ISSN: 0973-7510
E-ISSN: 2581-690X
Dengue and Zika are arboviruses transmitted by the Aedes aegypti mosquito and cause high morbidity and mortality rates. This study aimed to identify epidemiological and clinical factors related to dengue and Zika in febrile patients in the Lambayeque region of Peru from July 2022 to June 2023. This was a prospective observational cross-sectional study of 403 patients. Arboviruses were diagnosed serologically by enzyme immunoassay using NS1 antigen markers and IgM antibodies against dengue and Zika viruses. Clinical and epidemiological data were collected using clinical-epidemiologic research forms. The sample was characterized by a mean age of 31 years and 46.4% male. Headache (71.7%), myalgia (57.1%), and hand arthralgia (54.1%) were the most common symptoms. The prevalence of dengue and Zika was estimated at 19.9% and 3.4%, respectively. Patients from the province of Ferreסafe (RPa = 2.15, P = 0.011), those who reported no history of the disease (RPa = 2.99, P = 0.002) and those who reported low back pain (RPa = 1.75, P = 0.015) were more likely to present dengue. Comorbidities were associated with Zika (ORa = 13.57, P = 0.012). Dengue and Zika are prevalent arboviruses in febrile patients in the Lambayeque region of Peru, where the origin, lack of history, comorbidities, and some clinical manifestations are associated with a greater likelihood of these diseases.
Dengue Fever, Zika, Epidemiological Factors, Prevalence, Febrile Illnesses (DeCS-BIREME)
Dengue, an arbovirus with a global impact, is responsible for serious conditions such as hemorrhagic fever and shock, with approximately 390 million infections per year worldwide.1 Zika is associated with complications such as microcephaly and orofacial anomalies in newborns2 and Guillain-Barré syndrome in adults.3 Both viruses are transmitted mainly by Aedes aegypti and Aedes albopictus, vectors that represent critical public health challenges in tropical and subtropical areas, whose geographic distribution has expanded due to climate change, globalization of trade, and population movements.4
In vector-borne diseases, the basic reproductive number is a measure of the epidemic potential of a disease that is mainly temperature-dependent.5 Recent studies have shown that an increase in the environmental temperature significantly increases the efficiency of infection, dissemination, and viral transmission in mosquitoes, exacerbating the problem in tropical and subtropical areas.6 Associated risk factors, such as advanced age, secondary infections, and chronic comorbidities also contribute to this increase.7 Consequently, a 20% increase in cases of dengue, Zika, and chikungunya is expected over the next 30 years due to climate change.5
In December 2023, the World Health Organization (WHO) declared the current global dengue outbreak a grade 3 emergency, the highest level of alert, with the objective of strengthening surveillance and response capacities in affected countries.8 In Peru, a particularly alarming event was recorded in 2023, where, in the first 30 weeks of the year, 222,620 cases of dengue and 381 associated deaths were reported, figures that exceeded ten times the average of the previous five years.9 The Lambayeque region of northwestern Peru has been one of the most affected areas with recurrent dengue outbreaks since its reintroduction between 2016 and 2017.10 In addition, recent studies suggest a possible association between the simultaneous circulation of dengue and Zika in this region and an increase in cases of Guillain-Barré syndrome, which reinforces the need for integrated surveillance of these two arboviruses.11
Febrile patients in endemic areas represent a priority population for epidemiological surveillance, since the initial symptoms of dengue and other arboviruses often overlap with other prevalent infections, complicating differential diagnosis.12 In the Lambayeque region, local studies have identified key risk factors such as inadequate water storage, high vector mosquito density, unfavorable socioeconomic conditions, and limited community participation in prevention strategies.13 Despite this knowledge, there are still critical gaps in our understanding of the clinical and epidemiological determinants that explain the high prevalence of these viruses in this region, limiting the design of effective interventions.
Therefore, this study aimed to identify the epidemiological and clinical factors related to dengue and Zika in febrile patients in the Lambayeque region of Peru. These findings could help optimize prevention and control strategies in line with the WHO recommendations for the integrated management of arboviruses.
Type and design of research
This was a prospective observational, cross-sectional, analytical study.
Population and sample
The study population consisted of febrile patients assisted by an institution of the Health Services Network of the Department of Lambayeque, Peru, between July 2022 and June 2023.
The sample included 403 patients, statistically calculated to estimate the proportion, considering an unknown population, a confidence level of 95%, an error of 5%, and a theoretical expected proportion of 50%. Patients were selected probabilistically using systematic sampling by recruiting every second patient according to the correlative code assigned by the Laboratorio de Referencia Regional de Salud Publica (LARESA/L), from which a copy of the clinical epidemiological form and an aliquot of blood serum were obtained for subsequent serological analyses.
Patients older than one year were included; similarly, asymptomatic patients with incomplete data and those with more than 15 days of illness were excluded according to the epidemiological clinical form. In addition, participants whose samples were hemolyzed, lipemic, in obvious microbial contamination, or in insufficient quantity (less than 500 µL) were excluded.
Data collection techniques and instruments
The outcome variables studied were prevalence of dengue and Zika. The independent variables were the epidemiological and clinical characteristics of the patients. The epidemiological variables included sex, age, pregnancy status, travel in the previous 15 days, type of IPRESS (healthcare provider institution), and occupation. The clinical variables were the time of illness, frequent signs and symptoms (headache, myalgia, arthralgia of the hands, arthralgia of the feet, lumbar pain, nausea/vomiting, rash/exanthema, and conjunctivitis), alarm signs (severe abdominal pain, chest pain or dyspnea, serous effusion, and persistent vomiting), signs of severity (weak and undetectable pulse, differential BP <20 mmHg, and severe organ involvement), and dengue classification (no alarm signs, with alarm signs, and severe).
Arboviruses were measured by serological diagnosis using the following enzyme immunoassays: NS1 antigen (Bio-Rad Laboratories, USA) and IgM antibodies (Vircell Microbiologist, Spain) against dengue and Zika. The tests were performed at the Viral Immunology Laboratory of the Lambayeque Regional Hospital, following the procedures described by a commercial company. Serum samples with indeterminate results were repeated; if the result persisted, it was considered negative.
The clinical and epidemiological data were collected using the documentation technique of the “Clinical-epidemiological research form for the surveillance of dengue, chikungunya, Zika, yellow fever and other arboviruses”, official document of the Peruvian Ministry of Health for the surveillance of these diseases. They were then recorded on an ad hoc data collection sheet.
Statistical analysis
The data were recorded on a Microsoft Excel 2019 spreadsheet, considering the variables in the columns and cases in the rows. Statistical analyses were performed using Stata.14 Descriptive analyses were performed on the outcomes and independent variables according to their nature. Relative and absolute frequencies were calculated for categorical variables, and means and deviations (parametric) or medians with interquartile ranges (non-parametric) were calculated for numerical variables.
To relate the independent variables to the prevalence of dengue and Zika, bivariate analysis was performed according to the nature of the independent variables. Categorical variables were analyzed using the chi-square (polytomous) and Fisher’s exact (dichotomous) tests, and numerical variables were analyzed using Student’s t-tests. Likewise, crude and adjusted prevalence ratios and 95% confidence intervals were calculated as measures of strength of association between the independent variables and dengue, whereas odds ratios were calculated for Zika. Confounding variables were controlled using Poisson and logit multiple regression models for adjusted prevalence ratios and adjusted odds ratios, respectively. In addition, a variance inflation factor of less than 8 was assessed for correct model fit. Statistical significance was set at P-value of less than 0.05.
Ethical considerations
This study was approved by the Ethics Committee of the hospital in charge of the serum and epidemiological forms of the study patients (0914-016-22 CEI). All collected data were securely stored by the study authors, and access was restricted to the research team only.
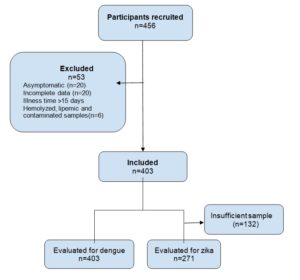
In total, 403 febrile patients from the Lambayeque region were studied (Figure). The sample showed prevalences of 19.9% and 3.4% of dengue and Zika, respectively (Table 1). It was also epidemiologically characterized by a mean age of 31 years and by 46.4% of the participants being men (Table 2). Table 3 shows the clinical characteristics, with a median of three days of illness and headache (71.7%), myalgia (57.1%), and arthralgia of hands (54.1%) as the most frequent manifestations. Table 4 shows the bivariate analysis of epidemiological and clinical characteristics and the prevalence of dengue and Zika.
Table (1):
Prevalence of dengue and Zika in febrile patients in the Lambayeque region, Peru, during July 2022 to June 2023
Arbovirosis |
N |
95% CI |
|---|---|---|
Dengue (n = 403) |
80 |
19.9 (16.0-23.8) |
Zika (n = 291) |
10 |
3.4 (1.3-5.5) |
Dengue + Zika (n = 291) |
3 |
1.0 (0.0-2.2) |
Table (2):
Epidemiological characteristics of febrile patients in the Lambayeque region, Peru, during July 2022 to June 2023 (n = 403).
| Epidemiological characteristics | N | % |
|---|---|---|
| Age (years)* | 31.7 | 18.3 |
| Sex | ||
| Male | 187 | 46.4 |
| Female | 216 | 53.6 |
| Pregnant | 19 | 4.7 |
| Trips 15 days prior | 52 | 12.9 |
| Type of IPRESS | ||
| Minsa | 334 | 82.88 |
| EsSalud | 58 | 14.39 |
| Police | 8 | 1.99 |
| Private | 3 | 0.74 |
| Occupation | ||
| Student | 108 | 26.8 |
| Homemaker | 77 | 19.1 |
| Farmer/Worker | 45 | 11.2 |
| Healthcare Professional | 20 | 5.0 |
| Commerce/Transportation | 15 | 3.7 |
| Teacher | 15 | 3.7 |
| Police Officer | 11 | 2.7 |
| Office Worker | 10 | 2.5 |
| Other | 102 | 25.3 |
| Province of origin | ||
| Chiclayo | 252 | 62.5 |
| Lambayeque | 101 | 25.1 |
| Ferreñafe | 39 | 9.7 |
| Other | 11 | 2.7 |
| Case Type | ||
| Autochthonous | 351 | 87.1 |
| Imported | 52 | 12.9 |
| History of dengue | ||
| Not | 302 | 74.9 |
| Yes | 49 | 12.2 |
| Unknown | 52 | 12.9 |
| Comorbidity | 51 | 12.7 |
| Unknown | 83 | 20.6 |
| Not | 269 | 66.7 |
| Yes | 51 | 12.7 |
| Type of comorbidity | ||
| Hypertension (n = 51) | 16 | 31.4 |
| Diabetes mellitus (n = 51) | 6 | 11.8 |
| Autoimmune disease (n = 51) | 5 | 9.8 |
| Other comorbidities (n = 51) | 16 | 31.4 |
*Mean and standard deviation; IPRESS = institution that provides health care services
Table (3):
Clinical characteristics of febrile patients in the Lambayeque region, Peru, from July 2022 to June 2023 (n = 403)
| Clinical features | N | % |
|---|---|---|
| Sick time (days)* | 3 | (2-5) |
| Common signs and symptoms | ||
| Headache | 289 | 71.7 |
| Myalgia | 230 | 57.1 |
| Arthralgia in hands | 218 | 54.1 |
| Arthralgia in feet | 176 | 43.7 |
| Eye pain | 183 | 45.4 |
| Lower back pain | 154 | 38.2 |
| Nausea/vomiting | 144 | 35.7 |
| Rash/exanthema | 42 | 10.4 |
| Conjunctivitis | 21 | 5.2 |
| Other | 58 | 14.4 |
| Warning signs | ||
| Severe abdominal pain | 31 | 7.7 |
| Chest pain or dyspnea | 29 | 7.2 |
| Serous effusion | 3 | 0.7 |
| Persistent vomiting | 16 | 4.0 |
| Other | 13 | 3.2 |
| Signs of severity | ||
| Weak, undetectable pulse | 1 | 0.2 |
| Differential BP <20 mmHg | 2 | 0.5 |
| Severe organ involvement | 1 | 0.2 |
| Dengue classification | ||
| Dengue SSA | 309 | 76.7 |
| Dengue CSA | 92 | 22.8 |
| Dengue grave | 2 | 0.5 |
*Median and interquartile range; BP = blood pressure; SSA = no alarm signs; CSA = with alarm signs
Table (4):
Association of epidemiological and clinical factors with dengue and Zika in febrile patients in the Lambayeque region, Peru, from July 2022 to June 2023
| Variables | Dengue/Total (%) | P-value | Zika/Total (%) | P-value |
|---|---|---|---|---|
| Age (years)* | 30.8-35.1 | 0.031 | 23.3-31.5 | 0.087 |
| Sick time (days) | 4.0-3.7 | 0.134 | 3.8-3.9 | 0.445 |
| Male sex | 38/187 (20.3) | 0.900 | 7/135 (5.19) | 0.196 |
| Pregnant | 5/19 (26.3) | 0.553 | 0/14 (0.0) | 0.999 |
| Province of Origin | ||||
| Chiclayo | 43/252 (17.1) | 0.006 | 8/186 (4.3) | 0.676 |
| Ferrenafe | 14/39 (35.9) | 1/28 (3.6) | ||
| Lambayeque | 18/101 (17.8) | 1/69 (1.5) | ||
| Other | 5/11 (45.5) | 0/8 (0.0) | ||
| Trips 15 days prior | 12/52 (23.1) | 0.576 | 1/41 (2.4) | 0.999 |
| History of dengue | ||||
| Not | 53/302 (17.6) | 0.078 | 8/221 (3.6) | 0.403 |
| Yes | 11/49 (22.5) | 0/35 (0) | ||
| Unknown | 16/52 (30.8) | 2/35 (5.7) | ||
| Comorbidity | ||||
| Not | 47/269 (17.5) | 0.073 | 5/203 (2.5) | 0.218 |
| Yes | 16/51 (31.4) | 3/37 (8.1) | ||
| Unknown | 17/83 (20.5) | 2/51 (3.9) | ||
| Arthralgia in hands | 50/218 (22.9) | 0.104 | 4/162 (2.5) | 0.347 |
| Arthralgia in feet | 45/176 (25.6) | 0.012 | 4/127 (3.2) | 0.999 |
| Myalgias | 54/230 (23.5) | 0.043 | 4/170 (2.4) | 0.328 |
| Headache | 64/289 (22.2) | 0.072 | 8/215 (3.7) | 0.738 |
| Eye pain | 45/183 (24.6) | 0.033 | 4/135(3.0) | 0.756 |
| Lower back pain | 45/154 (29.2) | <0.001 | 3/111(2.7) | 0.746 |
| Nausea/vomiting | 31/144 (21.5) | 0.602 | 3/105 (2.9) | 0.753 |
| Rash/exanthema | 13/42 (31.0) | 0.066 | 2/31 (6.5) | 0.607 |
| Conjunctivitis | 5/21 (23.8) | 0.778 | 1/15 (6.7) | 0.999 |
*Mean infected-uninfected patients; Student’s P-value. **Fisher Exact for dichotomous variables and Chi-square for polytomous variables
Table 5 shows the measures of the strength of association between the factors and dengue, where the origin, history of dengue, and manifestation of low back pain were associated with a greater likelihood of suffering from the disease (P < 0.050). Indeed, patients from the province of Ferrenafe had a 115% higher likelihood of dengue than those from Chiclayo (RPa = 2.15, P = 0.011). Also, those patients who stated that they were unaware of a history of dengue fever were 199% more likely to present it, compared to those who stated that they had no history (RPa = 2.99, P = 0.002). Similarly, patients who reported low back pain as a clinical manifestation were 75% more likely to have the disease (RPa = 1.75, P = 0.015).
Table (5):
Strength of association between epidemiological and clinical factors with dengue in febrile patients in the Lambayeque region, Peru, from July 2022 to June 2023
| Variables | Simple regression | Multiple regression | ||
|---|---|---|---|---|
| PRc (95% IC) | P-value | PRa (95% IC) | P-value | |
| Age (years) | 1.01 (1.00-1.02) | 0.039 | 1.00 (0.99-1.02) | 0.393 |
| Province of Origin | ||||
| Chiclayo | Ref. | Ref. | ||
| Ferreסafe | 2.10 (1.28-3.47) | 0.004 | 2.15 (1.19-3.89) | 0.011 |
| Lambayeque | 1.04 (0.63-1.72) | 0.865 | 0.92 (0.55-1.54) | 0.747 |
| Other | 2.66 (1.32-5.38) | 0.006 | 1.52 (0.77-3.03) | 0.231 |
| History of dengue | ||||
| Not | Ref. | |||
| Yes | 1.28 (0.72-2.27) | 0.402 | 1.15 (0.65-2.03) | 0.632 |
| Unknown | 1.75 (1.09-2.82) | 0.021 | 2.99 (1.48-6.05) | 0.002 |
| Comorbidity | ||||
| Not | Ref. | |||
| Yes | 1.80 (1.11-2.91) | 0.017 | 1.59 (0.93-2.71) | 0.092 |
| Unknown | 1.17 (0.71-1.93) | 0.531 | 0.50 (0.24-1.05) | 0.066 |
| Arthralgia in hands | 1.41 (0.94-2.13) | 0.096 | 0.79 (0.46-1.35) | 0.379 |
| Arthralgia in feet | 1.66 (1.11-2.46) | 0.012 | 1.49 (0.88-2.55) | 0.140 |
| Myalgias | 1.56 (1.02-2.39) | 0.040 | 1.08 (0.66-1.78) | 0.753 |
| Headache | 1.58 (0.95-2.61) | 0.076 | 1.21 (0.69-2.12) | 0.509 |
| Eye pain | 1.55 (1.04-2.30) | 0.031 | 1.16 (0.71-1.89) | 0.551 |
| Lower back pain | 2.08 (1.40-3.08) | <0.001 | 1.75 (1.12-2.75) | 0.015 |
| Rash/exanthema | 1.67 (1.01-2.75) | 0.046 | 1.42 (0.83-2.40) | 0.198 |
Table 6 shows the measures of the strength of association between the factors and Zika, showing that patients with some comorbidities were more than 13 times more likely to present Zika, compared to those who did not report comorbidities (ORa = 13.57, P = 0.012).
Table (6):
Strength of association between epidemiological and clinical factors with Zika in febrile patients in the Lambayeque region, Peru, from July 2022 to June 2023
| Variable | Simple regression | Multiple regression | ||
|---|---|---|---|---|
| ORc (95% IC) | P-value | ORa (95% IC) | P-value | |
| Age (years) | 0.97 (0.93-1.01) | 0.166 | 0.96 (0.92-1.02) | 0.169 |
| Sex | ||||
| Female | 0.36 (0.09-1.41) | 0.143 | 0.27 (0.05-1.43) | 0.123 |
| Male | Ref. | Ref. | ||
| Comorbidity | ||||
| Not | Ref. | Ref. | ||
| Yes | 3.5 (0.80-15.30) | 0.097 | 13.57 (1.79-102.65) | 0.012 |
| Unknown | 1.62 (0.30-8.58) | 0.573 | 0.76 (0.04-16.55) | 0.860 |
| Arthralgia in hands | 0.52 (0.14-1.88) | 0.318 | 0.96 (0.75-1.23) | 0.754 |
| Arthralgia in feet | 0.86 (0.24-3.10) | 0.813 | 4.8 (0.46-50.15) | 0.190 |
| Rash/exanthema | 2.17 (0.44-10.72) | 0.341 | 4.53 (0.60-34.37) | 0.144 |
Model adjusted for age, sex, origin, history, comorbidities and clinical manifestations (hand arthralgia, foot arthralgia, myalgia, headache, eye pain, low back pain, nausea/vomiting, rash/exanthema and conjunctivitis). Average variance inflation factor = 2.4
Model adjusted for age, sex, origin, history, comorbidities and clinical manifestations (hand arthralgia, foot arthralgia, myalgia, headache, eye pain, low back pain, nausea/vomiting, rash/exanthema and conjunctivitis).
The most frequent epidemiological and clinical characteristics of the febrile patients studied were an average age of 31 years, female sex, IPRESS of MINSA and origin from the province of Chiclayo, autochthonous cases, no history of dengue and no comorbidities, clinical illness between 2 and 5 days, headache, myalgia, and arthralgia. These characteristics are consistent with a local study in 2017, which reported similar characteristics.14 Therefore, it is likely that the characteristics of this population are mainly defined by the climatic (El Niño Costero), geographic, and cultural conditions that cause arbovirosis.
The prevalence of dengue observed in febrile patients in this study was 19.9%. These results, although relevant, were lower than those reported for other regions with high endemicity. Thus, a study of febrile patients in a Colombian hospital revealed that 51% presented dengue, 44% of whom corresponded to secondary infections.15 Brazil has also historically documented a high burden of disease, accounting for up to 60% of all cases reported worldwide.16 These differences can be explained by the geographical, climatic, and vector density characteristics. However, in Peru, areas such as Madre de Dios had a prevalence similar to that reported in Colombia and Brazil, given its status as an Amazonian region with active transmission.17 Furthermore, in the Lambayeque region, where this study was conducted, a prevalence of 28.1% has been previously documented in 2017,10 a figure that supports the prevalence of the virus in a range close to that currently detected.
Regarding Zika virus infection (3.4%), the prevalence was lower compared to that reported for other countries in the region. In Nicaragua, during the 2016 outbreak, seropositivity of 36% was observed in children, 46% in households, and 56% in adults.18 Similarly, in the general population, a review estimates that the average seroprevalence of Zika in the Americas was approximately 40%.19 These findings reflect considerable exposure to the virus in urban and rural contexts of high transmission, much higher than that observed in the Lambayeque region, mainly because in this study, active infection was sought through IgM detection, unlike previous studies that focused on seroprevalence (IgG). However, the low prevalence of Zika in Lambayeque should be actively monitored because of its epidemic potential in the context of co-circulation with other arboviruses.
In terms of age, most patients with positive serological markers for dengue were between 30.8 and 35.1 years old (P = 0.031), whereas for Zika, they were between 23.3 and 31.5 years old (P = 0.087, marginally not significant). These age groups belong to a young economically active population, in which intense physical activity may be related to the growth of bacteria that generate metabolites attractive to mosquitoes.20 CO2 emission is a factor that influences the attraction of mosquitoes, and children have lower emissions of CO2, which explains why more cases are observed in adults in the intradomiciliary environment.21 In addition, active metabolism in adults generates greater body heat and promotes the growth of metabolite-producing bacteria that are attractive to mosquitoes.21 However, a systematic review that evaluated the epidemiological impact of dengue in Colombia found that 50% of the cases occurred in children under 20 years of age, with the highest incidence in the age group of 5-14.22
Low back pain was the only clinical manifestation that showed an association with dengue. Although other classic symptoms, such as myalgia, eye pain, and exanthema, showed a possible association in the bivariate analysis, they lost statistical significance in the multivariate analysis, suggesting less diagnostic utility when other clinical or epidemiological variables were considered. The prevalence of low back pain as a clinical manifestation of dengue has also been previously reported in Peru: 65.9% in Amazonas,23 73% in La Libertad,24 and 74.8% in Lambayeque.14 These findings highlight the high frequency of this symptom in patients with dengue, reinforcing its value as a potentially relevant clinical marker in the context of epidemics.
This study identified a significant association between certain epidemiological factors and dengue infection, in particular, origin from Ferreסafe. Indeed, a high frequency of cases in this province has been reported previously25 and could be explained by environmental factors, urbanization conditions, variations in vector density, and sociocultural factors. In this regard, in Pueblo Nuevo, one of the most affected districts in the province, a marked community resistance to fumigation campaigns was reported, with only 47.4% of families allowing health personnel to enter their homes,26 which limits vector control actions and favors transmission.
Finally, this study revealed active circulation of dengue and Zika viruses in the Lambayeque region, in addition to a co-infection rate of 1.0%. These values are lower than those reported in highly endemic regions of Latin America, but confirm the persistence of epidemiological risks in this area of northern Peru.19 Cases of dengue and Zika together account for 23.4% of the febrile cases, accounting for only a quarter of the febrile cases, which can be explained by other prevalent febrile diseases such as leptospirosis, which has been reported to account for more than 26% of the febrile population in Lambayeque.27
As for Zika, only comorbidities were found to be associated with the highest prevalence, probably due to the increased susceptibility of the person to these conditions. In this regard, other Latin American studies have reported that comorbidities such as diabetes, hypertension, and sickle cell anemia were associated with severe disease and a fatal outcome due to this virus.28
The study had some limitations: probable information bias in the completion of the clinical-epidemiological forms and probable measurement bias in the detection of Zika by serological methodology, since the commercial ELISA kit used has not been standardized for use in the Peruvian population. However, the observed results are plausible in the existing theory and can be generalized to the febrile population of the Lambayeque region because of the representative sample size. Potential confounding factors were controlled in the association analyses.
It was concluded that dengue and Zika are prevalent arboviruses in febrile patients in the Lambayeque region of Peru, where the province of origin, unawareness of history, and low back pain were associated with a greater possibility of having dengue, whereas comorbidities were associated with Zika. This finding suggests the influence of environmental, urban and sociocultural variables, such as low acceptance of vector control measures, on local transmission dynamics. Overall, these results reinforce the need to strengthen epidemiological surveillance, promote sustained community interventions, and improve prevention and differential diagnostic strategies, especially in the context of arbovirus co-circulation.
ACKNOWLEDGMENTS
The authors are thankful to the Laboratorio de Referencia Regional de Salud Pública de Lambayeque for allowing access to the samples and clinical-epidemiological records.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
This study was supported by the University of San Martín de Porres, with code number E21102023010, and Cesar Vallejo University and Lambayeque Regional Hospital (non-monetary contribution).
DATA AVAILABILITY
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
ETHICS STATEMENT
This study was approved by the Ethics Committee, Lambayeque Regional Hospital (0914-016-22 CEI).
INFORMED CONSENT
Written informed consent was obtained from the participants before enrolling in the study.
- World Health Organization: WHO. Dengue. Published August 21, 2025. https://www.who.int/news-room/fact-sheets/detail/dengue-and-severe-dengue. (Accessed June 10, 2025)
- Scotto G, Massa S, Spirito F, Fazio V. Congenital Zika Virus Syndrome: Microcephaly and Orofacial Anomalies. Life. 2023;14(1):55.
Crossref - Beattie J, Parajuli S, Sanger M, et al. Zika Virus–Associated Guillain-Barre Syndrome in a Returning US Traveler. Infect Dis Clin. 2018;26(6):e80-e84.
Crossref - Lwande OW, Obanda V, Lindstrom A, et al. Globe-Trotting Aedes aegypti and Aedes albopictus: Risk Factors for Arbovirus Pandemics. Vector Borne Zoonotic Dis. 2020;20(2):71-81.
Crossref - Wyk HV, Eisenberg JNS, Brouwer AF. Long-term projections of the impacts of warming temperatures on Zika and dengue risk in four Brazilian cities using a temperature-dependent basic reproduction number. PLoS Negl Trop Dis. 2023;17(4):e0010839.
Crossref - Delrieu M, Martinet JP, O’Connor O, et al. Temperature and transmission of chikungunya, dengue, and Zika viruses: A systematic review of experimental studies on Aedes aegypti and Aedes albopictus. Curr Res Parasitol Vector-Borne Dis. 2023;4:100139.
Crossref - Liu LT, Huang SY, Lin CH, et al. The epidemiology and identification of risk factors associated with severe dengue during the 2023 dengue outbreak in Kaohsiung City, Taiwan. Travel Med Infect Dis. 2025;65:102852.
Crossref - Pan American Health Organization; World Health Organization. Dengue Multi-Country Grade 3 Outbreak. PAHO/WHO. Published online. https://www.paho.org/en/topics/dengue/dengue-multi-country-grade-3-outbreak. (Accessed June 10, 2025).
- Munayco CV, Rosales BYV, Lizarbe SYM, et al. Notes from the Field: Dengue Outbreak — Peru, 2023. Morb Mortal Wkly Rep. 2024;73(4):86-88.
Crossref - Coronado PET, Aguilar-Gamboa FR, Guevara-Vásquez G. Características epidemiológicas y serológicas de los pacientes con dengue probable, en un hospital de Lambayeque, Perú [Epidemiological and serological characteristics of patients with probable dengue, in a Lambayeque hospital, Peru]. Rev Exp Med. 2019;5(1):13-17.
Crossref - Aguilar-Gamboa FR. El Síndrome de Guillain-Barré en Perú: ¿Dengue y Zika detrás del aumento de casos?[Guillain-Barré Syndrome in Peru: Are Dengue and Zika behind the increase in cases?]. Rev Exp Med. 2023;9(3):123-127.
Crossref - Mayo Clinic. Dengue fever. https://www.mayoclinic.org/diseases-conditions/dengue-fever/diagnosis-treatment/drc-20353084. (Accessed June 10, 2025).
- Copaja-Corzo C, Santana-Tellez TN. Water management and reduction of Aedes Aegypti reservoirs: An unresolved public health problem in Peru. Rev Cuerpo Med Hosp Nac Almanzor Aguinaga Asenjo. 2023;16(1):174-175.
Crossref - Bravo Acuna KF, Paredes Huancas LM. Perfil clínico, epidemiológico y laboratorial del dengue en un centro de salud de Lambayeque, 2023 [Clinical, epidemiological, and laboratory profile of dengue fever in a health center in Lambayeque, 2023]. Universidad Senor De Sipan. 2024. https://repositorio.uss.edu.pe/handle/20.500.12802/12561. Accessed 10 Jun, 2025
- Sanchez Lerma L, Perez Gutierrez N, Pavas Escobar NC. Dengue: a frequent cause of acute febrile syndrome in the pediatric population. Rev Habanera Cienc Medicas. 2017;16(2):256-267.
- Fares RCG, Souza KPR, Anez G, Rios M. Epidemiological Scenario of Dengue in Brazil. BioMed Res Int. 2015;2015: 321873.
Crossref - Garcia MP, Padilla C, Figueroa D, Manrique C, Cabezas C. Emergencia del genotipo Cosmopolitan del virus dengue serotipo 2 (DENV2) en Madre de Dios, Perú, 2019. Revista Peruana De Medicina Experimental Y Salud Pública. 2022;39(1):126-128.
Crossref - Zambrana JV, Carrillo FB, Burger-Calderon R, et al. Seroprevalence, risk factor, and spatial analyses of Zika virus infection after the 2016 epidemic in Managua, Nicaragua. Proc Natl Acad Sci USA. 2018;115(37):9294-9299.
Crossref - Villarroel PMS, Hamel R, Gumpangseth N, et al. Global seroprevalence of Zika virus in asymptomatic individuals: A systematic review. PLoS Negl Trop Dis. 2024;18(4):e0011842.
Crossref - Liu F, Coutinho-Abreu IV, Raban R, et al. Engineered skin microbiome reduces mosquito attraction to mice. PNAS Nexus. 2024;3(7): 267.
Crossref - Romero S. Descubre si eres un blanco fácil para las picaduras de mosquitos [Find out if you’re an easy target for mosquito bites. Very interesting]. 2024. https://www. muyinteresante.com/ciencia/29043.html. Accessed 10 Jun, 2025.
- Rodriguez-Morales AJ, Lopez-Medina E, Arboleda I, et al. The Epidemiological Impact of Dengue in Colombia: A Systematic Review. Am J Trop Med Hyg. 2025;112(1):182-188.
Crossref - Cumpen Silva J, Llamo Sullon M, Sanchez Tejada E, et al. Prevalence of dengue in patients treated at the José Olaya health center from February to June 2017. Salud Vida Sipanense. 2017;4(1):9-14. https://revistas.uss.edu.pe/index.php/SVS/article/view/686. Accessed 10 Jun, 2025.
- Ruiz Chang WBClinical characterization of patients with dengue from the District Hospital Santa Isabel – El Porvenir and the Hospital District Laredo – Laredo, referring to Regional Reference Laboratory of La Libertad, Perú – 2019. Arnaldoa. 2020;27(1):237-246. http://www.scielo.org.pe/pdf/arnal/v27n1/2413-3299-arnal-27-01-237.pdf. Accessed 10 Jun, 2025.
- Municipalidad Provincial de Ferreñafe Situacional de dengue en la provincia [Provincial Municipality of Ferrenafe. Situational of dengue in the province] https://www.gob. pe/institucion/muniferrenafe/noticias/877151- situacional-de-dengue-en-la-provincia. Accessed 10 Jun, 2025
- Gerencia Regional de Salud Lambayeque Solo el 47.36 % de viviendas en Ferreñafe acceden a intervenciones contra el dengue [Lambayeque Regional Health Management. Only 47.36% of homes in Ferreñafe have access to dengue interventions]. 2024. https://www.gob.pe/institucion/regionlambayeque-geresa/noticias/923046-solo-el-47-36-de-viviendas-en-ferrenafe-acceden-a-intervenciones-contra-el-dengue. Accessed 10 June, 2025.
- Silva-Diaz H, Llatas-Cancino DN, Campos-Sanchez MJ, et al. Leptospirosis frequency and socio-demographic characteristics associated in febrile patients from northern Perú. Rev Chil Infectol. 2015;32(5):530-535.
Crossref - Sarmiento-Ospina A, Vasquez-Serna H, Jimenez-Canizales CE, et al. Zika virus associated deaths in Colombia. Lancet Infect Dis. 2016;16(5):523-524.
Crossref
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.