ISSN: 0973-7510
E-ISSN: 2581-690X
This manuscript reports a chemical investigation involving reverse-phase semipreparative HPLC-based isolation, followed by mass spectrometric and NMR-based characterization of three endophytic fungi recovered from Albizia lebbeck (L.) Benth. The study yielded two compounds (1-2) from ALE-75 (Sacharicola bicolor), three (3-5) from ALE-85 (Aspergillus pseudoglaucus), and one (6) from ALE-121 (Aspergillus eurotium). The extract from Aspergillus pseudoglaucus exhibited antioxidant potential (IC50 = 11.92 µg/mL in DPPH assay) and moderate cytotoxicity against lung (A549, IC50 = 35 µg/ml), colon (HCT-116, IC50 value of 45.4 µg/ml), and breast (T47D, IC50 value of 2.8 µg/ml) cancer cells, with growth inhibition of 89%, 77%, and 74% at 100 µg/ml. ALE-121 extract showed strong cytotoxicity against lung (A549, IC50 = 11.2 µg/ml), colon (HCT-116, IC50 = 12.5 µg/ml) and breast (T47D, IC50 = 15 µg/ml) cancer cells, with inhibition rates of 85%, 84%, and 75% at 100 µg/ml. Compound 6 (Austrocortinin) showed strong cytotoxicity against these cancer lines, with inhibition rates of 89%, 87%, and 67% at 10 µg/ml. Compound 1 was initially identified through the fermentation process of the endophytic fungus Coriolopsis sp. J5, which was isolated from the mangrove Ceriops tagal and compound 2 in Penicillium chrysogenum LF066 from the marine sponge Tethya aurantium. Compounds 3-5, originally found in Chaetomium globosum and Aspergillus flavus, whereas Compound 6 was initially discovered in the Australian Cortinarius toadstool. Although these compounds have been identified previously, this is the first instance of their reporting in the aforementioned endophytic fungi isolated from Albizia lebbeck.
Endophytic Fungi, Albizia lebbeck, Sacharicola bicolor, Aspergillus pseudoglaucus, Aspergillus eurotium
Endophytic fungi have gained significant attention as a promising source of unique pharmaceutical compounds with various uses in agriculture, medicine and industry.1 These microorganisms colonize plant tissues without causing harm and produce broad variety of secondary metabolites, such as steroids, polyketides, terpenoids, peptides, and alkaloids.2
Numerous biological effects, including antibacterial, antiviral, anticancer, antioxidant, and anti-inflammatory properties have been demonstrated by such compounds.3,4 Remarkably, certain endophytic fungi can generate bioactive compounds including vinblastine, camptothecin, podophyllotoxin, and paclitaxel that are identical to or analogous to those produced by their host plants.5,6 This ability has led to increased research on endophytes as a sustainable alternative for producing valuable plant-derived compounds. The siris plant, Albizia lebbeck, has applications in Indian ancient herbal medicine,7 has been studied for its antibiotic,8 antioxidant, liver-protective properties, anti-allergic, anti-asthmatic, and analgesic properties.9 Traditional medicine has utilized this herb to treat epilepsy, skin diseases, snake bites, detoxification of the blood, and diarrhea.10,11 Secondary metabolites with bioactive potential may be produced by endophytes linked to this plant.
As part of an investigation on fungal endophytes from medicinal plants,12-15 210 fungal strains were recovered from stem and leaf tissues sourced from A. lebbeck (Fabaceae) and underwent an analysis of diversity.16 Fungal strain Rosellinia sanctae-cruciana, isolated from this plant produced jammosporin A, a novel bioactive compound that exhibited anticancer efficacy in response to MOLT-4 which is blood cancer cell line.17 Such ability of plants to host endophytic fungi that generate bioactive compounds highlights their importance in traditional medicine and pharmaceutical research. It has been proven that endophytic fungus, especially Aspergillus species, are abundant sources of bioactive substances with an array of benefits in agriculture and medicine. Several studies have reported the recovery and characterization of bioactive compounds from Aspergillus strains, showcasing their abilities as antimicrobial, anticancer, antioxidant, and antiviral agents. Aspergillus sydowii and Aspergillus flavus, isolated from Ricinus communis and Psidium guajava, respectively, were found to produce extracts with antifungal, antibacterial, anticancer, and antioxidant properties.18 Similarly, Aspergillus fumigatus sourced from leaf tissues of Moringa oleifera exhibited antimicrobial, antibiofilm, antimutagenic, antiproliferative, and antioxidant activities. The chloroformic extract of A. fumigatus contained various phenolic compounds, including caffeic acid, rutin, ellagic acid, quercetin, and kaempferol.19 Further research into the isolation, characterization, and optimization of bioactive compound production from Aspergillus endophytes can result in the discovery of novel pharmaceutical drugs. Numerous bioactive compounds with significant antifungal activity have been shown to be produced by the endophytic fungus Saccharicola bicolor, which was isolated from Bergenia purpurascens.19 Through the application of the one strain-many compounds strategy, nine novel halogenated cyclopentenones, designated as bicolorins A-I, in addition with three previously described cyclopentenones, were characterized from this fungus.20 When tested against five phytopathogenic fungi, these compounds exhibited antifungal properties. Saccharicola bicolor’s bioactive compounds were discovered, highlighting the possibility of endophytic fungus as sources of new antifungal drugs. The isolation of bioactive molecules from Saccharicola bicolor has not been extensively documented. The aim was to isolate compounds from this species with the anticipation that it would yield novel and bioactive substances. Building on our previous work concerning the extraction of natural compounds from endophytes,15-17 we initiated a study focused on isolating secondary metabolites from three specific endophytic fungi: ALE-75 (Sacharicola bicolor), ALE-85 (Aspergillus pseudoglaucus), and ALE-121 (Aspergillus eurotium) isolated from Albizia lebbeck. The main goal of the investigation aimed to identify the compounds from these endophytic fungi and assess their cytotoxic and antioxidant properties.
Host plant isolation and sampling method
In September 2021, leaf specimens of Albizia lebbeck that showed no symptoms of disease were acquired from the Jammu region of J&K, India (32° 43′ 58″ N, 74° 48′ 32″ E). Under carefully monitored temperature conditions, these samples were brought to the lab in sterile containers. After that, the samples were processed with the objective to isolate the endophytic fungus. The collected plant sample was initially washed thoroughly under flowing tap water to get rid of any dirt and clinging soil particles. To eliminate microbial epiphytes on the surface, the sample underwent surface sterilization. The sterilization process involved rinsing with double-distilled water for two to three minutes after each treatment step, which included using 1% sodium hypochlorite (NaOCl) and a thirty-second exposure to 75% ethanol (C2H5OH).
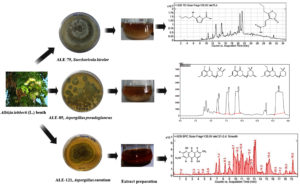
After sterilization, the plant sample was air-dried between sterile filter paper. After trimming the edges of the sterilized plant material with a sterile knife to expose the inner tissue, the sample was then divided into small pieces, approximately 5 mm each. These tissue pieces were placed in petri dishes containing three different types of media: Water Agar (WA), Yeast Malt Agar (YMA), and Potato Dextrose Agar (PDA). In each media streptomycin sulfate (0.2 mg/ml) was used as an antibiotic to inhibit bacterial growth. The medium supported the growth of the endophytic fungus as it emerged from the plant tissue. After being Parafilm-sealed, the petri dishes were kept at 28 °C in the dark. Due to the meticulous nature of some fungi, which can lead to overlapping growth if not monitored, observations commenced on the second day. Fresh PDA plates were utilized to collect the hyphal tips from the emerging fungal growth. Subculturing was repeated multiple times until a pure culture was obtained (Supplementary Figure S1, S2 and S3).
Molecular Characterization of Endophytic fungi
Both Internal Transcribed Sequence (ITS) analysis and morphological assessment were conducted to characterize the isolated endophytic fungi, as nucleotide-to-nucleotide BLAST query revealed ALE-75, as Sacharicola bicolor, ALE-85, as Aspergillus pseudoglaucus, and ALE-121, identified as Aspergillus eurotium as its closest match.
Large scale fermentation of Endophytic fungi
For one week, the fungus was grown on PDA media at a temperature of 28 °C. In a 500 mL Erlenmeyer flask, 200 mL of sterilized potato dextrose broth was inoculated with plugs taken from the agar. These cultures were kept as seed cultures for 48 hours at 28 °C in darkness, rotating at 150 rpm on a rotary shaker. Subsequently, thirty 1 L glass conical flasks, each containing 350 mL of Potato Dextrose Broth, were inoculated with 35 mL of the seed culture. These flasks were then incubated for 12 days under conditions identical to those used for the seed culture.
Extraction and Isolation
Extraction and isolation of secondary metabolites from Endophytic Fungus, ALE-75
Following a 12 day incubation period at 150 rpm and 28 °C in darkness on a rotary shaker, the culture was harvested upon incorporating 10% methanol solvent and subsequently homogenized. The homogenized culture underwent extraction with EtOAc (12 L) thrice. The organic extracts, EtOAc were concentrated in vacuo using a rotary evaporator to yield 1.5 g of yellow oil extract. The crude combined extract weighing 1 gram was subsequently dissolved in methanol and subjected to purification utilizing semi-preparative reversed-phase high-performance liquid chromatography (RP-HPLC) using column having dimension of 250 × 10 mm, particle size of 5 µm, and a flow rate of 3 mL/min. The isocratic solvent system employed consisted of 50% methanol and 50% water with 0.1% formic acid. Injection volume of 100 µL and 40 °C column temperature was maintained throughout the 37-minute run time. Two distinct peaks, labeled as 1 and 2, were individually isolated, resulting in the acquisition of previously identified furan derivatives, with yields of 30 mg and 12 mg, respectively (Supplementary Figure S4).
Extraction and isolation of secondary metabolites from Endophytic Fungus, ALE-85
Following a 12 day incubation period at 150 rpm and 28 °C in darkness on a rotatory shaker, the culture was harvested upon incorporating 10% methanol solvent and subsequently homogenized. The homogenized culture underwent extraction with EtOAc (12 L) and CH2Cl2 (6 L) thrice. The organic extracts, EtOAc and CH2Cl2, were concentrated in vacuo using a rotary evaporator to yield 1.5 g and 1 g of brown oil extract, respectively. In light of the comparable peak profiles observed in the HPLC chromatograms of the two extracts, they were amalgamated to yield 2.5 g of extract. The crude combined extract, weighing 1 g, was dissolved in methanol and subjected to purification through semi-preparative reversed-phase high-performance liquid chromatography (RP-HPLC) (250 × 10 mm, 5 µm, 3 mL/min). This process employed an isocratic solvent system composed of methanol (45%) and water (55% with 0.1% formic acid). Throughout the 20-minute procedure, 100 µL the injection volume and 40 °C column temperature was maintained. Three distinct peaks, labeled as 1-3, were individually isolated, resulting in the acquisition of previously identified asperentin derivatives, with yields of 14 mg, 25 mg and 32 mg, respectively (Supplementary
Figure 5).
Extraction and isolation of secondary metabolites from Endophytic Fungus, ALE-121
Following a growth period of 10 days in an orbital shaker at 150 rpm and 28 °C under dark conditions, the culture was harvested by the addition of 10% methanol and subsequently homogenized. The homogenized culture underwent extraction with EtOAc (12 L) thrice. The organic extracts, EtOAc were concentrated in vacuo using a rotary evaporator to yield 2 g of orange colour extract. Isolation of secondary metabolite was done using glass column chromatography. The ethyl acetate (EtOAc) extract was processed with open glass column chromatography utilizing silica gel of 60-120 mesh size. A minimal amount of silica gel was employed to adsorb the sample, which was subsequently positioned at the top of the column. Using a range of n-hexane and ethyl acetate combinations from 10:0 to 0:10, a gradient elution was carried out. About 10 mL of fractions were gathered in test tubes, and then evaporated using rotatory evaporator in order to identify the results using thin layer chromatography (TLC). A developing solvent system consisting of various ratios of n-hexane and ethyl acetate was used to apply each fraction to a TLC plate. Before being sprayed with a vanillin/sulphuric acid reagent, the produced TLC plates were inspected under ultraviolet light at short 254 nm and long 365 nm wavelengths. Using Sephadex LH20 (GE Healthcare Bioscience, Sweden), fractions from silica gel chromatography were mixed according to their TLC patterns and then subjected through size exclusion chromatography. Sephadex LH20 was soaked in chloroform for the entire night before the column was packed, and chloroform was used for the elution. One compound has been isolated and characterized, identified as the previously reported anthraquinone, with a yield of 38 mg (Supplementary Figure S6).
Anti-oxidant assay (DPPH)
The potential of the extract and compounds to sequester free radicals was evaluated using 1,1-diphenyl-2-picryl-hydrazyl (DPPH) within a 96-well plate. The sample was prepared at 10 mg/ml (w/v) in DMSO, and DPPH was 0.13 mM by dissolving 0.532 mg in 10 ml of methanol. To establish the IC50 value, the extract and compound were tested at concentrations ranging from 100 µg/mL to 3.125 µg/ml using serial dilution method. Ascorbic acid (10 mg/ml in DMSO) was used as the positive control, while 100 µL of MeOH and DPPH were used as the negative control. 100 µL of methanol was added to each well after the first one, which held 4 µl of the sample and 196 µL of methanol. Using a serial dilution method, values ranging between 100 µg/ml to 3.125 µg/ml was obtained. Following the addition of 100 µL DPPH, the mixture was mixed well, wrapped with foil, and then incubated for 30 minutes in incubator at 37 °C in the dark. At 517 nm, the absorbance was recorded employing an ELISA plate reader.
DPPH scavenging effect (%) = [(A0-A1/A0) × 100], where A0 denotes negative control absorbance whereas A1 denotes sample absorbance, was employed for determining antioxidant capacity. By plotting the percentage of scavenging activity against the sample concentration, the IC50 value was determined. In the linear equation y = mx + c, where x represents the IC50 value, 50 was substituted for y to ascertain the IC50, which is the concentration that exhibits 50% scavenging activity. Each assay was conducted in triplicate, and the mean values along with standard deviations were calculated to summarize the results.
In vitro cytotoxicity assay
The SRB assay was used to evaluate cytotoxic activity. Seed culture of 100 µl was added on 96-well tissue culture plate and kept for 24 hours’ incubation. Then, 100 µl of extract in complete growth medium was added, followed by a 48 hour incubation in a carbon dioxide incubator. 50 µl of 50% trichloroacetic acid has been added to each well to terminate growth, and the plate was then held at 4 °C for an hour to anchor the cells on the well bottom of the plate. To remove residual trichloroacetic acid, the plate was rinsed thrice with flowing tap water and kept for drying. After 30 minutes, sulforhodamine B dye (0.4%, made in 1% acetic acid, 100 µl) was applied, again washed thrice with 1% acetic acid, and allowed to dry. The plate was shaken gently for ten minutes after the attached dye was dissolved in 100 milliliters of 0.01M Tris-HCl Buffer (pH 10.4). At 540 nm, OD was measured utilizing a ELISA plate reader. GraphPad Prism Software Version 5.0 was used to determine IC50 value.
By deducting the mean OD of the corresponding blank from the mean OD of the treated cell, cell viability was computed. The growth inhibition percentage was calculated by taking the growth without test material as 100% and then calculating the growth percentage in the presence of test material (extract/compound).
% of cell viability = (Absorbance of Treated cells – Absorbance of Blank) / (Absorbance of Control cells – Absorbance of Blank) × 100
% of Growth inhibition = (100 – % cell viability)
A total of 210 endophytic fungi were isolated, described, and their diversity examined from leaves and stems gathered from twelve distinct locations in Jammu and Kashmir.16 This current research expands on that work, focusing on the extraction of bioactive compounds from three endophytic fungi, ALE-75, ALE-85 and ALE-121, identified as Aspergillus pseudoglaucus, Saccharicola bicolor and Aspergillus eurotium, respectively. The extract underwent screening for two bioactive assays: a free radical scavenging assay using DPPH and an in vitro cytotoxic assay using SRB assay.
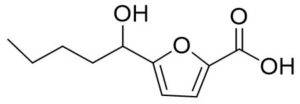
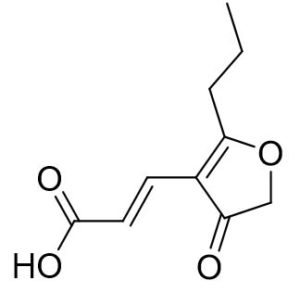
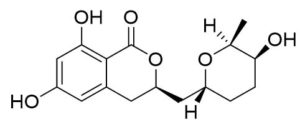
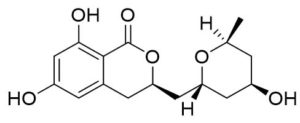
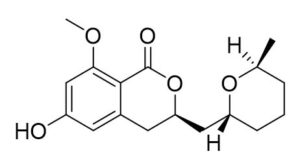
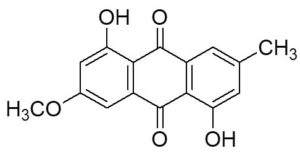
Two compounds were recovered and described from the endophytic fungus ALE-75. Through the application of 1-Dimensional and 2-Dimensional NMR spectroscopy, ESIMS measurements, and comparison with existing literature, the first compound was identified as a furan derivative, specifically 5-(1-hydroxypent-4-en-1-yl)-furan-2-carboxylic acid (1) (Supplementary Figure S7, S8, S9). This metabolite was first identified in the fermentation of solid rice by the endophytic fungal strain Coriolopsis sp. J5, whichwas obtained from the Ceriops tagal mangrove plant.21 The second compound was characterized as cillifuranone (2) (Supplementary Figure S10, S11, S12), a known compound previously reported from the endophytic fungi Penicillium chrysogenum strain LF066, isolated from the marine sponge Tethya aurantium.22 Additionally, three compounds were purified and described from the endophytic fungus ALE-85, identified as Aspergillus pseudoglaucus. The identification of these compounds was conducted using NMR spectroscopy, including both 1D and 2D techniques, as well as ESIMS measurements. The results were compared to previous research, specifically identifying them as 5’-hydroxyasperentin23-27 (3) (Supplementary Figure S13, S14 and S15)., 4-hydroxyasperentin23,24 (4) (Supplementary Figure S16, S17, S18)., and 8-methyl ether asperentin23-25 (5) (Supplementary Figure S19, S20, S21). These metabolites have been previously isolated from an entomogenous strain of Aspergillus flavus. Furthermore, one compound was extracted and identified from the endophytic fungus ALE-121, characterized as Aspergillus eurotium. Through the precise application of ESIMS measurements, coupled with the rigorous analysis of 1D and 2D NMR spectroscopy, and a meticulous comparison with established research, it has been conclusively determined that this compound is an anthraquinone derivative. Specifically, it is identified as 1,4-dihydroxy-2-methoxy-7-methyl-9,10-anthraquinone (6) (Supplementary Figure S22, S23, S24) commonly called austrocortinin. This metabolite was initially discovered in the fruiting bodies of a red Australian mushroom belonging to the Cortinarius genus28 and was subsequently found in the genera Alternaria29 and Dermocybe30 (Details including Mass, and name of all the compounds are listed in Table, whereas structure of compounds are shown in Figure).
Table:
Details of compounds isolated from endophytic fungi associated with Albizia lebbeck (L.) benth including their chemical structure, source of isolation and mass spectral data
In the DPPH assay, the extract from the endophytic fungus Saccharicola bicolor (ALE-75) displayed 34.84% antioxidant potential at 100 µg/ml. Conversely, the pure compounds showed no activity. In the cytotoxicity assay, the extract demonstrated cytotoxicity levels of 25%, 25%, and 50% against Lung (A549), Colon (HCT-116), and Breast (T47D) human cancerous cell lines respectively. However, pure compounds 1 and 2 showed no cytotoxicity against these cell lines, except for the lung cancer cell line (A549), where they exhibited cytotoxicity levels of 11% and 22% at 10 µg/ml, respectively. The bioactive potential assessment of the Aspergillus pseudoglaucus, ALE-85 extract revealed significant antioxidant properties, with an IC50 value of 11.92 µg/mL in the DPPH assay. Additionally, the extract demonstrated moderate anticancer effects against three human cancer cell lines: lung (A549, IC50 = 35 µg/ml), colon (HCT-116, IC50 = 45.4 µg/ml), and breast (T47D, IC50 = 2.8 µg/ml), with growth inhibition rates of 89%, 77%, and 74%, respectively, at 100 µg/ml. The endophytic fungus Aspergillus eurotium (ALE-121) extract exhibited a weak antioxidant potential of 5.41% at 100 µg/ml in anti-oxidant assay. Furthermore, the extract demonstrated moderate anticancer effects against three human cancer cell lines: lung (A549, IC50 = 11.2 µg/ml), colon (HCT-116, IC50 = 12.5 µg/ml), and breast (T47D, IC50 = 15 µg/ml), with growth inhibition rates of 85%, 84%, and 75%, respectively, at 100 µg/ml. Compound 6 exhibited strong cytotoxicity against lung (A549), colon (HCT-116), and breast (T47D) cancerous cell lines with growth inhibition rates of 89%, 87%, and 67%, respectively, at 10 µg/ml. These results demonstrate the potential of Aspergillus species as source of biologically active compounds with promising anti-cancer and antioxidant activities. The significant antioxidant activity of A. pseudoglaucus extract, coupled with its moderate anticancer effects, suggests it may be a valuable candidate for further investigation in drug discovery efforts. The differing results between extracts from A. pseudoglaucus and A. eurotium underscore the importance of conducting species-specific screenings when searching for new bioactive compounds from endophytic fungi. The strong cytotoxicity exhibited by Austrocortinin against multiple cancer cell lines warrants further exploration of its mechanism of action and potential as an anticancer agent. Several anthraquinonic pigments reported in the literature includes catenarin, emodin, erythroglaucin, rubrocristin, physcion, physcion-9-anthrone, questin, viocristin and isoviocristin isolated from Aspergillus cristatus mycelia. Among them, emodin, catenarin, viocristin, and isoviocristin have demonstrated antibacterial properties. In Bacillus brevis, these compounds disrupt macromolecular synthesis, whereas in Ehrlich ascites carcinoma cells, catenarin, emodin, and viocristin interfere with nucleotide incorporation. Additionally, catenarin and emodin inhibit DNA-dependent RNA polymerase in Escherichia coli in vitro.31 Catenarin, which is also isolated from Pyrenophora tritici-repentis (Died.) Drechsler demonstrated antibacterial activity.32 Catenarin and emodin also show antifungal activity against fungi linked to P. tritici-repentis, suggesting they play a role in the pathogen’s life strategy.33 Some anthraquinones, including emodin and diacerein, possess antidiabetic properties by improving glucose tolerance and insulin sensitivity and reducing inflammatory cytokine levels. Despite its diverse pharmacological effects, emodin can be toxic at high doses and has poor oral bioavailability in rats.34,35 Chrysophanol, another anthraquinone, protects renal cells from hypoxia/reoxygenation damage by modulating apoptosis, endoplasmic reticulum stress, and ferroptosis. It also shows promise in treating neurological damage associated with intracerebral hemorrhage and cerebral ischemia-reperfusion injuries.36 Research has shown that Chrysophanol effectively enhances neurobehavioral alterations, lowers the levels of PI3K, AKT, and mTOR proteins, influences markers of apoptosis, reduces pro-inflammatory cytokines such as IL-1β, IL-6, and TNF-α, and restores neurotransmitters such as GABA, glutamate, acetylcholine, and dopamine. It also improves oxidative stress markers, suggesting that CPH could be a promising therapy for neurological damage associated with intracerebral hemorrhage.37 The study suggests that chrysophanol may have therapeutic potential for treating cerebral ischemia-reperfusion injury by inhibiting mitochondrial autophagy and reducing hippocampal damage.38 Helminthosporin, anthraquinone isolated from EtOAc extract of Rumex abyssinicus Jacq, demonstrates potential as a brain-permeable dual cholinesterase inhibitor, warranting further exploration for Alzheimer’s disease treatment.39 Furthermore, the photoactive extracts of the genus Cortinarius, previously unreported to contain photoactive compounds, yielded austrocortinin and xanthorin for further investigation. Notably, austrocortinin had a singlet oxygen yield of 0.03, using [Ru(bpy3)Cl2] in d4-MeOH as a reference.40 These findings highlight the potential of anthraquinones as versatile compounds with applications in various fields of medicine and biotechnology. The diverse pharmacological effects of anthraquinones, including antibacterial, antifungal, antidiabetic, and neuroprotective properties, underscore their significance in drug discovery and development. Further research is needed to fully elucidate the mechanisms of action, optimize the dosing, and address the potential toxicity concerns associated with these compounds.
This study highlights a new source of isolation for these compounds by reporting their isolation from endophytic fungi isolated from Albizia lebbeck (L.) Benth, a traditional medicinal plant. The study’s significance lies in its novel approach to isolating these compounds from these endophytic fungi for the first time and evaluating their bioactive properties. This study adds important knowledge to the expanding body of knowledge regarding endophytic fungus and possible therapeutic potential. These findings highlights that these endophytic fungi could be a promising repertoire of naturally occurring compounds for developing antioxidant and anticancer therapies. Furthermore, this study highlights the importance of exploring endophytes as a rich repertoire of bioactive secondary metabolites, potentially leads to uncovering new therapeutic compounds for various diseases, including cancer. The research also underscores the value of interdisciplinary approaches, combining mycology, analytical chemistry, and pharmacology to unlock the therapeutic potential of bioactive compounds derived from endophytes. It represents a significant advancement in the field of endophytic fungi research, offering new perspectives on the potential of these organisms as sources of pharmaceutically relevant compounds. In addition to demonstrating the anticancer and antioxidant potential of the isolated metabolites, the study opens up new possibilities for drug development and discovery. This study underscores the untapped potential of endophytic fungi within the pharmaceutical industry by elucidating the diverse array of secondary metabolites produced by the endophytes. Moreover, the interdisciplinary nature of this research highlights the importance of collaborative efforts in scientific discovery. By combining expertise from mycology, analytical chemistry, and pharmacology, the study provides a comprehensive approach to understanding and harnessing the therapeutic potential of fungal metabolites. This holistic methodology not only enhances the quality and depth of the findings but also sets a precedent for future research in the field. The results of the study highlight the need for more research into endophytic fungi as a potentially rich and sustainable source of bioactive compounds. These compounds could help develop new, nature-inspired pharmaceutical agents and lead to breakthroughs in healing variety of illnesses, including cancer.
Additional file: Figures S1-S24.
ACKNOWLEDGMENTS
The authors express their gratitude to Rajneesh Anand, Chief Scientist, CSIR-IIIM, for providing analytical support throughout the study.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
SJ provided resource for experimentation. NS performed experimental work. MK executed LC-MS/MS studies. AQ and SKS performed in vitro cytotoxicity assay. UG performed formal analysis and supervision. NS, SJ and MK wrote the manuscript. UG reviewed the manuscript. MK revised the manuscript. All authors read and approved the final manuscript for publication.
FUNDING
This study was funded by CSIR-IIIM for major lab projects MLP-1009, MLP21002 & MLP21004.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript and/or in the supplementary files.
ETHICS STATEMENT
Not applicable.
- Frisvad JC, Smedsgaard J, Larsen TO, Samson RA. Mycotoxins, drugs and other extrolites produced by species in Penicillium subgenus Penicillium. Stud Mycol. 2004;49(201):e41.
- Pham-Huy LA, He H, Pham-Huy C. Free radicals, antioxidants in disease and health. Int J Biomed Sci. 2008;4(2):89-96.
- Manganyi MC, Ateba CN. Untapped potentials of endophytic fungi: A review of novel bioactive compounds with biological applications. Microorganisms. 2020;8(12):1934.
Crossref - Ortega HE, Torres-Mendoza D, Caballero EZ, Cubilla-Rios L. Structurally uncommon secondary metabolites derived from endophytic fungi. J Fungi. 2021;7(7):570.
Crossref - Aly AH, Debbab A, Kjer J, Proksch P. Fungal endophytes from higher plants: a prolific source of phytochemicals and other bioactive natural products. Fungal Divers. 2010;41:1-16.
Crossref - Zhao J, Shan T, Mou Y, Zhou L. Plant-derived bioactive compounds produced by endophytic fungi. Mini Reviews in Medicinal Chemistry. 2011;11(2):159-168.
Crossref - Faisal M, Singh P, Irchhaiya R. Review on Albizia lebbeck a potent herbal drug. Int Res J Pharm. 2012;3(5):63-68.
- Bobby MN, Wesely EG, Johnson M. In vitro anti– bacterial activity of leaves extracts of Albizia lebbeck Benth against some selected pathogens. Asian Pacific Journal of Tropical Biomedicine. 2012;2(2):S859-S862.
Crossref - Zia-Ul-Haq M, Ahmad S, Qayum M, Ercisli S. Compositional studies and antioxidant potential of Albizia lebbeck (L.) Benth. Pods and seeds. Turk J Biol. 2013;37(1):25-32.
Crossref - Sivakumar B, Velmurugan C, Bhargava A, Kumar PL. Diuretic activity of methanolic extract of Albizia lebbeck. Research and Reviews: J Pharm Toxicol Stud. 2013;1(1):1-3.
- Balkrishna A, Sakshi, Chauhan M, Dabas A, Arya V. A Comprehensive Insight into the Phytochemical, Pharmacological Potential, and Traditional Medicinal Uses of Albizia lebbeck (L.) Benth. Evid Based Complement Alternat Med. 2022(1):5359669.
Crossref - Devari S, Jaglan S, Kumar M, et al. Capsaicin production by Alternaria alternata, an endophytic fungus from Capsicum annum; LC–ESI–MS/MS analysis. Phytochemistry. 2014;98:183-189.
Crossref - Deshidi R, Devari S, Kushwaha M, et al. Isolation and quantification of alternariols from endophytic fungus, Alternaria alternata: LC ESI MS/MS analysis. ChemistrySelect. 2017;2(1):364-368.
Crossref - Sharma V, Singamaneni V, Sharma N, et al. Valproic acid induces three novel cytotoxic secondary metabolites in Diaporthe sp., an endophytic fungus from Datura inoxia Mill. Bioorg Med Chem Lett. 2018;28(12):2217-2221.
Crossref - Arora D, Sharma N, Singamaneni V, et al Isolation and characterization of bioactive metabolites from Xylaria psidii, an endophytic fungus of the medicinal plant Aegle marmelos and their role in mitochondrial dependent apoptosis against pancreatic cancer cells. Phytomedicine. 2016;23(12):1312-1320.
Crossref - Sharma N, Murtaza M, Ahmed W, Goutam U, Jaglan S. Isolation, characterization, and diversity of fungal endophytes from Albizia lebbeck in Jammu & Kashmir, India. J Appl Biol Biotechnol. 2025;13(4):89-99.
Crossref - Sharma N, Kushwaha M, Arora D, et al. New cytochalasin from Rosellinia sanctae-cruciana, an endophytic fungus of Albizia lebbeck. J Appl Microbiol. 2018;125(1):111-120.
Crossref - El-Sayed E-SR, Hazaa MA, Shebl MM, Amer MM, Mahmoud SR, Khattab AA. Bioprospecting endophytic fungi for bioactive metabolites and use of irradiation to improve their bioactivities. AMB Expr. 2022;12(1):46.
Crossref - Kaur N, Arora DS, Kalia N, Kaur M. Antibiofilm, antiproliferative, antioxidant and antimutagenic activities of an endophytic fungus Aspergillus fumigatus from Moringa oleifera. Mol Biol Rep. 2020;47(4):2901-2911.
Crossref - Zhao M, Guo D-L, Liu G-H, et al. Antifungal halogenated cyclopentenones from the endophytic fungus Saccharicola bicolor of Bergenia purpurascens by the one strain-many compounds strategy. J Agric Food Chem. 2020;68(1):185-192.
Crossref - Chen L-L, Wang P, Chen H-Q, et al. New furan derivatives from a mangrove-derived endophytic fungus Coriolopsis sp. J5. Molecules. 2017;22(2):261.
Crossref - Wiese J, Ohlendorf B, Blumel M, Schmaljohann R, Imhoff JF. Phylogenetic identification of fungi isolated from the marine sponge Tethya aurantium and identification of their secondary metabolites. Mar Drugs. 2011;9(4):561-585.
Crossref - Grove JF. New metabolic products of Aspergillus flavus. Part IV. 4′-Hydroxyasperentin and 5′-hydroxyasperentin 8-methyl ether. J. Chem. Soc., Perkin Trans. 1. 1973;22:2704-2706.
Crossref - Wang S, Li X-M, Teuscher F, et al. Chaetopyranin, a benzaldehyde derivative, and other related metabolites from Chaetomium globosum, an endophytic fungus derived from the marine red alga Polysiphonia urceolata. J Nat Prod. 2006;69(11):1622-1625.
Crossref - Tang Q, Guo K, Li X-Y, et al. Three new asperentin derivatives from the algicolous fungus Aspergillus sp. F00785. Mar Drugs. 2014;12(12):5993-6002.
Crossref - Leshchenko EV, Antonov AS, Dyshlovoy SA, et al. Meroantarctines A–C, meroterpenoids with rearranged skeletons from the alga-derived fungus Penicillium antarcticum KMM 4685 with potent p-glycoprotein inhibitory activity. J Nat Prod. 2022;85(12):2746-2752.
Crossref - Wang X, Radwan MM, Tarawneh AH, et al. Antifungal activity against plant pathogens of metabolites from the endophytic fungus Cladosporium cladosporioides. J Agric Food Chem. 2013;61(19):4551-4555.
Crossref - She Z-G, Huang H-R, Lin Y-C, Vrijmoed L, Jones EBG. 1, 4-Dihydroxy-2-methoxy-7-methylanthracene-9, 10-dione. Struct Rep. 2006;62(9):3737-3738.
Crossref - Xia G, Li J, Li H, et al. Alterporriol-type dimers from the mangrove endophytic fungus, Alternaria sp.(SK11), and their MptpB inhibitions. Mar Drugs. 2014;16;12(5):2953-2969.
Crossref - Keller G, Moser M, Horak E, Steglich W. Chemotaxonomic investigations of species of Dermocybe (Fr.) W nsche (Agaricales) from New Zealand, Papua New Guinea and Argentina. Sydowia. 1988;40:168-87.
- Anke H, Kolthoum I, Laatsch H. Metabolic products of microorganisms. 192. The anthraquinones of the Aspergillus glaucusgroup. II. Biological activity. Arch Microbiol. 1980;126(3):231-236.
Crossref - Wakulinski W, Kachlicki P, Sobiczewski P, et al. Catenarin production by isolates of Pyrenophora tritici-repentis(Died.) Drechsler and its antimicrobial activity. J Phytopathol. 2003;151(2):74–79.
Crossref - Bouras N, Strelkov, SE. The anthraquinone catenarin is phytotoxic and produced in leaves and kernels of wheat infected by Pyrenophora tritici-repentis. Physiol Mol Plant Pathol. 2008;72(1-3):87-95.
Crossref - Martorell M, Castro N, Victoriano M, et al. An update of anthraquinone derivatives emodin, diacerein, and catenarin in diabetes. Evid Based Complement Altern Med. 2021;2021(1):3313419.
Crossref - Dong X, Fu J, Yin X, Cao S, Li X, Lin L, Huyiligeqi, Ni J. Emodin: A review of its pharmacology, toxicity and pharmacokinetics. Phytother Res. 2016;30(8):1207–1218.
Crossref - Lin CH, Tseng HF, Hsieh, PC, et al. Nephroprotective role of chrysophanol in hypoxia/reoxygenation-induced renal cell damage via apoptosis, ER stress, and ferroptosis. Biomedicines. 2021;9(9):1283.
Crossref - Jadaun, KS, Mehan S, Sharma A, Siddiqui, EM, Kumar S, Alsuhaymi N. Neuroprotective effect of chrysophanol as a PI3K/AKT/mTOR signaling inhibitor in an experimental model of autologous blood-induced intracerebral hemorrhage. Curr Med Sci. 2022:1-18.
Crossref - Cui WH, Zhang, HH, Qu ZM, Wang Z, Zhang DJ, Wang S. Effects of chrysophanol on hippocampal damage and mitochondrial autophagy in mice with cerebral ischemia reperfusion. Int J Neurosci. 2022;132(6):613–620.
Crossref - Augustin N, Nuthakki VK, Abdullaha M, Hassan QP, Gandhi SG, Bharate SB. Discovery of helminthosporin, an anthraquinone isolated from Rumex abyssinicusJacq as a dual cholinesterase inhibitor. ACS Omega. 2020;5(3):1616-1624.
Crossref - Fiala J, Battlogg M, Bosking J, et al. Photoantimicrobial anthraquinones in Australian fungi of the genus Cortinarius. Fitoterapia. 2025;182:106402.
Crossref
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.