of the microbial extract; and (iii) to characterize the microbial extract using FTIR spectroscopy.
Chemicals
Chemicals, nutrient agar media, and reagents were procured from standard chemical suppliers HiMedia Laboratories and SRL Pvt. Ltd. (Mumbai, India). All reagents were used without further purification unless otherwise specified. ZnO-NPs (Size ≤100 nm) was procured from AdNano Technologies Pvt. Ltd. (Karnataka, India). As per the manufacturer’s data, the crystalline nature of ZnO-NPs was confirmed by X-ray Diffraction analysis.
Development of double mutant microalga
A double mutant N. pupukensis DMA5 was generated through sequential mutagenesis using UV-C radiation (200-280 nm) for 15 min, followed by treatment with 1% (v/v) ethyl methanesulfonate for 60 min from a native freshwater green microalga, N. pupukensis DTAS7 (GenBank Accession No. PQ084675.1), which was isolated from the ancient temple pond located in Ayyangarkulam (12.78°N, 79.66°E latitude and longitude), Kanchipuram District, Tamil Nadu, India. The obtained double mutant N. pupukensis DMA5 was maintained in modified Chu-13 medium30 and subsequently used for biomass production using ultrasonic-pretreated municipal wastewater (UPMWW).
Preparation of UPMWW
To prepare UPMWW, raw MWW was collected from the main outlet of a wastewater treatment facility in Kanchipuram, Tamil Nadu, India. Subsequently, the MWW was transported to the laboratory and stored under refrigeration until analysis. Ultrasonic pretreatment of MWW was performed following a modified procedure of Dhandayuthapani et al.25 Approximately 100 mL of 75% MWW was taken in a stainless-steel beaker and the treatment was performed using an ultrasonicator equipped with a metal probe (1.2 cm). Sonication was applied for 20 min at 25 kHz with an energy density of 0.35 W/mL, while maintaining gentle stirring. The temperature during the operation was kept at 30 ± 2 °C. The ultrasonically pretreated municipal wastewater (UPMWW) was then utilized as a sole medium for production of biomass using the double mutant strain N. pupukensis DMA5.
Biomass production by batch cultivation
Batch cultivation of the double mutant N. pupukensis DMA5 was carried out in five-liter Erlenmeyer flasks containing 2 L of UPMWW. The flask was inoculated with 7% (v/v) of a fresh, actively growing double mutant N. pupukensis DMA5 and incubated at 30 °C with constant shaking at 150 rpm. 15 watts white fluorescent light at an intensity of 150 µmol photons m2/s was used for illumination, following a 10 hrs light and 14 h dark photoperiod. Cultivation was continued for 8 days. End of experiment, sample was withdrawn and the biomass was separated by centrifugation using a centrifuge (Remi, R-8C BL, Maharashtra, India) at 14,000 rpm for 15 min. The harvested biomass was then used for DB preparation.
Preparation of DB
The harvested biomass was thoroughly washed twice with deionized water and subsequently centrifuged to eliminate any remaining impurities. The cleaned biomass was dried in a hot-air oven (Remi RDHO-50, Maharashtra, India) at 60 °C until a constant weight was obtained. Lipids were then extracted from the dried biomass using the conventional Bligh and Dyer method.31 Following lipid extraction, residual solvent in the DB was evaporated by air-drying at room temperature. The resulting DB was used for further investigations.
Preparation of immobilized cells
Bacillus stearothermophilus (GenBank Accession Number: KT282130) was obtained from our own department, immobilized with Ca-alginate and used for fermentation of DB to extract antioxidant and anti-inflammatory compounds. Immobilized cells were prepared using a modified method of Srinivasulu et al.32 A 2% (w/v) sodium alginate slurry was autoclaved and mixed with washed selected microbial cells harvested from 100 mL of an active culture (107 cells/mL). The cell-alginate mixture was stirred to ensure uniform distribution and then dropped into a 0.25 mol/L CaCl2 using a sterile syringe with needle to form beads, which were cured at 4 °C for 1 hr. The beads were thoroughly washed with deionized water before being used for SmF. All steps were performed aseptically under a laminar flow hood.
Extraction by SmF
SmF was performed with 100 mL of hydrolysate obtained from 25 g/L of DB. The initial pH of the hydrolysate was adjusted to 7.0 and supplemented with 150 mg/L ZnO-NPs prior to sterilization at 121 °C for 15 min under 15 psi. The medium was then cooled to room temperature. SmF was initiated by inoculating the sterile medium with 50 g/L of freshly prepared immobilized cells of B. stearothermophilus. The cultures were incubated at 45 °C and 150 rpm for 48 hrs under aerobic conditions in an orbital shaker incubator. After fermentation, sample was collected and estimated the TPC and TFC. A 75% (v/v) methanol extract was used as the control.
Separation of extract from fermented broth
The clarified fermentation broth was subjected to triple extraction with ethyl acetate according to the procedure described by Xue et al.33 After fermentation, the immobilized beads were separated by filtration using Whatman No. 1 filter paper. The filtrate was then centrifuged at 9000 rpm for 15 min at 4 °C. The resulting supernatant was acidified to pH 2.5 with 1 N HCl and subsequently extracted three times with equal volumes of ethyl acetate using a separatory funnel. The ethyl acetate extract was concentrated under reduced pressure using a rotary evaporator (BUCHI R-210) to obtain the extract. The microbial extract (ME) was air-dried at room temperature and used for further investigation.
Estimation of TPC
The TPC of the ME was estimated using the colorimetric method.34 Approximately 200 µL of the ME was placed in a test tube and mixed with 1 mL of diluted Folin–Ciocalteu reagent (1:10, v/v). After incubation for 4 min, 800 µL of Na2CO3 solution was added. The mixture was then incubated in the dark at room temperature for 2 h and subsequently centrifuged at 6000 rpm for 10 min. The absorbance of the resulting supernatant was measured at 765 nm using a UV–Visible spectrophotometer (Systronics Model 2206, Gujarat, India). The optical density (OD) values of the samples were interpolated from a gallic acid calibration curve (0-500 mg/L) to determine TPC. Results were expressed as milligrams of gallic acid equivalents (mg GAE) per gram of dry weight (DW) of DB.
Estimation of TFC
The TFC of the ME was determined using the AlCl3 colorimetric method with quercetin as the standard.35 Quercetin stock solution (10 mg) was prepared in 80% methanol, and standard solutions ranging from 25-100 µg/mL were used to construct the calibration curve. For each reaction, 0.5 mL of the sample was mixed with 1.5 mL of 95% methanol, 0.1 mL of 10% AlCl3 solution, 0.1 mL of 1 M potassium acetate, and 2.8 mL of distilled water. The mixture was incubated at room temperature for 30 min to allow color development, after which the absorbance was measured at 415 nm using a UV-Visible spectrophotometer (Systronics Model 2206, Gujarat, India) against a reagent blank. The concentration of TFC was calculated from the standard curve (y = 0.0063x + 0.0086, R2 = 0.9993) and expressed as milligrams of quercetin equivalents (mgQE) per gram of DW of DB.
Evaluation of anti-inflammatory activity
In vitro anti-inflammatory activity of the ME was evaluated through inhibition of albumin denaturation,36 anti-proteinase,36 and anti-lipoxygenase assays.37 In this study, the ME of N. pupukensis DMA5 was prepared at concentrations of 100, 200, 300, 400, and 500 µg/mL in distilled water. Ibuprofen (IPF), a nonsteroidal anti-inflammatory drug, and distilled water were used as the positive and negative controls, respectively. The percentage inhibition for each assay was calculated using the following equation:
% Inhibition = [AoC – AoS / AoC] × 100
Where,
AoC: Absorbance of control, AoS: Absorbance of sample
Inhibition of albumin denaturation
The reaction mixture comprised 1.5 mL of the ME or ibuprofen at varying concentrations and 1.5 mL of 1% (w/v) bovine serum albumin (BSA) solution prepared in distilled water. The pH of the mixture was adjusted to 6.5 using 1 N HCl and incubated at 27 ± 1 °C for 15 min. The samples were subsequently heated at 70 °C for 10 min to induce protein denaturation, followed by cooling to room temperature. Absorbance was recorded at 660 nm using a UV–Visible spectrophotometer (Systronics Model 2206, Gujarat, India).
Anti-proteinase activity
The reaction mixture comprised 0.5 mL of the ME or ibuprofen prepared at varying concentrations and 0.5 mL of trypsin solution (0.06 mg/mL) prepared in 20 mM Tris-HCl buffer (pH 7.4). Subsequently, 1.0 mL of 0.8% (w/v) casein solution was added, and the mixture was incubated at 37 °C for 20 min. The reaction was terminated by adding 2.0 mL of 70% perchloric acid, followed by centrifugation at 3000 rpm for 10 min to remove the precipitated protein. The absorbance of the resulting supernatant was recorded at 210 nm using a UV-Visible spectrophotometer (Systronics Model 2206, Gujarat, India) with buffer serving as the blank.
Anti-lipoxygenase activity
The reaction mixture comprised 1.0 mL of the ME or ibuprofen at varying concentrations and 1.0 mL of lipoxygenase enzyme solution (20,000 U/mL) prepared in 0.1 M borate buffer (pH 9.0). The mixture was incubated at room temperature for 5 min, and the reaction was then initiated by adding 1.0 mL of linoleic acid substrate solution (10 mM in borate buffer, pH 9.0). The increase in absorbance, corresponding to the formation of conjugated dienes, was recorded at 234 nm using a UV–Visible spectrophotometer (Systronics Model 2206, Gujarat, India).
Evaluation of antioxidant activity
The in vitro antioxidant activity of the ME was assessed using DPPH and ABTS radical scavenging assays.38,39 For the DPPH assay, various concentrations of the extract (50-300 µg/mL) were mixed with an equal volume of DPPH solution and incubated in the dark at 37 °C for 25 min. The absorbance of the reaction mixture was then measured at 517 nm using a UV-Visible spectrophotometer (Systronics Model 2206, Gujarat, India), while a DPPH solution without extract served as the control. For the ABTS assay, 5 µL of the extract (50-300 µg/mL) was added to 4 mL of the ABTS+• solution and kept in the dark at room temperature for 2 hrs. The absorbance was recorded at 734 nm using a UV-Visible spectrophotometer (Systronics Model 2206, Gujarat, India), with ascorbic acid used as the reference standard for both assays. The radical scavenging activity (%) was determined using the following equation:
Scavenging activity (%) = [AoC – AoS / AoC ] × 100
Half maximal inhibitory concentration determination
The half maximal inhibitory concentration (IC50) represents the concentration of ME essential to achieve 50% inhibition of DPPH and ABTS radicals, BSA denaturation, proteinase activity, and lipoxygenase activity. The IC50 values were calculated by plotting the percentage inhibition against varying extract concentrations and analyzing the trend using linear regression. Notably, a lower IC50 value reflects higher antioxidant and anti-inflammatory potential.
FTIR spectroscopy analysis
FTIR spectroscopy investigation of the ME of DB of N. pupukensis DMA5 was performed to find the key functional groups present in the bioactive constituents by the method of Sudhakar and Premalatha.40 Approximately 1.5 mg of extract was finely ground with 100 mg of spectroscopic-grade KBr and compressed into a transparent pellet under vacuum. Spectra were obtained in the wavenumber range of 4000-500 cm-1 at a 4 cm-1 resolution using an FTIR spectrometer (FT/IR-4700 Type A, JASCO, Tokyo, Japan), with a clean background used as the reference. Replicate spectra were collected to confirm reproducibility, and the obtained spectra were analyzed against reference data to identify functional groups.
Cellulase activity assay
Cellulase activity was assayed using the dinitrosalicylic acid (DNS) method with slight modifications.41 In this procedure, 0.5 mL of the crude fermented broth was combined with 0.5 mL of 1% (w/v) carboxymethyl cellulose prepared in 50 mM sodium citrate buffer (pH 4.8) and incubated at 50 °C for 30 min. The reaction was terminated by adding 1 mL of DNS reagent, and the mixture was subsequently boiled for 5 min to develop color. After cooling to room temperature, the absorbance was recorded at 540 nm using a UV-Visible spectrophotometer (Systronics Model 2206, Gujarat, India). One unit (U) of cellulase activity was defined as the amount of enzyme required to liberate 1 µmol of glucose equivalents per minute under the assay conditions.
Statistical analysis
All data are expressed as the mean ± standard deviation (SD) of three replicates. Statistical differences among treatments were evaluated using one-way analysis of variance (ANOVA) in MINITAB version 12, and significance was considered at p ≤ 0.05.
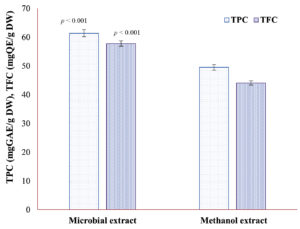
Maximum TPC and TFC of ME of DB of UPMWW-grown mutant N. pupukensis DMA5 were found to be 61.37 ± 0.09 mgGAE/g DW and 57.77 ± 0.11 mgQE/g DW, following SmF with immobilized B. stearothermophilus. Whereas, the TPC and TFC extracted from the DB using 75% (v/v) methanol were found to be 49.52 ± 0.11 mgGAE/g DW and 44.09 ± 0.07 mgQE/g DW, respectively (Figure 1). However, the obtained TPC and TFC values were lower than those achieved through microbial fermentation using immobilized B. stearothermophilus (p < 0.001). These findings suggest a substantial accumulation of phenolic and flavonoid compounds in the DB. In addition, the incorporation of ZnO-NPs during SmF significantly enhanced enzyme activity and phenolic release. The cellulase activity of immobilized B. stearothermophilus reached 82.43 ± 0.11 U/mL under ZnO-NPs supplemented conditions, whereas the unsupplemented control exhibited only 8.28 ± 0.11 U/mL. These results indicate that microbial extraction provided superior recovery of phenolic and flavonoid compounds compared to solvent-based extraction.
Figure 1. Total phenolic content and total flavonoid content of microbial and methanol extracts of defatted biomass of N. pupukensis DMA5
Inhibition effect of ME on albumin denaturation
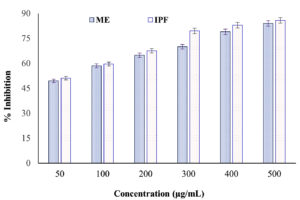
The in vitro anti-inflammatory activity of the ME obtained from N. pupukensis DMA5 was evaluated through the albumin denaturation inhibition assay, using IPF as the reference standard. As illustrated in Figure 2, the ME demonstrated a concentration-dependent enhancement in protein denaturation inhibition, increasing from 49.54% at 50 µg/mL to 84.03% at 500 µg/mL, whereas IPF showed inhibition values ranging from 51.05% to 85.88%. A paired t-test indicated a statistically significant difference between the two treatments (t = -4.25, p = 0.008), demonstrating that the standard drug produced marginally higher inhibition values than the ME. However, the difference in activity decreased notably at higher concentrations (400-500 µg/mL), where the ME achieved inhibition values comparable to IPF. This suggests that the BACs in the ME possess potent anti-inflammatory properties capable of stabilizing albumin against heat-induced denaturation.
Figure 2. Inhibitory activity of microbial extract of defatted biomass of N. pupukensis DMA5 on albumin denaturation
Anti-proteinase activity of ME
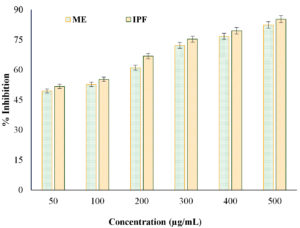
The in vitro anti-proteinase activity of the ME derived from N. pupukensis DMA5 was evaluated in comparison with IPF as the standard drug. The results presented in Figure 3, show that both the ME and IPF displayed concentration-dependent inhibition of proteinase activity. The ME showed inhibition values extending from 49.51% at 50 µg/mL to 82.45% at 500 µg/mL, whereas IPF showed slightly higher inhibition, from 51.77% to 85.34%. Statistical analysis by a paired t-test (t = -6.20, p = 0.0016) confirmed a significant variance (p < 0.01) among the ME and IPF, indicating that while both samples effectively inhibited proteinase activity, the standard drug remained marginally more potent. However, at higher concentrations (≥400 µg/mL), the inhibition levels of the ME approached those of IPF, suggesting strong dose-dependent anti-inflammatory efficacy.
Anti-lipoxygenase activity of ME
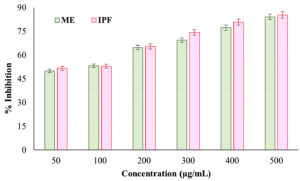
The anti-lipoxygenase activity of the ME from N. pupukensis DMA5 was compared with IPF as a standard reference to assess its potential in inhibiting enzyme-mediated inflammatory responses. As shown in Figure 4, both the ME and IPF demonstrated dose-dependent inhibition of lipoxygenase activity. The ME exhibited inhibition values ranging from 50.01% at 50 µg/mL to 84.09% at 500 µg/mL, whereas IPF exhibited inhibition extending from 51.66%-85.34% over the same concentration range. Statistical evaluation using a paired t-test revealed that the difference between ME and IPF was not significant (t = -2.50, p = 0.054), suggesting that the ME exhibits comparable lipoxygenase inhibitory potential to the standard drug. This result highlights the strong anti-inflammatory efficacy of the ME, particularly at higher concentrations (≥300 µg/mL), where its inhibitory performance closely matched that of ibuprofen (IPF).
The inhibitory concentration (LC50) values of the ME from N. pukensis DMA5 were determined using logistic regression analysis for albumin denaturation, proteinase inhibition, and lipoxygenase inhibition assays. The results revealed LC50 values of approximately 52.6 µg/mL, 57.5 µg/mL and 50.0 µg/mL, respectively, indicating strong and concentration-dependent anti-inflammatory potential. The LC50 values for albumin denaturation and proteinase inhibition were only slightly higher than those of the standard anti-inflammatory drug ibuprofen (IPF) (≈50 µg/mL), while the lipoxygenase inhibition value was nearly identical to that of ibuprofen (IPF), demonstrating the comparable potency of the N. pupukensis DMA5 extract. At higher concentrations (400-500 µg/mL), the inhibitory effects of the extract were almost equivalent to those of the standard drug across all assays, further supporting its potential as a natural anti-inflammatory agent.
Antioxidant activity of ME
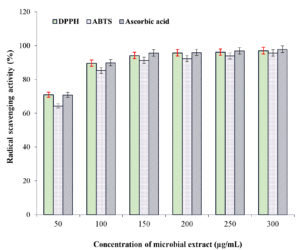
The in vitro antioxidant potential of ME of DB of N. pupukensis DMA5 was evaluated using DPPH and ABTS assays across concentrations of 50-300 µg/mL (Figure 5) and compared with ascorbic acid as the standard antioxidant. A progressive, concentration-dependent enhancement in radical scavenging activity was observed in both assays. For the DPPH assay, scavenging activity increased from 70.97% at 50 µg/mL to 97.04% at 300 µg/mL. Likewise, the ABTS assay showed an increase in radical scavenging activity from 64.32%-95.71% at 50 and 300 µg/mL, respectively. However, DPPH assay consistently showed slightly higher values than ABTS. At concentrations ranging from 150-300 µg/mL, the ME exhibited strong antioxidant performance, with DPPH scavenging values ranging from approximately 94%-98% and ABTS values between 92%-97%. Over the same concentration range, ascorbic acid also showed consistently high inhibition levels of about 96%-98%. At these elevated concentrations, the antioxidant activity of the ME closely matched that of the standard, with only a minimal difference of around 1%-3%, indicating comparable radical scavenging efficiency. The IC50 values determined through regression analysis were 41.6 µg/mL for DPPH and 46.8 µg/mL for ABTS, indicating a strong antioxidant potential. These findings support the potential of ME derived from DB of N. pupukensis DMA5 as natural antioxidants.
FTIR characterization of ME
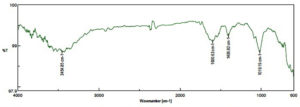
The FTIR spectrum of the ME derived from DB of N. pupukensis DMA5 revealed four prominent absorption peaks, indicating the occurrence of diverse functional groups related with bioactive metabolites (Figure 6). An intense and broad absorption band appearing at 3454.85 cm-1 corresponded to O-H stretching vibrations of hydroxyl groups, indicative of the presence of phenolic and flavonoid compounds. Another strong peak appeared at 1600.53 cm-1, attributed to C=O stretching of conjugated carbonyl or amide groups, suggesting the presence of proteins, esters, and polyphenolic compounds generated during microbial fermentation.
Figure 6. Functional group characterization of microbial extract of defatted biomass of N. pupukensis DMA5 by FTIR spectroscopy analysis
Furthermore, the absorption peak observed at 1468.22 cm-1 was attributed to C=C stretching vibrations of aromatic rings, signifying the presence of aromatic structures typically associated with phenolic and flavonoid compounds. The absorption band observed at 1019.19 cm-1 corresponded to C-O-C stretching vibrations of alcohols, esters, or polysaccharides, indicating the presence of glycosidic linkages and carbohydrate components in the extract. These functional group characteristics confirm that the ME of N. pupukensis DMA5 is rich in hydroxyl, carbonyl, aromatic, and ether compounds, typical of antioxidant and anti-inflammatory metabolites.
The high TPC and TFC values observed in N. pupukensis DMA5 indicate that its DB represents a sustainable and efficient source of BACs with considerable medicinal potential. The ME exhibited notably higher phenolic and flavonoid levels compared to the methanolic extract, yielding approximately 1.24 fold higher TPC and 1.31 fold higher TFC, respectively. This enhancement can be accredited to the enzymatic degradation of cell wall of microalgal by B. stearothermophilus, a thermophilic bacterium that secretes thermostable hydrolytic enzymes such as cellulases, which effectively break down cell wall polymers and release bound phenolics and flavonoids inaccessible to solvent extraction.
In contrast, conventional methanolic extraction primarily depends on solvent diffusion and solubility, limiting its efficiency in accessing tightly bound intracellular phenolic compounds. The ME approach is not only more effective but also environmentally sustainable, as it eliminates the use of organic solvents. Similar enzymatic enhancement of phenolic recovery was reported by Sivakumar and Dhandayuthapani,11 who demonstrated that thermophilic Bacillus species improved phenolic extraction from microalgal biomass through enzymatic hydrolysis. The inclusion of ZnO-NPs further improved fermentation efficiency by stabilizing enzyme conformation and enhancing catalytic activity through Zn2+ ion mediation, which promotes cellulolytic enzyme function and facilitates greater hydrolysis of polysaccharides and phenolic release.42,43 The highest TPC recorded in this study was about 4.5 fold greater than that reported for the mutant Pseudochlorella pringsheimii EMM2 produced by SmF.11
When compared with previous studies on solvent-based extraction, such as Eriobotrya japonica leaf extract containing 47.99 mgGAE/100 g DW of TPC and 4.78 mgQE/100 g DW of TFC, Chlorella sp. E53 (35.5 ± 0.14 mgGAE/g DW)41 and Scenedesmus sp. ME02 (5.40 ± 0.28 mgGAE/g DW), N. pupukensis DMA5 exhibited substantially higher phenolic and flavonoid yields.44-46 Overall, the ZnO-NPs assisted microbial fermentation process using immobilized B. stearothermophilus significantly improved the extraction efficiency of phenolic and flavonoid compounds compared to conventional solvent extraction. This finding reinforces the potential of microbial-assisted fermentation as a greener, enzyme-driven, and highly effective approach for the recovery of BACs from microalgal biomass, contributing to the development of sustainable natural antioxidant sources for pharmaceutical and nutraceutical applications.
The anti-inflammatory potential of the ME from N. pupukensis DMA5 is primarily attributed to its ability to prevent protein denaturation and inhibit key inflammatory enzymes through antioxidant stabilization and modulation of inflammatory mediators. The prevention of protein denaturation, a key step in inflammation, occurs by stabilization of protein conformation through hydrogen bonding and free radical scavenging.47 The strong inhibitory activities observed in protein denaturation, proteinase, and lipoxygenase assays are linked to the extract’s high phenolic and flavonoid contents, which suppress proteolytic enzyme activity and chelate metal ions involved in lipoxygenase catalysis.48 Microbial-assisted fermentation significantly enhanced this anti-inflammatory potential by enzymatically breaking down complex biomolecules and releasing low-molecular-weight phenolic derivatives with greater bioavailability and efficacy. Similar improvements have been reported in fermented C. vulgaris and Tetraselmis suecica, where microbial fermentation markedly increased antioxidant, anti-proteinase, and anti-lipoxygenase activities compared to non-fermented extracts.49 The LC50 analysis and the comparable inhibitory effects of the ME and IPF across the three inflammation-related assays-albumin denaturation, proteinase inhibition, and lipoxygenase inhibition-demonstrate that the extract possesses potent and concentration-dependent anti-inflammatory activity. The close similarity in efficacy between the ME and the standard drug confirms that microbial-assisted fermentation is an efficient strategy for enhancing the natural anti-inflammatory potential of microalgal biomolecules through the release of bioavailable phenolic and flavonoid compounds. Collectively, these findings establish the fermented N. pupukensis DMA5 extract as a sustainable, biocompatible, and biologically safe natural alternative to synthetic anti-inflammatory drugs, with significant potential for use in inflammation management and pharmaceutical applications.
The ME of DB of N. pupukensis DMA5 exhibited strong, concentration-dependent antioxidant activity as confirmed by DPPH and ABTS radical scavenging assays. The near equivalence to ascorbic acid at higher concentrations underscores its suitability for possible nutraceutical, functional food, or pharmaceutical applications. The low IC50 values of 41.6 µg/mL (DPPH) and 46.8 µg/mL (ABTS) reflect high radical scavenging efficiency, indicating the presence of potent antioxidant constituents comparable to those reported for microalgal phenolic extracts.50 The slightly higher activity in the DPPH assay suggests a greater hydrogen-donating ability of the phenolic compounds, consistent with earlier observations on algal polyphenols.51 The high antioxidant potential is attributed to the hydroxyl groups in phenolics that facilitate radical neutralization and prevent oxidative damage. Similar enhancements in antioxidant activity have been observed in fermented microalgal biomass, where fermentation improves phenolic extractability and efficacy.11,52 Overall, these results affirm that TPC and TFC of N. pupukensis DMA5 serve as effective natural antioxidants with promising applications in nutraceutical and functional food formulations.53
The FTIR analysis of the ME from N. pupukensis DMA5 confirmed the biochemical transformations induced by microbial-assisted fermentation, enhancing the release of bioactive metabolites rich in hydroxyl, carbonyl, aromatic, and ether groups. The presence of broad O-H and C=O bands indicate abundant phenolic and flavonoid compounds responsible for antioxidant and anti-inflammatory activities through hydrogen donation and protein stabilization. The C=C and C-O-C stretching bands further suggest the formation of aromatic and glycosidic compounds resulting from enzymatic hydrolysis during fermentation, which improves bioavailability and functional diversity.54 Similar findings in other fermented microalgal extracts have shown enhanced phenolic and polysaccharide content contributing to bioactivity.53 Overall, these findings confirm that microbial-assisted fermentation offers an efficient and sustainable approach to improving the biochemical profile and therapeutic potential of N. pupukensis DMA5 extracts for nutraceutical and pharmaceutical applications.
This study demonstrated that ZnO NPs assisted microbial fermentation employing immobilized B. stearothermophilus serves as an efficient, eco-friendly, and sustainable approach for enhancing the extraction of phenolic and flavonoid compounds from the DB of N. pupukensis DMA5. The process valorizes DB, a biofuel byproduct, into a rich source of BACs with strong anti-inflammatory and antioxidant potential. ZnO-NPs synergistically enhanced microbial enzymatic hydrolysis, yielding higher TPC and TFC than conventional extraction, while FTIR confirmed the presence of key functional groups linked to these bioactivities. By integrating nanocatalyst-assisted microbial biocatalysis, this study establishes a novel, green platform for sustainable recovery of high-value compounds, promoting zero-waste biorefinery and advancing the circular bioeconomy.
ACKNOWLEDGMENTS
The authors gratefully acknowledge the Principal and the Head of the Department, Department of Botany, and the authorities of Thiruvalluvar University for their support and for providing the facilities necessary to carry out this work.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
KD conceptualized the study. NT applied methodology. KD performed project administration and supervision. NT and KD performed formal analysis, Investigation and data curation. NT performed visualization and validation. NT wrote the original draft. KD wrote, reviewed and revised the manuscript. Both authors read and approved the final manuscript for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This article does not contain any studies on human participants or animals performed by any of the authors.
- Manful CF, Fordjour E, Ikumoinein E, Abbey L, Thomas R. Therapeutic strategies targeting oxidative stress and inflammation: a narrative review. BioChem. 2025;5(4):35.
Crossref - Parveen B, Rajinikanth V, Narayanan M. Natural plant antioxidants for food preservation and emerging trends in nutraceutical applications. Disc Appl Sci. 2025;7(8):845.
Crossref - Silva LBAR, Pinheiro-Castro N, Novaes GM, de Freitas Laiber Pascoal G, Ong TP. Bioactive food compounds, epigenetics and chronic disease prevention: focus on early-life interventions with polyphenols. Food Res Int. 2019;125:108646.
Crossref - Alongi M, Anese M. Re-thinking functional food development through a holistic approach. J Funct Foods. 2021;81:104466.
Crossref - Xu D-P, Li Y, Meng X, et al. Natural antioxidants in foods and medicinal plants: extraction, assessment and resources. Int J Mol Sci. 2017;18(1):96.
Crossref - Pereira AG, Echave J, Jorge AOS, et al. Therapeutic and preventive potential of plant-derived antioxidant nutraceuticals. Foods. 2025;14(10):1749.
Crossref - Coulombier N, Jauffrais T, Lebouvier N. Antioxidant compounds from microalgae: a review. Mar Drugs. 2021;19(10):549.
Crossref - Ferdous UT, Nurdin A, Ismail S, Shaari K, Yusof ZNB. A comparative study on antioxidant properties, total phenolics, total flavonoid contents, and cytotoxic properties of marine green microalgae and diatoms. J Gen Eng Biotech. 2025;23(1): 100456.
Crossref - Sikiru AB, Arangasamy A, Alemede IC, Egena SSA, Bhatta R, Rao SBN. In vitro evaluation of antioxidant properties of Chlorella vulgaris and its derivatives for use as antioxidant supplements in animal production. Indian J Anim Sci. 2024;94(1):88-91.
Crossref - Fernandes RPP, Trindade MA, Tonin FG, et al. Evaluation of antioxidant capacity of 13 plant extracts by three different methods: cluster analyses applied for selection of the natural extracts with higher antioxidant capacity to replace synthetic antioxidant in lamb burgers. J Food Sci Technol. 2016;53(1):451-460.
Crossref - Sivakumar N, Dhandayuthapani K. Total phenolic content and in vitro evaluation of antioxidant activity of microbial extract of defatted biomass of mutant Pseudochlorella pringsheimii EMM2. J Appl Nat Sci. 2025;17(1):31-38.
Crossref - Sander K, Murthy GS. Life cycle analysis of algae biodiesel. Int J Life Cycle Assess. 2010;15(7):704-714.
Crossref - Ahmad A, Hassan SW, Banat F. An overview of microalgae biomass as a sustainable aquaculture feed ingredient: food security and circular economy. Bioengineered. 2022;13(4):9521-9547.
Crossref - Nayak M, Swain DK, Sen R. Strategic valorization of de-oiled microalgal biomass waste as biofertilizer for sustainable and improved agriculture of rice (Oryza sativa L.) crop. Sci Total Environ. 2019;682:475-484.
Crossref - Fetyan N a. H, El-Sayed AEKB, Ibrahim FM, Attia YA, Sadik MW. Bioethanol production from defatted biomass of Nannochloropsis oculata microalgae grown under mixotrophic conditions. Environ Sci Pollut Res Int. 2022;29(2):2588-2597.
Crossref - da Fontoura JT, Rolim GS, Mella B, Farenzena M, Gutterres M. Defatted microalgal biomass as biosorbent for the removal of Acid Blue 161 dye from tannery effluent. J Environ Chem Eng. 2017;5(5):5076-5084.
Crossref - Wang T, Zhu L, Mei L, Kanda H. Extraction and separation of natural products from microalgae and other natural sources using liquefied dimethyl ether, a green solvent: a review. Foods. 2024;13(2):352.
Crossref - Gligor O, Mocan A, Moldovan C, Locatelli M, Crisan G, Ferreira ICFR. Enzyme-assisted extractions of polyphenols – a comprehensive review. Trends Food Sci Technol. 2019;88:302-315.
Crossref - Quiterio E, Grosso C, Ferraz R, Delerue-Matos C, Soares C. A critical comparison of the advanced extraction techniques applied to obtain health-promoting compounds from seaweeds. Mar Drugs. 2022;20(11):677.
Crossref - Hernandez Becerra E, De Jesus Perez Lopez E, Zartha Sossa J W. Recovery of biomolecules from agroindustry by solid–liquid enzyme-assisted extraction: a review. Food Anal Methods. 2021;14:1744-1777.
Crossref - Lemes AC, Egea MB, de Oliveira Filho JG, Gauterio G V, Ribeiro BD, Coelho MAZ. Biological approaches for extraction of bioactive compounds from agro-industrial by-products: a review. Front Bioeng Biotechnol. 2022;9:802543.
Crossref - Marathe SJ, Jadhav SB, Bankar SB, Kumari Dubey K, Singhal RS. Improvements in the extraction of bioactive compounds by enzymes. Curr Opin Food Sci. 2019;25:62-72.
Crossref - Sarumathi V, Dhandayuthapani K, Mazher S. Antioxidant activity and total phenolic content of defatted Scenedesmus quadricauda KDPSC2 biomass extract by microbial fermentation. J Drug Deliv Ther. 2019;9(4-s):58-263.
Crossref - Martinez-Espinosa R. Introductory chapter: A brief overview on fermentation and challenges for the next future. In: New Advances on Fermentation Processes. R M Martinez-Espinosa (Ed.). London, UK: IntechOpen. 2020.
Crossref - Dhandayuthapani K, Kumar PS, Chia WY, et al. Bioethanol from hydrolysate of ultrasonic proceessed robust microalgal biomass cultivated in dairy wastewater under optimal strategy. Energy. 2022;244:122604.
Crossref - Martínez-Espinosa RM. Introductory chapter: a brief overview on fermentation and challenges for the next future. In: Martínez-Espinosa RM, ed. New Advances on Fermentation Processes. London, UK: IntechOpen; 2020.
Crossref - Rambabu K, Bharath G, Banat F, Hai A, Show PL, Nguyen THP. Ferric oxide/date seed activated carbon nanocomposites mediated dark fermentation of date fruit wastes for enriched biohydrogen production. Int J Hydrog Energy. 2021;46(31):16631-16643.
Crossref - El-Sheekh M, Elshobary M, Abdullah E, Abdel-Basset R, Metwally M. Application of a novel biological-nanoparticle pretreatment to Oscillatoria acuminata biomass and coculture dark fermentation for improving hydrogen production. Microb Cell Fact. 2023;22(1):34.
Crossref - Sasidharan S, Chen Y, Saravanan D, Sundram KM, Yoga Latha L. Extraction, isolation and characterization of bioactive compounds from plants’ extracts. Afr J Tradit Complement Altern Med. 2011;8(1):1-10.
- Chu SP. The influence of the mineral composition of the medium on the growth of planktonic algae. Part I. Methods and culture media. J Ecol. 1942;30(2):284-325.
Crossref - Bligh EG, Dyer WJ. A rapid method of total lipid extraction and purification. Can J Biochem Physiol. 1959;37:911-917.
Crossref - Srinivasulu B, Adinarayana K, Ellaiah P. Investigations on neomycin production with immobilized cells of Streptomyces marinensis NUV-5 in calcium alginate matrix. AAPS Pharm Sci Tech. 2003;4(4):E57.
Crossref - Xue S, Jones AD, Sousa L, et al. Water-soluble phenolic compounds produced from extractive ammonia pretreatment exerted binary inhibitory effects on yeast fermentation using synthetic hydrolysate. PLoS One. 2018;13(3):e0194012.
Crossref - Singleton VL, Rossi JA. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic. 1965;16(3):144-158.
Crossref - Chang C-C, Yang M-H, Wen H-M, Chern J-C. Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J Food Drug Anal. 2002;10(3):178-182.
Crossref - Phukan K, Devi R, Chowdhury D. Green synthesis of gold nano-bioconjugates from onion peel extract and evaluation of their antioxidant, anti-inflammatory, and cytotoxic studies. ACS Omega. 2021;6(28):17811-17823.
Crossref - Eshwarappa RSB, Ramachandra YL, Subaramaihha SR, Subbaiah SGP, Austin RS, Dhananjaya BL. Anti-Lipoxygenase activity of leaf gall extracts of Terminalia chebula (Gaertn.) Retz. (Combretaceae). Pharmacogn Res. 2016;8(1):78.
Crossref - Qureshi MN, Kuchekar BS, Logade NA, Haleem MA. In-vitro antioxidant and in-vivo hepatoprotective activity of Leucas ciliata leaves. Rec Nat Prod. 2010;4(2):124-130.
- Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med. 1999;26(9-10):1231-1237.
Crossref - Sudhakar K, Premalatha M. Characterization of micro algal biomass through FTIR/TGA/CHN analysis: application to Scenedesmus sp. Ene Sou, Part A: Reco Utiliz and Enviro Eff.2015;37(21):2330-2337.
Crossref - Miller GL. Use of Dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem. 1959;31(3):426-428.
Crossref - Neha S, Manish S, Mishra PK, Ramteke PW. Application of ZnO nanoparticles for improving the thermal and pH stability of crude cellulase obtained from Aspergillus fumigatus AA001. Front Microbiol. 2016;7:514.
Crossref - Bautista-Hernández I, Aguilar CN, Martínez-Ávila GCG, et al. Phenolic compounds and antioxidant activity of Lippia graveolens Kunth residual leaves fermented by two filamentous fungal strains in solid-state process. Food Bioproducts Proceess. 2022;136:24-35.
Crossref - Pawlowska AM, Zurek N, Kapusta I, De Leo M, Braca A. Antioxidant and antiproliferative activities of phenolic extracts of Eriobotrya japonica (Thunb.) Lindl. fruits and leaves. Plants. 2023;12(18):3221.
Crossref - Choochote W, Suklampoo L, Ochaikul D. Evaluation of antioxidant capacities of green microalgae. J Appl Phycol. 2014;26(1):43-48.
Crossref - Bulut O, Akın D, Sönmez Ç, Öktem A, Yücel M, Öktem HA. Phenolic compounds, carotenoids and antioxidant capacities of a thermo-tolerant Scenedesmus sp. (Chlorophyta) extracted with different solvents. J Appl Phycol. 2019;31(3):1675-1683.
Crossref - Sakat SS, Juvekar AR, Gambhire MN. In vitro antioxidant and anti-inflammatory activity of methanol extract of Oxalis corniculata Linn. Int J Pharm Pharm Sci. 2010;2(1):146-155.
- Sabalingam S. In-vitro approaches to evaluate the anti-inflammatory potential of phytochemicals: A Review. J Drug Deliv Ther. 2025;15(1):187-192.
Crossref - Nor Shahril NS, Abu Bakar MH, Kassim MA, Karunakaran T, Shariff KA. The in-vitro antioxidant, anti-obesity, and anti-inflammatory properties of microalgae extracts of Tetraselmis suecica cultivated under sodium acetate and sodium nitrate supplementations. J Appl Biotechnol Rep. 2025;12(1):1554-1569.
Crossref - Mirzapour-Kouhdasht A, Garcia-Vaquero M, Huang JY. Algae-derived compounds: bioactivity, allergenicity and technologies enhancing their values. Bioresour Technol. 2024;406:130963.
Crossref - Godlewska K, Pacyga P, Najda A, Michalak I. Investigation of chemical constituents and antioxidant activity of biologically active plant-derived natural products. Molecules. 2023;28(14):5572.
Crossref - Garofalo C, Norici A, Mollo L, Osimani A, Aquilanti L. Fermentation of microalgal biomass for innovative food production. Microorganisms. 2022;10(10):2069.
Crossref - Shahidi F, Ambigaipalan P. Phenolics and polyphenolics in foods, beverages and spices: antioxidant activity and health effects – a review. J Funct Foods. 2015;18(part B):820-897.
Crossref - Kanimozhi NV, Sukumar M. Harnessing probiotic fermentation to enhance the bioavailability and health impact of dietary phytochemicals. Food Wellness. 2025;1(1):100018.
Crossref
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.