ISSN: 0973-7510
E-ISSN: 2581-690X
There are increments to the need of the usage of new non-chemical medicines, pharmaceuticals and health products. Plantsare traditionally used as medicine over the world for their pharmacological values. They effectively used as anti-fungal, antiviral, anti-bacterial, anti-inflammatory, anti-diabetic, anti-oxidant anti-ulcer, and antitussive. This study aims to investigate the effects of plant extracts (Glycyrrhiza glabra, Cuminum cyminum, Zingiber officinale, Origanum majorana and Petroselinum crispum) against different types of gram-positive and negative bacterial isolates. Antimicrobial activity of tested aqueous extracts by well-diffusion method against various bacterial isolates were done, to estimate their antibacterial activity. Results showed that the aquatic extracts of different plants produce a good antibacterial effects when compared with the synthetic antibiotic ciprofloxacin. As all bacterial isolates in this study where sensitive to these extracts with variable ranges of inhibition zones ranging from 18-32mm in diameter. Thus we can concluded that the aquatic extracts of Glycyrrhiza glabra, Cuminum cyminum, Zingiber officinale, Origanum majorana and Petroselinum crispum can be beneficial as treatment of UTI-causing bacteria.
Glycyrrhiza glabra, Cuminum cyminum, Zingiber officinale, Origanum ajorana, Petroselinum crispum
Plants are rich sources of natural products that are extracted from them due to their content of active substances that used for centuries. These extracts can be used as antimicrobial activity to treat various diseases caused by pathogenic organisms in addition to their numerous pharamaceutical effects such as antibacterial, antifunfial, antiparasitic and anticarcinogenic1.The use of crude plant extracts in herbal medicine for treatment of diseases can be seen in different form like infusion or tincture2. Bacterial resistance to different types of antibiotics is growing because of genetic variability and mutation therfore we must understand the genetic mechanisms of resistance to develop new drugs from plants that have natural products with safety and efficiency for human being3.
Plant extracts widly used to reduce the bacterial growth of most infections. The antimicrobial activity of them dependent on their methods of extraction and chemical structures of plants. Glycyrrhiza glabra has important chemical components called licorice extracted from its roots that have medical benefits as antibacterial, antiviral and anti-ulcerative activity,it used for treatment of upper respiratory and liver diseases4,5.
Cuminum cyminum has anticholesterol and antimicrobial activity and the active components of it is cumin oil or cuminol, these extracts are important in stimulation of digestive system enzymes secration to treat different types of diseases and used as bactericides against plant bacterial diseases 6,1.
Ginger (Zingiber officinale) has anti-microbial activity that can be used in treatment of bacterial infections through its aromatic and medicinal properties. Both ethanolic and aqueus extraction of Ginger have bactericidal effects on some types of pathogenic organismis7,8. Petroselinum crispum extractes have antibacterial activity, damage bacterial cell wall and inhibit the bacterial growth in addition to their cytotoxic activity at higher concentration9.
Many researches found the Origanum majorana has an essential oils that affect the bacterial growth through altering cell membrane permeability, and causing a distortion of the membrane structure, also the extractes of this plant has agricultural, pharmaceutical and interfer with cosmetic industries10.
The antimicrobial activity of plant extracts have been comparad with the effects of different types of antibiotics through measuring the inhibition zones of bacterial growth, ciprofloxacin has a wide antibacterial sepectrum against the pathogenic gram-positive and -negative bacteria that caused different types of urinary truct, respiratory truct, skin infections and other types of infections through inhibition the action of enzymes that interfer with bacterial repliction11.
Aim
This study aims to investigate the action of plant extracts(Glycyrrhiza glabra, Cuminum cyminum, Zingiber officinale, Origanum majorana and Petroselinum crispum) against a number of positively- and negatively-gram stained bacterial isolates and copmarad them with the effects of ciprofloxacin.
Extraction Methods
Plants were collected fromthe market. Aqueous extracts were prepared by soaking 30 grams of powder in 100ml of distillated water, leave them to stand for 72 hours, and sterilized by using Millipore 0.45 filter paper. 50% concentration of the extract was obtained by this way12.
Bacterial Isolates
An overall of eight negatively-gram stained and three positively-gram stained bacterial isolates were collected from urine clinical samples (UTI) and used in this study. These bacterial isolates were; S. saprophyticus, S. epidermidis,S. aureus, P. fluresence, P.aeruginosa, E. aerugenes, E.coli, K. pneumoniae, P. vulgaris, Acinetobacter, Proteus mirabilis. All these bacterial isolates were cultivated for activation and re-cultured as three successive times on nutrient agar plates. Identification and diagnosis of these isolates were applied by conventional biochemical procedures13.
Agar Well Diffusion Assay for In vitro Testing the Antimicrobial Activity (14)
Bacterial growths of Loopfull from each bacterial isolates were taken and applied into nutrient broth, incubation at 37°C for 18 hours. Normal saline was used to dilute the bacterial suspensions to adjust the turbidity and comparing with standard McFarland tube number 0.5. Cotton swab dipped into the tube of suspension and streaked Mueller-Hinton agar plates, left them to dry for 5-15 minutes at room temperature. Four wells of about 5mm in diameter were made in the media by cork borer, 0.1ml of the extracts were added. Then incubation overnight at 37 C°. Zone of inhibition was measured, disks of ciprofloxacin were add in the center of agar plate to compare the results of bacterial inhibition13.
The evaluation of bacterial inhibitory activity of aquatic extracts of Glycyrrhiza labra was processed using the agar diffusion test, as in Figure (1). This figure shows that Glycyrrhiza labra exhibited an inhibitory activity against all the bacterial isolates of the study with inhibition zones ranging from 18-25mm, with the largest inhibition against E.coli (25mm) followed by Pseudomonas aerogenosa and Staphylococcuss aprophyticus (24 and 23mm) respectively. Lowest inhibition against Klebsiella pneumonia and Enterobacter aerogenes (18mm) for each.
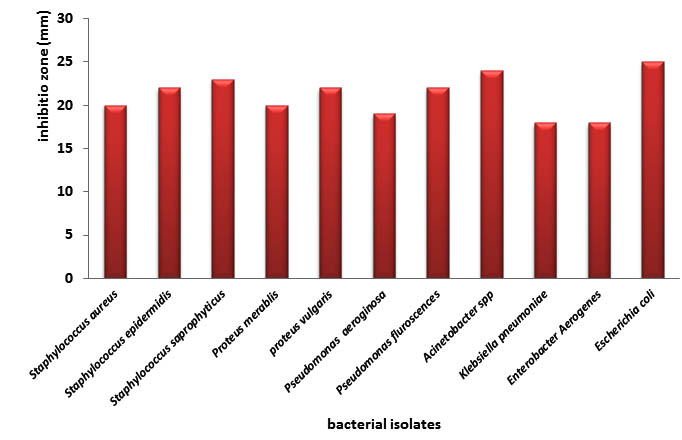
Fig. 1. Antibacterial activity of Glycyrrhiza labra against bacterial isolates
Figure (2) shows the inhibitory activity of aquatic extracts of Cuminum cyminum against different bacterial isolates in the study. Results demonstrated that inhibition zones were the same (20mm) against all the tested bacterial isolates. While the diameter of inhibition zones produced by Ginger were ranging from 23mm against Enterobacter aerogenes to 18mm with Klebsiella pneumonia; and 22mm against most of isolates, figure (3).
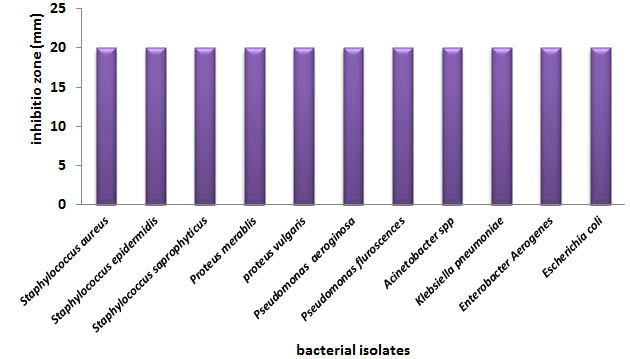
Fig. 2. Antibacterial activity of Cuminum cyminum against bacterial isolates
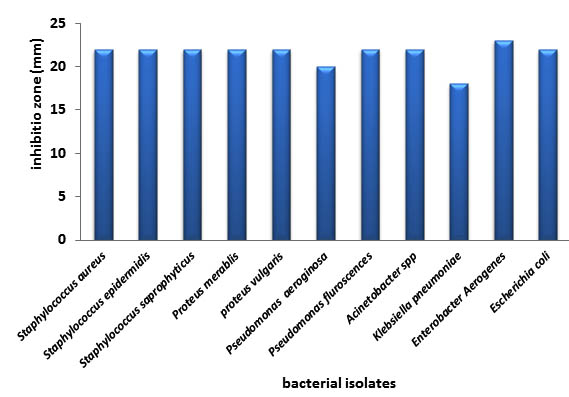
Fig. 3. Antibacterial activity of ginger against bacterial isolates
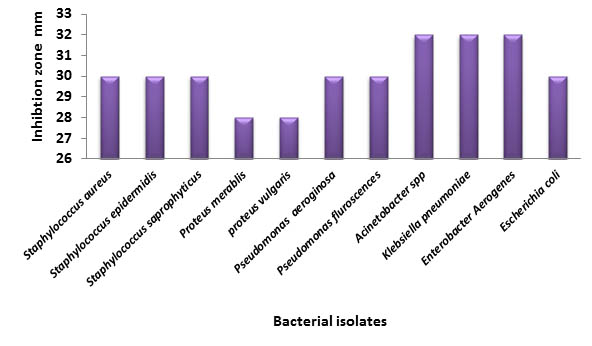
Fig. 4. Antibacterial activity of Petroselinum crispum against bacterial isolates
The antibacterial effect of Petroselinum crispum exhibited as a high inhibition level as compared with other extracts, as the inhibition zone diameters range from 32-28mm; higher effects against Klebsiella pneumonia, Enterobacter aerogenes and Acinetobacter spp (32mm), followed by 30mm as an inhibition against 6 bacterial isolates, and the lowest one (28mm) with Proteus mirabilis and Proteus vulgaris (4).
Results in table (5) revealed the antibacterial inhibitory effect of Origanum majorana Gram positive and negative bacteria with the highest inhibition 32 mm against E. coli, 30 mm against seven bacterial isolates, while the least inhibition against Pseudomonas aeruginosa and Pseudomonas fluroscence (28) and 27mm respectively. In contrast to all these aquatic extracts, Ciprofloxacin antibiotic is used as a synthetic antibacterial reagent; figure (6). This showed resistance of bacteria to this reagent; especially Pseudomonas aeruginosa and Pseudomonas fluroscence 0 mm, while the largest effect were only reach to about (17 mm) with staphylocci and E.coli, smallest zones were 12 mm with Proteus spp.
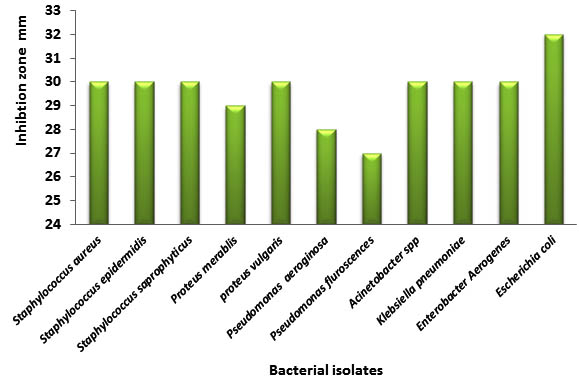
Fig. 5. Antibacterial activity of Origanum majorana against bacterial isolates
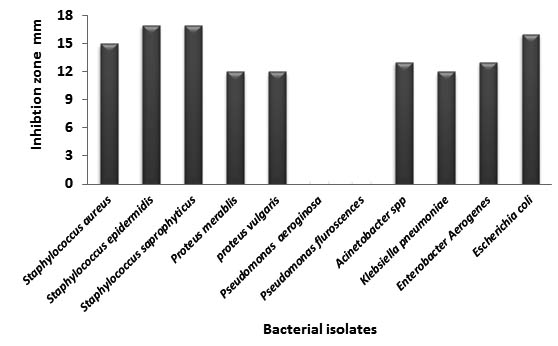
Fig. 6. Antibacterial activity of Ciprofloxacin against bacterial isolates
Extract of Licorice root has been used traditionally for a long period od history as medicines and folk preparations, and being used in many diseases from hundred years before15. It contains a large number of active components. About 40-50% of its total dry material weight is accounted of water-soluble, biologically active complexes.
Glycyrrhizin represents about 10-25% of liquor ice root extract. It is considered to be the most commonly used of the folk medicines in Asia. Its structure is a saponin compound16, 17. Thus due of the existence of these different secondary metabolites such as flavonoids, alkaloids, and saponins in its aquatic-root extract, it exhibits potent antibacterial activity18, 19, also it has a role as antiviral agent against many viruses20, 21.
In vitro works had demonstrated that aquatic and ethanolic extracts of liquor ice display an inhibitory action on the cultures of Strept. pyogenes and Staph. aureus (22), and many pathogenic organisms23.
In vitro testing of the extracts from liquor ice root was carried out against13 bacterial isolates by the use of agar diffusion method, that showed various antibacterial activities. The inhibitory antibacterial properties of this extract to prevent the growth of S. flexneri, Shigella sonnei, S.paratyphi B. Salmonella typhi and ETEC E. coli was revealed24.
Glycyrrhizin had been studied by Alonso and Tratado22, as it is regularly used among the orally administered foods, and it obstructs the growth of some bacterial sorts, also can prevent dental caries formation, it demonstrated antimicrobial effects against both Gram-positive and -negative bacteria by Gupta et al25.
Cumin spice is used popularly as a traditional therapy, for the reason that aromatic substances are presenting this herb, it is prescribed for the mild digestive sicknesses, and used in the treatment of diseases caused by K. pneumoniae; especially the essential oil of its seeds that had displayed to have a significant in vitro antibacterial activity26,27. Cumin aldehyde and para-cymene are the main two active components of cumin; Cumin aldehyde has made known diverse activities like antifungal, antibacterial, anti-diabetic and anti-platelet28. Antimicrobial effect of both aqueous and oil extracts of it, had judged against a wide collection of valuable, pathogenic microbial strains of both gram-positive and -negative, as it is reported to obstruct Salmonella spp, E.coli, Aspergillus niger and Bacillus cereus growth29, 30.
Alcoholic extract and oil of Cumin seeds have ability to inhibit growth of Klebsiella pneumoniae, especially its clinical isolates, with improvement in the morphology of cells, decrement of urease activity and impeding capsule expression. Additionally, have capability to prevent properties of Streptococcus pyogenes and Streptococcus mutans in biofilm-formation. Moreover the cumin extracts has anti-fungal properties against human, soil, food and animal pathogens, yeasts, mycotoxin and aflatoxins producers31,32,33.
Ginger exhibited robust antibacterial and to a less degree antifungal properties. Main active ingredients of ginger have been studied in vitro to inhibit multiplication and growth of colon bacteria; as ginger can counteract bacterial ability to produce flatulence due to fermention of undigested carbohydrates. It constrains growth of Staphylococci, Salmonella, Proteus sp, E. coli and Streptococci(34).Gingerol, Ingenol and Shogaol, isolated from ginger rhizome are the main active components that exhibited antiviral and antibacterial activity against many pathogens; as M. avium and M. tuberculosis in vitro, H. pylori and many periodontal bacteria35, 36, 37.
Hindi et al.12 showed that the aquatic extract of ginger give a significant influence to inhibit most of the studied sixteen bacterial isolates both gram positive and negative, also in a study of Suhad and her coworkers displayed that the ginger aquatic extract gave the peak effect against Staphylococcus aureus, Streptococcus pyogenes, Proteus vulgaris, and Klebsiella pneumonia. A number of antibiotics like chloromphenicol, erythromycin, gentamicin, nalidixic acid and trimethoprim were used as a comparison with this extract of ginger, using antibiogram test. Aa results he instituted that the inhibitory action of ginger was superior than that to Gentamicin and Chloromphenicol against Proteus vulgaris, Klebsiella pneumoniae, Staphylococcus aureus and Streptococcus pyogenes38.
Various compounds from different phytochemical categories have been identified in Parsley. Also, different pharmacological activities have been attributed to it. In Iran Parsley as seeds or leaves has many usages for renal system, as inflammation, kidney stone, renal and urinary tract disease, and urinary tract infections39,40,41. Also, extracts from its leaves are widely used in Serbia as an antimicrobial agent42.
As Parsley leaves and stems possess antibacterial activity on B. subtilis and E. coli. Both hot and cold water extract from them demonstrated antibacterial activity against pseudomonas aeruginosa, S. aureus, S. epiderm and S. pyogenes, with higher inhibition zone in hot water extract43. Leaf extracts showed higher cell damage on both bacteria with higher activity with methanol extract, Coumarins are responsible components for this property44, 45.
Microbial analysis carried out by Nessrien et al46 and Gutierrez et al47 showed that, essential oils of marjoram has antimicrobial properties, as it is rich in phenolic compound that being particularly active as antimicrobials against both bacteria and fungi.
The essential oils from its leaves showed antibacterial effects on various bacteria (Bacillus cereus, E. coli, Staphylococcus aureus, Proteus spp., Enterobacter spp., Klebsiella spp., Acinetobacter spp. and Pseudomonas spp.) in agar diffusion assay. Also the ethanol and water extracts of majorana have shown antimicrobial activity against both gram positive and negative bacteria and its possible food applications by minimum inhibitory concentration estimation48.
Farooqi and Sreeramu49 have reported the antimicrobial activity of majorana against Bacillus anthracis, Proteus vulgaris, Salmonella spp, Streptococcus agalactiae, Streptococcus spp and Aspergillus fumigatus.Shahidi50 screened some used medical Iranian traditional plants for antibacterial properties against two E. coli strains and found that Majorana showed anti-E.coli activity.Leelavathi51 has conducted a comparative study of the antibacterial activity of crude extract of in vivo and in vitro leaves of majorana against Staphylococcus aureus and reported that in vitro leaf extract showed better antibacterial activity.
At present, most pathogenic bacteria develop an antibiotic resistance. Thus too verwhelm this frightening problem, it is an urgency to discover a numer of novel active compounds. Organic solvents and water are used to prepare extracts from these spices which are biologically active compounds, and can be applied for the synthesis of potent drugs. Thus spices, that considered as ausual components of our routine food preparations, which may give defense to a certain extent against bacterial pathogens the natural enemies.
- Bameri, Z., Amini-Boroujeni, N., Saeidi, S., Bazi, S. Antimicrobial Activity of Cyminum cuminum Against Biofilm E. coli.Inter. Res. J. Appl. Basic. Sci., 2013; 5(10): 1232-4.
- Masoud, E.A., Gouda, H.A. Effect of some plant extracts against gram negative bacteria in Njran Area, Saudia Arabia, Egypt. Acad . J. Biolog., 2013; 4: 85-92.
- Hindi, N.K.K., Chabuck, Z.A.G.Antimicrobial Activity of Different Aqueous Lemon Extracts. J. Appl. Pharmaceut. Sci., 2013; 3(6): 74-8.
- Nitalikar, M.M., Munde, K.C., Dhore, B.V., Shikalgar, S.N. Studies of Antibacterial Activities of Glycyrrhiza glabra Root Extract. Inter. J. Pharm. Tech. Res., 2010; 2(1): 899-901.
- Gupta, A., Maheshwari,D.K. Khandelwal,G.Antibacterial activity of Glycyrrhiza glabra roots against certain gram-positive and gram-negative bacterial strains. J. Appl. Nat. Sci., 2013; 5(2): 59-464.
- Iacobellis, N.S., Cantore, P., Capasso, F., Senatore, F.Antibacterial Activity of Cuminumcyminum L. and Carum carvi L. Essential Oils. J. Agric. Food Chem., 2005; 53: 57-61.
- Islam, K., Rowsni, A.A., Khan, M., Kabir S. Antimicrobial Activity of Ginger (Zingiber Officinale) Extracts Against Food- Borne Pathogenic Bacteria.Inter. J. Sci. Environm. Technol., 2014; 3(3): 867-71.
- Nikoli, M., Vasi, S., urevi, J., Stefanovi, O., omi, L.Antibacterial and Anti-biofilm Activity of Ginger Zingiber officinale (Roscoe)) Ethanolic Extract. Kragujevac J. Sci., 2014; 36: 129-36.
- Al-Hadi, A.H., Al-Rahbi, S.S., Akhtar, M.S., Said, S., Weli, A., Al-Riyami, Q. Phytochemical Screening, Antibacterial and Cytotoxic Activities of Petroselinum Crispum Leaves Grown in Oman. Iran. J. Pharma. Sci., 2013; 9(1): 61-5.
- Oliveira, J., Diniz, M., Lima, E., Souza, E., Trajano, V., Santos, B. Effectiveness of Origanum vulgare L. and Origanum majorana L. essential oils in inhibiting the growth of bacterial strains isolated from the patients with conjunctivitis. Braz. arch. biol. technol., 2009; 52: (1).
- Soni, K.Fluoroquinolones: Chemistry and Action-A Review. Indo Global J. Pharmaceut. Sci., 2012; 2(1): 43-53.
- Hindi, N.K.K., AL-Mahdi, Z.K.A., Chabuck, Z.A.G. Antibacterial activity of the aquatic extract of fresh, dry powder ginger, apple vinegar extract of fresh ginger and crud oil of ginger (Zingiber officinale) against different types of bacteria in Hilla City, Iraq. Inter. J. Pharm. Pharmaceut. Sci., 2014; 6(5): 414-7.
- Forbes, B.A., Sahm, D.F., Weissfeld, A.S. Bailey and Scotts’ Diagnostic microbiology. 12th ed. Elsevier. China.2007.
- NCCLS (National Committee for Clinical Laboratory Standards). Methods for dilution antimicrobial susceptibility tests of bacteria that grow aerobically. Approved Standard M100-S12. Wayne. PA, NCCLS.2002.
- Parvaiz, M., Hussain, K., Tufail, M., William,G., Shoaib, M., Jamil, M.D. EthnobotanicalSurvey of Wild Plants Used to Cure Piles in DistrictGujrat, Punjab, Pakistan. Global J.Pharmacol., 2013; 7(3): 337-41.
- Rahman, S., Sultana, S. Glycyrrhizin exhibits potential chemopreventive activity on 12-O-tetradecanoyl phorbol-13-acetate-induced cutaneous oxidative stress and tumor promotion in Swiss albino mice. J. Enzyme Inhibition Med. Chemist., 2007; 22(3): 363-9.
- Bhanuben, N., Road, V.M. Glycyrrhizaglabra(Liquorice) – a potent medicinal herb. IJHM., 2014; 2(2): 132-6.
- Sharma, V., Agrawal, R.C., Pandey, S. Phytochemical screening and determination of anti-bacterial and antioxidant potential of Glycyrrhizaglabra root extracts. J. Environ. Res. Develop., 2013; 7(4):1552-8.
- Parvaiz, M., Hussain, K., Khalid, S., Hussnain, N., Iram, N., Hussain, Z., Azhar, M. A review: medicinal importance of Glycyrrhizaglabra L. (Fabaceae family). Global J. Pharmacol., 2014; 8(1): 8-13.
- Iino, S., Tango, T., Matsushima, T. Therapeutic effects of stronger neo-minophagen C at different doses on chronic hepatitis and liver cirrhosis. Hepatol. Res., 2001; 19: 31-40.
- Khare, C.P. Encyclopedia of Indian Medicinal Plants. New York: Springer-Verlag, 2004; 233-5.
- Alonso, J., Tratado, F. www.fitoterapia.net. Barcelona: Corpus, 2004; 905-11.
- Hojoa, H., Satob, J. Antifungal Activity of Licorice (Glycyrrhizaglabra) and Potential Applications in Beverage Foods Food Ingredients J. Japan. 2002; 203.
- Shirazi, M.H., Ranjbar, R., Eshraghi, S., Sadeghi, G., Jonaidi, N., Bazzaz, N., Izadi, M., Sadeghifard, N., An Evaluation of Antibacterial Activity of Glycyrrhizaglabra Linn Extract on the Growth of Salmonella, Shigella and ETEC E. coli, J. Biolog. Sci., 2007; 7(5): 827-9.
- Gupta, V.K., Fatima, A., Faridi, U., Negi, A.S., Shanker, K., Kumar, J.K., Rahuja, N., Luqman, S., Sisodia, B.S., Saikia, D., Darokar, M.P., Khanuja, S.P. Antimicrobial potential of Glycyrrhizaglabra Linn roots. J. Ethnopharmocol., 2008; 116(2): 377-80.
- Derakhshan, S., Sattari, M., Bigdeli, M. Effect of subinhibitory concentrations of cumin (Cuminum cyminum L.) seed essential oil and alcoholic extract on the morphology, capsule expression and urease activity of Klebsiella pneumoniae. Int. J. Antimicrob. Agents, 2008; 32: 432-6.
- Singh, R.P., Gangadharappa, H.V., Mruthunjaya, K. Cuminum cyminum – A Popular Spice: An Updated Review. Pharmacogn J. 2017; 9(3): 292-301.
- Jirovetz, L., Buchbauer, G., Stoyanova, A.S., Georgiev, E.V., Damianova, S.T. Composition, quality control and antimicrobial activity of the essential oil of cumin (Cuminum cyminum L.) seeds from Bulgaria that had been stored for up to 36 years. Inter. J. Food Sci. Technol., 2005; 40: 305-10.
- Stefanini, M.B., Figueiredo, R.O., Ming, L.C., Junior, A.F. Antimicrobial activity of the essential oils of some spice herbs. Acta Horticulturae 2003; 597: 215-6.
- Chaudhary, N., Husain, S.S. Ali, M. Chemical composition and antimicrobial activity of cumin oil (Cuminum cyminum, Apiaceae). J. Pharm. Pharmaceut. Sci., 2014; 3(7): 1428-41.
- Shayegh, S., Rasooli, I., Taghizadeh, M., Astaneh, S.D. Phytotherapeutic inhibition of supragingival dental plaque. Nat. Prod. Res., 2008; 22: 428-39.
- Derakhshan, S., Sattari, M., Bigdeli, M. Effect of cumin (Cuminum cyminum L.) seed essential oil on biofilm formation and plasmid integrity by Klebsiella pneumoniae. Pharmacog. Mag., 2010; 6: 57-61.
- Hajlaoui, H., Mighri, H., Noumi, E., Snoussi, M., Trabelsi, N., Ksouri, R. Chemical composition and biological activities of Tunisian Cuminum cyminum L. essential oil: A high effectiveness against Vibrio spp. strains. Food Chem. Toxicol., 2010; 48: 2186-92.
- Zadeh, J.B., Kor, N.M. Physiological and pharmaceutical effects of Ginger (Zingiber officinale Roscoe) as a valuable medicinal plant. European J. Experiment. Biol., 2014; 4(1): 87-90.
- Mahady, G.B., Pendland, S.L., Yun, G.S., Lu, Z.Z., Stoia, A. Ginger (Zingiber officinale Roscoe) and the gingerols inhibit the growth of Cag A+ strains of Helicobacter pylori. Anticancer Res., 2003; 23(5): 3699-702.
- Miri, P., Bae, J., Lee, D.S. Antibacterial activity of [10]-gingerol and [12]-gingerol isolated from ginger rhizome against periodontal bacteria. Phytothery Res., 2008; 22: 1446-9.
- Yadav, S., Sharma, P.K., Alam, M.A. Ginger medicinal uses and benefits. Ejpmr., 2016; 3(7): 127-35.
- Suhad, A., Ahmed, I., Jabbar, I., Hamssah, E. Study the Antibacterial Activity of Zingiber officinale roots against Some of Pathogenic Bacteria. Al- Mustansiriya J. Sci., 2012; 23(3): 63-70.
- Moazedi, A.A., Mirzaie, D.N., Seyyednejad, S.M. Spasmolytic effect of Petroselinum crispum (Parsley) on rat’s ileum at different calcium chloride concentrations. Pak. J. Biol. Sci., 2007; 10(22): 4036-42.
- Behtash, N., Kargarzadeh, F., Shafaroudi, H. Analgesic effects of seed extract from Petroselinum crispum (Tagetes minuta) in animal models. Toxicol. Lett., 2008; 180(Suppl5): S127-S128.
- Aghili, M.H, Makhzan, A., Rahimi, R., Shams, A.R., Farjadmand, F. Tehran: Tehran University of Medical Sciences, 2009; 329-30.
- Savikin, K., Zdunic, G., Menkovic, N. Ethnobotanical study on traditional use of medicinal plants in South-Western Serbia, Zlatibor district. J. Ethnopharmacol., 2013; 146(3): 803-10.
- Aljanaby, A.A. Antibacterial activity of an aqueous extract of Petroselinumcrispum leaves against pathogenic bacteria isolated from patients with burns infections in Al-najaf Governorate, Iraq. Res. Chem. Intermed. 2013; 39(8): 3709-14.
- Ojala, T., Remes, S., Haansuu, P. Antimicrobial activity of some coumarin containing herbal plants growing in Finland. J Ethnopharmacol., 2000; 73(1): 299-305.
- Viuda-Martos, M., Mohamady, M.A., Fernández-López, J. In vitro antioxidant and antibacterial activities of essentials oils obtained from Egyptian aromatic plants. Food Control 2011; 22(11): 1715-22.
- Nessrien, M., Yasin, N., Mohamed, A. Antioxidant and Antimicrobial Effects of Marjoram and Thyme in Coated Refrigerated Semi Fried Mullet Fish Fillets. World J. Dairy & Food Sci., 2007; 2(1): 1-9.
- Gutierrez, J., Barry-Rya, C., Bourke, P. The antimicrobial efficacy of plant essential oil combinations and interactions with food ingredients; Inter. J. Food Microbiol., 2008; 124: 91-7.
- Freire, J.M., Cardoso, M.G., Batista, L.R. Andrade, M.A. Essentia oil of Origanum majorana L., Illicium verum Hook. F. and Cinnamomum zeylanicum. Blume, chemical and antimicrobial chracterization, Braz. J. Med. Biol. Res., 2011; 13(2): 209-14.
- Farooqi, A.A., Sreeramu, B. S. Cultivation of medicinal and aromatic crops. Universities Press, India. 2004; 465-70.
- Shahidi, B.G. Evaluation of antibacterial properties of Iranian medicinal-plants against Micrococcus luteus, Serratia marcescens, Klebsiella pneumoniae and Bordetella bronchoseptica. Asian J. Plant sciences. 2004; 3(1): 82-86.
- Leelavathi, D. In vitro morphogenetic studies in some important aromatic plants. PhD Thesis, Bangalore University 2009.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


