ISSN: 0973-7510
E-ISSN: 2581-690X
Extra pulmonary tuberculosis presents a diagnostic dilemma for both physicians as well as for clinical microbiologists . The laboratory diagnosis of tuberculosis ranges from simple microscopy,culture to complex molecular assays. To evaluate the sensitivity, specificity and turnaround time of microscopy, culture and PCR in the diagnosis of Extrapulmonary tuberculosis & to evaluate the use of PCR in the early diagnosis of Extrapulmonary tuberculosis. A total of 71 samples patients with strong clinical suspicion of extra-pulmonary tuberculosis were processed and evaluated by ZN staining, fluorescent microscopy, LJ culture, BacT Alert culture and PCR. The positivity rates by microscopy, LJ culture, BacT Alert culture and PCR were 11.26%, 8.45%, 14.08% and 14.08% respectively. The sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) of both staining methods was 50%, 92.3%, 37.5% and 95.2% respectively. The sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) of BacT Alert culture was 83.3%, 92.3%, 50% and 98.4% respectively. The recovery rate was higher by BacT Alert culture (90.9%) compared to LJ culture (63.63%). The mean turnaround time for culture positivity was 36.3 days with LJ culture and 14.6 days with BacT Alert culture. The sensitivity, specificity, positive predictive value and negative predictive value of PCR assay was 66.66%, 90.76%, 40% and 96.72% respectively. PCR has high sensitivity, specificity, substantial level of agreement with BacT Alert culture and shorter turnaround time. Therefore, use of PCR in combination with other diagnostic modalities is a useful tool to detect additional EPTB cases which may be missed otherwise.
Extrapulmonary tuberculosis, diagnostic methods.
Tuberculosis is one of the leading infectious diseases in the world and remains a major public health problem causing considerable morbidity and mortality. 1, EPTB constitutes about 15-20% of all TB cases.2,3 With HIV pandemic, the EPTB scenario is further complicated, as EPTB constitutes more than 50% of all cases of TB in HIV positive patients.4
The definitive & rapid diagnosis of EPTB is challenging since conventional techniques have limitations .The major pitfalls in the diagnosis of EPTB are atypical clinical presentations resulting in delay or deprivation of treatment & the lack of accurate diagnostic resources. 5 This often leads to empirical treatment based on clinical grounds without pathological and/or bacteriological confirmation, leading to over-diagnosis and unnecessary treatment.6
Delayed diagnosis results in increasing morbidity, mortality and cost to the health care system. The outcome for the patient could be improved if rapid, simple & reliable tests are available.7 There are several methods are available for the laboratory diagnosis of tuberculosis ranging from simple microscopy to complex molecular biological techniques.8 The PCR assays targets various genes for rapid detection of M tuberculosis complex with encouraging results. The aim of this study was to evaluate the sensitivity, specificity and turnaround time of microscopy, culture and PCR in the diagnosis of Extrapulmonary tuberculosis and to evaluate the use of PCR in the early diagnosis of Extrapulmonary tuberculosis.
Patients with strong clinical suspicion of extra-pulmonary tuberculosis willing to participate after informed consent were included in the study. The cases already on Anti-tubercular therapy or had been confirmed as having tuberculosis was excluded from study.
A total of 71 samples were included in our study & were processed were processed and evaluated by ZN staining, fluorescent microscopy, LJ culture, BacT Alert culture and PCR. The 71 clinical specimens included in this study were pus (15), endometrial biopsy (14), lymph node aspirate (10), peritoneal fluid (9), pleural fluid (7), tissue (5), CSF (5), synovial fluid (3), urine (3) as shown in the Table 1.
Table (1):
Distribution of Extrapulmonary tuberculosis samples( N =71)
Samples |
Number |
|---|---|
Pus |
15 |
Endometrial biopsy |
14 |
Lymph node aspirate |
10 |
Peritoneal fluid |
09 |
Pleural fluid |
07 |
CSF |
05 |
Tissue |
05 |
Synovial fluid |
03 |
Urine |
Processing of clinical samples
Sterile body fluids were centrifuged, where as pus ,urine specimen were digested & decontaminated by using Modified Petroff’s (4% NaOH) method . Tissue & lymphnode aspirates were homogenized using sterile tissue homogenizer. The Sediment thus obtained were subjected to ZN staining, fluorescent microscopy, LJ culture, BacT Alert culture and PCR .
Auramine rhodamine stain was used in fluorescent microscopy,In house prepared Lowenstein Jensen medium were used for solid culture media. and for liquid culture method , BacT/Alert MP culture media (BioMerieux, ) were used by following manufacturer’s instructions .
Polymerase Chain Reaction
Extraction of DNA, amplification & detection were done in physically separate areas.
The DNA was extracted by spin column method ( Qiagen tissue extraction kit)DNA was extracted from 71 clinical samples, M. tuberculosis standard strain (H37RV). Each step of the extraction protocol was performed inside bio safety cabinet, using protected tips and dedicated pipettes at room temperature.
PCR amplification of DNA
The primers used for the assay were based on the published sequence.The species specific primer amplified a 245 base pair nucleotide sequence in IS 6110 present in strains of the M.tuberculosis
The sequences of the species specific primers were:
Forward: 5’ CGT GAG GGC ATC GAG GTG GC 3’
Reverse: 5’ GCG TAG GCG TCG GTG ACA AA 3’
The conditions were :
Initial delay: 94o C for 5 minutes.
94o C for 2 minutes
68o C for 2 minutes
72o C for 2 minutes
Final delay: 72 o C for 5 minutes
Detection of Amplification products: PCR products were detected on 1.5% agarose gel in 1X TE buffer containing ethidium bromide at 10µg/ml concentration under ultra violet illumination.
The positivity rates by microscopy, LJ culture, BacT Alert culture and PCR were 11.26%, 8.45%, 14.08% and 14.08% respectively as shown in Table 2.
Table (2):
Positivity rate of individual diagnostic method employed in detection of M tuberculosis ( N =71)
Sl No |
Diagnostic Method |
Positive |
Positivity rate (% age) |
|---|---|---|---|
1 |
Ziehl Neelsen Stain |
8 |
11.26 |
2 |
Fluorescent stain (Auramine Rhodamine Stain) |
8 |
11.26 |
3 |
Lowenstein Jensen Medium (Solid Culture) |
6 |
8.45 |
4 |
Bact Alert (Liquid Culture) |
10 |
14.08 |
5 |
Polymerase Chain reaction |
10 |
14.08 |
The sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) of both staining methods was 50%, 92.3%, 37.5% and 95.2% respectively as shown in Table 3. The mean turnaround time for culture positivity was 36.3 days with LJ culture (range 5 to 49 days) and 14.6 days with BacT Alert culture (range 4 to 21 days). The use of BacT Alert culture has reduced the mean detection time by 2.5 times when compared to LJ culture.
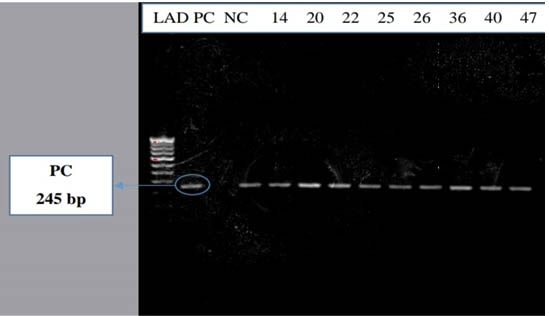
Fig. 1. Gel documentation picture of samples positive by PCR assay (LAD – Ladder 100 bp, PC- positive control, NC- negative control)
The sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) of BacT Alert culture was 83.3%, 92.3%, 50% and 98.4% respectively as shown in Table 3. BacT Alert culture was positive in additional 5 samples when compared to LJ culture. The recovery rate was higher by BacT Alert culture (90.9%) compared to LJ culture (63.63%).
Table (3):
Sensitivity, Specificity, Positive predictive value (PPV), Negative predictive value (NPV) of individual diagnostic method Reference : Lowenstein Jensen culture as Gold Standard
Sl No |
Diagnostic Method |
Sensitivity ( % age ) |
Specificity ( % age ) |
PPV (% age) |
NPV ( % age ) |
|---|---|---|---|---|---|
1 |
Ziehl Neelsen Stain |
50 |
92.3 |
37.5 |
95.2 |
2 |
Fluorescent stain (Auramine Rhodamine Stain) |
50 |
92.3 |
37.5 |
95.2 |
3 |
Bact Alert (Liquid Culture) |
83.3 |
92.3 |
50 |
98.4 |
4 |
Polymerase Chain reaction |
66.7 |
90.8 |
40 |
96.7 |
PCR was positive in 10 samples. The sensitivity, specificity, positive predictive value and negative predictive value of PCR assay was 66.66%, 90.76%, 40% and 96.72% respectively. The mean turnaround time for ZN staining , fluorescent staining, LJ culture, BacT Alert culture & PCR are 40 minutes , 30 minutes,36.3 days 14.6 days & 5 hours respectively as shown in Table 4.
Table (4):
Mean Turn Around Time (TAT ) of individual diagnostic method
Sl No |
Diagnostic Method |
TAT |
|---|---|---|
1 |
Ziehl Neelsen Stain |
40 minutes |
2 |
Fluorescent stain (Auramine Rhodamine Stain) |
30 minutes |
3 |
Lowenstein Jensen Medium (Solid Culture) |
36.3 days |
4 |
Bact Alert (Liquid Culture) |
14.6 days |
5 |
Polymerase Chain reaction |
5 hours |
Statistical analysis
Statistical analysis was done using ‘SPSS 22’ software and the sensitivities, specificities, positive predictive values, negative predictive values were calculated considering LJ culture as gold standard. Cohen’s kappa value was also obtained to assess the reproducibility and level of agreement between the PCR assay and other diagnostic tests employed.
In our study, the level agreement between BacT Alert and PCR was ‘substantial’ with a kappa value of 0.767 whereas ‘moderate’ level of agreement between LJ culture and PCR with kappa value of 0.441
Tuberculosis remains a major health problem in the developing countries in the world especially in India . In clinical practice the diagnosis of EPTB is difficult because of its non-specific, misleading and variety of clinical manifestations 9,10. The microscopy and culture are still the methods of choice for the diagnosis of tuberculosis in most of the microbiological laboratories.
The sensitivity of microscopy and culture on LJ media are low in EPTB owing to its paucibacillary nature. Cultivation of M.tuberculosis is considered as the gold standard in the diagnosis of tuberculosis. This gold standard lacks sensitivity and is negative in specimens from majority of paucibacillary cases .Recent introduction of liquid culture has reduced the time taken for culture positivity and also increased the rate of isolation of M tuberculosis 11, 12.The role of PCR in early diagnosis of EPTB has been evaluated with the hope of shortening the time required for diagnosis of EPTB.13,14.
Out of 71 samples processed, 16 samples (22.5%) were positive by one or more methods employed for the detection of acid fast bacilli. Microscopy showed the positivity of 11.26% by both the methods (ZN and fluorescent staining method). A study done by Sudhindra KS et al also reported equal positivity rates of ZN and fluorescent staining method 8. The sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) of both ZN staining and fluorescent staining were 50%, 92.3%, 37.5% and 95.2% respectively which is much higher when compared to studies done by others.
Among 8 cases which were positive by ZN staining and fluorescent microscopy, 2 were also positive by both culture methods and PCR.
Our study showed least positivity rate by LJ culture i.e. 8.45%. The low positivity rate may be attributed to paucibacillary nature of the disease and the sampled site may not represent the site of active infection13,14,15,16.Low positivity rates were also reported by Chhina D et al (2.1%)17, Ajantha GS et al (5%)18, Sharma K et al (11.3%)19 and Siddiqui MAM et al (15%)20.
BacT Alert had positivity rate of 14.08% which is much higher than reported by Angeby Kak et al (3.44%)21, Carricago A et al (4.07%)22 and Piersimoni C et al (7.07%)23. PCR was positive in 10 samples accounting for 14.08% of positivity rate. In studies by Hajia M et al 12, Pednekar SN et al 24 and Chawla K et al 25 reported higher positivity rates of 41%, 53% and 74 % respectively.
One lymph node aspirate was positive by BacT Alert and PCR but failed to grow on LJ media. This could be due to non uniform distribution of bacilli in the aliquots apportioned for the diagnostic tests 26,27. Two samples were PCR negative but culture positive and later diagnosed as Non tuberculous mycobacteria (NTM ) . Three samples were negative by both culture methods and PCR. The reason for smear positive but culture and PCR negative could be the availability of small quantity of sample for processing.
In our study sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) of BacT Alert culture was 83.3%, 92.3%, 50% and 98.4% respectively which correlates with study by Martinez MS et al 28 who reported sensitivity of 87.7% and specificity of 99.2%.
The percentage of isolation of M tuberculosis by BacT Alert and LJ culture from synovial fluid was 33.3%, whereas in lymph node it was 30% by BacT Alert compared to 20% by LJ culture. In pleural fluid, percentage of isolation by BacT Alert was 28.57% whereas isolation by LJ was none; however, in pus samples, isolation percentage was higher by LJ culture (20%) compared to BacT Alert (13.33%). Endometrial biopsy, peritoneal fluid, tissue samples, CSF and urine samples did not yield growth of M tuberculosis. Since the number of synovial fluid samples processed were less, the isolation percentage by both culture methods in present study is high. In study by Ghadage et al has reported maximum isolation of M tuberculosis by LJ culture in pus (33%), followed by pleural fluid (26.3%), fine needle aspiration biopsy (25%) and CSF (12.5%)29.
BacT Alert culture was positive in additional 5 samples when compared to LJ culture. The recovery rate was higher by BacT Alert culture (90.9%) compared to LJ culture (63.63%). Though BacT Alert had higher recovery rate, in one case of suspected Pott’s spine only LJ culture was positive. This underlines the need to use the combination of liquid and solid media especially in the diagnosis of EPTB.
PCR was positive in 10 samples. The sensitivity, specificity, positive predictive value and negative predictive value of PCR assay was 66.66%, 90.76%, 40% and 96.72% respectively. These results are comparable to studies by Oberoi A et al 30 and Siddqui MAM et al 20.
PCR was positive in 2 samples (endometrial biopsy, lymph node aspirate) where both the staining techniques and both the culture methods were negative. This could be due to paucibacillary nature and high sensitivity of PCR.
However in one case of suspected Pott’s spine, only LJ culture was positive but other methods failed to detect M tuberculosis. This false negative PCR result could be due to nonuniform distribution of bacilli in the aliquots apportioned for the diagnostic tests26, ineffective extraction of DNA or the presence of PCR inhibitors 31.
The mean turnaround time for ZN staining was 40 minutes as compared to fluorescent staining (30 minutes). The mean turnaround time for culture positivity was 36.3 days with LJ culture (range 5 to 49 days) and 14.6 days with BacT Alert culture (range 4 to 21 days). The use of BacT Alert culture has reduced the mean detection time by 2.5 times when compared to LJ culture. PCR mean detection time was 5 hours. Thus, PCR reduces detection time when compared with culture. PCR provides additional information when positive microscopy results are combined with PCR results and it also differentiates M tuberculosis from NMT.
The Cohen’s kappa statistics was applied to assess the reproducibility and the level of agreement between the PCR assay and other diagnostic tests. In our study, the level agreement between BacT ALERT and PCR was ‘substantial’ with a kappa value of 0.767 whereas ‘moderate’ level of agreement between LJ culture and PCR with kappa value of 0.441.
The ability of PCR to detect even few organism from clinical sample makes it very attractive diagnostic tool in the diagnosis of EPTB. In the present study, PCR had sensitivity of 66.7%, specificity of 90.76% and short detection time (5 hours). The sensitivity of PCR from clinical samples reported from different studies varies between 55% and 90%, which is much higher when compared to any other test used in the diagnosis of EPTB. This makes PCR a valuable screening test, especially when limitations of conventional diagnostic modalities have negative impact on patient care. Though PCR has been reported to have high sensitivity and specificity, it has few drawbacks. It is very expensive, needs expertise and proper standardization and risk of false negative and false positive results.
PCR has high sensitivity, specificity, substantial level of agreement with BacT ALERT culture and shorter turnaround time; hence in the era of evidence based clinical practice, it adds meaningful evidence to the results of conventional method employed in the diagnosis of EPTB, to rule-in or rule-out the disease. Therefore, use of PCR in combination with other diagnostic modalities helps to provide maximum information to clinicians in the diagnosis of EPTB.
- http://www.who.int/tb/publications/global_report/en/
- Lee JY. Diagnosis and Treatment of Extrapulmonary Tuberculosis. Tuberc Respir Dis 2015; 78: 47-55.
- World Health Organization Global Tuberculosis Report 2015. Available from:http://:www.health-e.org.2a/wp-contact/uploads/2015/10/global-TB-report-2015
- Purohit M, Mustafa T. Laboratory Diagnosis of Extrapulmonary Tuberculosis (EPTB) in Resource- Constrained Setting: State of the Art, Challenges and the Need. J Clin Diagn Res 2015; 9: EE01-EE06.
- Sharma S K, Mohan A. Extrapulmonary Tuberculosis. Indian J Med Res 2004; 120: 316-53.
- Sekar B, Selvaraj L, Alexis A, Ravi S, Arunagiri K, Rathinavel L. The Utility of IS Sequence Based Polymerase Chain Reaction in Comparison to Conventional Methods in the Diagnosis of Extra-Pulmonary Tuberculosis. Indian J Med Microbiol 2008; 26: 352-5.
- Pingle P, Apte P, Trivedi R. Evaluation of Microscopy, Culture and PCR Methods in the Laboratory Diagnosis of Genito-urinary Tuberculosis. American Journal of Infectious Diseases and Microbiology 2014; 2:17-21.
- Sudhindra KS, Prakash RC, Fathima F, Venkatesh VN, Rao A, Vijaynath V. Evaluation of PCR in Early Diagnosis of Tuberculosis. J Med Res Pract 2012; 1: 24-28.
- Arvind N, Chand P, Baliga S, Bharati B, Shenoi S, Saralaya V. A Comprehensive Comparison of PCR Based Assay Versus Microscopy and Culture in the Diagnosis of Tuberculosis in Different Clinical Specimens. Int J Biol Med Res 2014; 5: 3763-68.
- Mercy EG, Aditya T, Amarendra KE, Dan MM. Review on Mycobacterium Tuberculosis. RRJMB 2016: 9-17
- Lange C, Mori T. Advances in the Diagnosis of Tuberculosis. Journal of Asian Pacific Society of Respiriology 2010; 15:220-240.
- Hajia M, Rahbar M, Amini R. Is PCR assay reliable for diagnosis of extrapulmonary tuberculosis? Afr J Microbiol Res 2009; 3:877-881
- Winn W, Allen S, Janda W, Koneman E, Procop G, Schreckenberger P, Woods G. Koneman’s Color Atlas and Textbook of Diagnostic Microbiology. 6th ed:2006.1064-1124.
- Arora VK, Gupta R. Trends of extrapulmonary tuberculosis under revised national tuberculosis control program; a study from south Dehli. Indian J Tuberc 2006; 53:73-83
- Musellim B, Erturan S, Duman ES, Ongen G. Comparison of extrapulmonary and pulmonary tuberculosis case; Factors influencing the site of reactivation. Int J Tuberc Lung Dis 2005; 9: 1220-1223
- Srivastava I, Bhatambare G.S, Deshmukh A.B, Bajpai P, Bajpai T, Singh T. Genital Tuberculosis: Evaluating Microscopy, Culture, Histopathology and PCR for Diagnosis All Play Their Role. Int J Curr Microbiol App Sci 2014; 3: 439-445
- Chhina D, Mohi G, Gupta R, Deeplyall K, Kaur J. Role of gen-probe amplified Mycobacterium tuberculosis Direct Test (AMTD) in the diagnosis of extrapulmonary tuberculosis. IJPRBS 2015; 4: 135-143
- Ajantha GS, Shetty PC, Kulkarni RD, Biradar U. PCR as a Diagnostic Tool for Extra-pulmonary Tuberculosis. JCDR 2013; 7: 1012-1015
- Sharma K, Appannanavar SB, Modi M, Singh M, Sharma A, Varma S. Role of multiplex polymerase chain reaction using IS6110 and Protein b for the diagnosis of extrapulmonary tuberculosis: North India. Indian J Pathol Microbiol 2015; 58:27-30
- Siddqui MAM, Anuradha PR, Nagamani K, Vishnu PH. Comparison of conventional diagnostic modalities, BACTEC culture with polymerase chain reaction for diagnosis of extrapulmonary tuberculosis. J Med Allied Sci 2013; 3: 53-58
- Angeby KA, Werngren J, Toro JC et al. Evaluation of the BacT/ALERT 3D system for recovery and drug susceptibility testing of Mycobacterium tuberculosis. Clin Microbiol Infect 2003; 9: 1148 –52.
- Carricajo A, Fonsale N, Vautrin A C, Aubert G. Evaluation of BacT/Alert 3D Liquid Culture System for Recovery of Mycobacteria from Clinical Specimens Using Sodium Dodecyl (Lauryl) Sulfate-NaOH Decontamination J Clin Microbiol 2001; 39:3799-3800
- Piersimoni C, Scarparo C, Callegaro A Tosi CP, Nista D, Bornigia S et al.Comparison of MB/BacT ALERT 3D System with Radiometric BACTEC System and Lo¨wenstein-Jensen Medium for Recovery and Identifihcation of Mycobacteria from Clinical Specimens: a Multicenter Study. J Clin Microbiol 2001; 39: 651-657
- Pednekar SN, Desai SL, Pol S, Kagal AS, Dharmashale SN, Bharadwaj RS. A Comprehensive Comparision Of Mpb64 Based Pcr Assay Versus Microscopy And Culture In The Diagnosis Of Clinically Suspected Cases Of Extrapulmonary Tuberculosis. Int J Recent Advances in Multidisciplinary Research 2015; 2: 0436-0440
- Chawla K, Johar R, Vishwanath S, Mukhopadhyay C. Role of PCR in the Diagnosis of Pulmonary and Extra-Pulmonary Tuberculosis. NJLM 2015; 4:43-46.
- Anmam V, Kulkarni MH, Puranik RB. Comparison of modified fluorescent method and conventional Ziehl-Neelsen method in detection of acid fast bacilli in Lymph node aspirate. CytoJournal 2009; 6:13
- Chakravorty S, Sen MK, Tyagi JS. Diagnosis of Extrapulmonary Tuberculosis by Smear, Culture, and PCR Using Universal Sample Processing Technology. J Clin Microbiol 2005; 49: 4357-4362
- Martinez MS, Sardinas M, Garcia G, Mederos LM, Diaz R. Evaluvation of BacT ALERT 3D System for Mycobacteria isolates. Journal of Tuberculosis Research 2014; 9: 59-64.
- Ghadage DP, Muley VA, Pednekar S, Bhore AV. Evaluation of polymerase chain reaction using primer MPB 64 for diagnosis of clinically suspected cases of extra-pulmonary tuberculosis. J Sci Soc 2014; 41:89-93
- Oberoi A, Aggarwal A. Comparison of the conventional diagnostic techniques, BACTEC and PCR. JK Sci 2007; 9:179-181
- Zakham F, Bazoui H, Akrim M, Lamrabet S, Lahlou O, Elmzibri M et al. Evaluation of conventional molecular diagnosis of mycobacterium tuberculosis in clinical specimens from Morocco. J Infect Dev Ctries 2012; 6 :40-45.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


