ISSN: 0973-7510
E-ISSN: 2581-690X
This project was carried out to study the effect of five different doses of electron-beam irradiation, including: 0 as control, 1.5, 3.5, 5.5 and 7.5 kGy on peroxidase (POD), superoxide dismutase (SOD) and polyphenol oxidase (PPO) activities of brown button mushroom (Agaricus brunnescens). The experiment was conducted using an electron beam accelerator (ESS-010-03) and measurements were made during 1, 4th, 8th, 12th and 16th day storage at 4 °C and 80 percent relative humidity. There was a significant difference between irradiated and non-irradiated (control) mushrooms in different enzymes (P<0.01). Most Peroxidase activity was observed in mushrooms treated with 7.5 kGy. The irradiated mushroom with 7.5 kGy also showed the highest SOD activity. The lowest SOD activity in mushrooms was related to control in all days of storage. The mushrooms irradiated with 0 and 7.5 kGy contained higher and lower PPO activity using pyrocatechol as substrate respectively, during 12th and 16th day compared with other doses. Treatment of 0 kGy induced the highest PPO activity using pyrogallol as substrate in storage days. The data increased the current understanding of the effects of electron-beam irradiation on the enzyme activity changes associated with postharvest senescence and should lead to more targeted strategies for reducing postharvest quality loss in A. brunnescens.
Agaricus brunnescens, Electron-beam, Irradiation, Enzyme activity and postharvest
The button mushroom is the most widely cultivated and consumed mushroom throughout the world and includes about 40 percent of total world mushroom production (Giri and Prasad., 2007). The production and fresh use of white and brown button mushroom in Iran have increased rapidly during the last decade. However, the high perishable nature of mushrooms remains a problem for the progress of this industry (Beaulieu, Lacroix, Charbonneau and et al., 1992; Gautam, Sharma and Thomas., 1998). A potentially attractive procedure to extend the shelf-life of mushrooms is exposure to ionizing radiation, and previous papers have suggested this method is highly effective in inhibiting physical changes associated with postharvest deterioration and maintaining a fresh product appearance (Kader., 1986). Food processing by employing radiation is well established as a physical, non-thermal mode of food preservation that processes foods at or nearly at ambient temperature. Irradiation of food products causes minimal modification in the flavor, color, nutrients, taste, and other quality attributes of food. However, the levels of modification (in flavor, color nutrients, taste etc.) might vary depending on the basic raw material used, irradiation dose delivered, and on the type of radiation source employed (gamma, X-ray, UV, electron beam) (Bhat and Sridhar., 2008; Bhat, Sridhar and Yokotani., 2007; Mexis, Badeka, Chouliara et al., 2009). Electron beams are produced from machines capable of accelerating electrons to near the speed of light (~190,000 miles/second). This electron beam generator uses commercial electricity as an energy source and can be simply switched on or off. Compared with gamma rays or X-rays, the electron beam is limited to treating relatively thin packages, because of the low penetrating power (< 2 inches) of electrons (Park and Vestal., 2002). Electron-beam could be applied to fresh produce to improve microbial safety while simultaneously maintaining their “fresh” characteristics. International agencies including IAEA, FAO and WHO concluded that irradiation of any food commodity up to a dose of 10 kGy exhibits no health risks (WHO., 1981; Diehl., 2002). The enzymes are responsible for different changes in chemical constituents that would affect the quality characteristics and deterioration of mushroom either fresh or processed (Ogawa and Uritani., 1970). Fungi display several antioxidant enzymes against ROS, including superoxide dismutase (SOD), catalase (CAT), and peroxidases (POD) and glutathione reductases, capable of removing oxygen radicals and their products and/or repairing oxidative damage (Jamieson., 1998; Bai, Harvey, and Mcneil., 2003). The native PPO is a tetramer described as an H2L2 structure; where the H subunits have molecular weights of 45–55KDa and the L subunits 13–15Kda (Robb., 1984). with no disulfide linkages between subunits (Robb and Gutteridge., 1981). The polyphenol oxidase (PPO) present in the pileus (cap) and stipe (stalk) of mushrooms plays also an important role. PPO is a copper containing enzyme (Vamos-Vigyazo and Haard., 1981), which catalyzes two different reactions: (i) the hydroxylation of monophenols to the corresponding o-dihydroxy compounds and (ii) the oxidation of o-dihydroxy phenols to o-quinones, which condense to form the brown melanin pigments (Long, J.T., Alben., 1969; Stussi, Rats., 1981). Browning on the mushrooms tissue can be a consequence of enzymatic activity and/ or microbial infection (Royse and Wuest, 1980). Enzymatic browning in fruit and vegetables has been postulated by many authors as the action of peroxidase (POD) and polyphenol oxidase (PPO) activity (Jolivet, Arpin, Wichers et al., 1998; Nerya, Ben-Arie, Luzzato et al., 2006). Although both POD and PPO activities are present in mushrooms (Zhang and Flurkey., 1997) most researchers agree that the copper containing enzyme tyrosinase, of the PPO group, is largely responsible for enzymatic discoloration in mushrooms (Nerya, Ben-Arie, Luzzato et al., 2006). Gamma irradiation has also been shown to reduce polyphenol oxidase (PPO) activity, and respiration, and browning, and extend consumer acceptability (Beaulieu, Lacroix, Charbonneau et al., 1992; Benoit, Aprano and Lacroix., 2000). The aim of this investigation was mainly directed to the effect of different doses of electron-beam irradiation on enzyme activities of brown button mushroom.
Study site
The experiments were conducted in the Faculty of Agricultural, University of Guilan; Rasht and Yazd Radiation Processing Center (YRPC), Yazd, Iran, in 2011-2012. The experiment was set up in a Split Plot in Time (SP-T) design with three replications.
Mushroom samples
Freshly harvested, mature sporophores of Agaricus brunnescens of similar size and free from physical defects were obtained from a commercial mushroom-growing operation (Mehriz) located near Yazd. Immediately after harvesting, fruit bodies were packed into polystyrene trays (20 × 10 × 1 cm), covered with polyethylene film, stored at 4 °C and transported to the irradiation center of the Yazd Radiation Processing Center. Time of harvest until the start of irradiation was approximately 2 h. The temperature range of mushrooms during this 4 h period was 4 °C to 6 °C. Each replication was containing 10 trays with 200g in weight.
Irradiation
Fruit bodies (200 g) were placed in plastic trays and irradiated at 20 °C. Irradiation was carried out using an electron beam accelerator (ESS-010-03 electron linear accelerator).The samples were irradiated under various dose intervals of 0.5, 1, 2 and 4 kGy (Table 1). The dosimetry was made using the Fricke reference standard dosimetry system (Holm and Berry., 1970). During irradiation and transportation, the temperature of mushrooms was maintained between 4 °C and 6 °C. Measurements were made during 1, 4th, 8th, 12th and 16th day for the mushrooms stored continuously at 4 °C and 80 percent relative humidity. Ten mushrooms were analyzed from each treatment.
Fruit body extract
Frozen A. brunnescens tissue (0.5 g) was ground in a cold mortar and pestle with 10 ml of 0.05 mol/l phosphate buffer (pH 7)containing polyvinylpyrrolidone (0.1 g/ml)and EDTA (0.1 mol/L). Homogenates were centrifuged twice (14000 rpm, 15 min, 4 °C), and the supernatants were used for enzyme assays.
Peroxidase assay
POD activity was measured spectrophotometrically (model PG Instrument + 80, (Leicester, UK), using the substrate guaiacol (Moerchbacher, Noll, Flott et al., 1988). The reaction mixture for determination of POD activityconsisted of 50 mmol/l sodium phosphate buffer (pH 6.0), 5 mmol/l guaiacol, 5 mmol/l H2O2, and 50 µl of tissue extract. One time of POD activity was defined as the amount of enzyme that caused a change in absorbance at 470 nm of 0.01min1.
Superoxide dismutase assay
Superoxide dismutase (SOD) activity was determined by measuring the inhibition of the photochemical reduction of nitroblue tetrazolium (NBT) according to the method of Constantine and Stanly (1977). Reaction mixtures contined (in a total volume of 3 ml): 13 mmol/l riboflavin, and 0.1 ml enzyme extract in 50 mmol/l phosphate buffer (pH 7.8). One unit of SOD activity was defined as the amount of enzyme required to inhibit the rate of NBT photoreduction by 50 percent, and SOD activity values are presented as U/g Fw of mushroom.
Polyphenol oxidase assay
Polyphenol oxidase (PPO) activity was assayed by measuring the liner increase in absorbance at 410 nm and 30 °C as described by Galeazzi, Sgarbieri and Constantinides (1981) using catechol and pyrogallol as the substrate. Reaction mixtures contained 2.0 ml of 50 mmol/l phosphate buffer (pH 7), catechol (1, 2-dihydroxybenzene) or pyrogallol (PYGL, 1, 2, 3-trihydroxybenzene) (20 mg/ml) and 0.2 ml extract added to initiate the reaction. One unit (U) of PPO activity was defined as the amount of enzyme catalyzing an increase in absorbance at 410 nm of 0.01/min, using an UV/Vis spectrophotometer model PG Instrument + 80, (Leicester, UK). PPO activity values are presented as U/min/g Fw (Fresh weight) of mushroom.
Effect of electron-beam irradiation on peroxidase (POD) activities
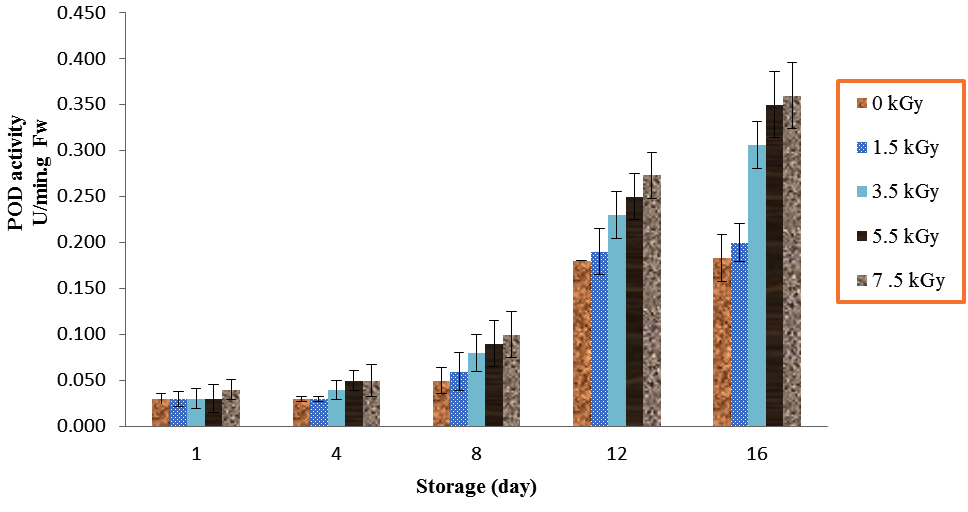
The results showed significant differences for POD activity (Table 1). Peroxidase activities of mushrooms were increased significantly by application of 1.5, 3.5, 5.5 and 7.5 irradiation as compared with control over 8, 12 and 16 days storage (P<0.01). In addition, the mushrooms which treated with 7.5 kGy had relatively higher POD activities at all storage periods (Fig. 1). However, the mushrooms treated with 7.5 kGy had no significance different with doses of 5.5 kGy (P<0.01).
Fig. 1. Effect of Electron-Beam Irradiation on POD Activity in Agaricus Brunnescens fruit Bodies During Storage at 4 °C. Vertical Bars Represent the Standard Deviation about the Mean (r = 3)
Table (1):
Analysis of variance (ANOVA) effects due to irradiation with five different doses (0, 1.5, 3.5, 5.5 and 7.5 kGy) on peroxidase (POD), super oxidase (SOD) and polyphenol oxidase (PPO).
Source |
df |
POD (U/g F.w) |
SOD (U/g F.w) |
PPO (pyrocatechol) (U/min.g F.W) |
PPO (pyrogallol) (U/min.g F.W) |
|---|---|---|---|---|---|
A |
4 |
00.013** |
21.78** |
0.12** |
288.999** |
Error (a) |
10 |
0.0008 |
0.036 |
0.0001 |
0.164 |
B |
4 |
0.196** |
522.517** |
0.089** |
9701.164** |
A×B |
16 |
0.003** |
2.978** |
0.002** |
41.931** |
B |
8 |
0.0009 |
0.025 |
0. 0005 |
0. 12 |
CV |
– |
2.12 |
1.64 |
9.75 |
1.21 |
** significant at P<0.01, ANOVA
CV, coefficient of variation; df, degrees of freedom; FW, fresh weight
Effect of electron-beam irradiation on superoxide dismutase (SOD) activities
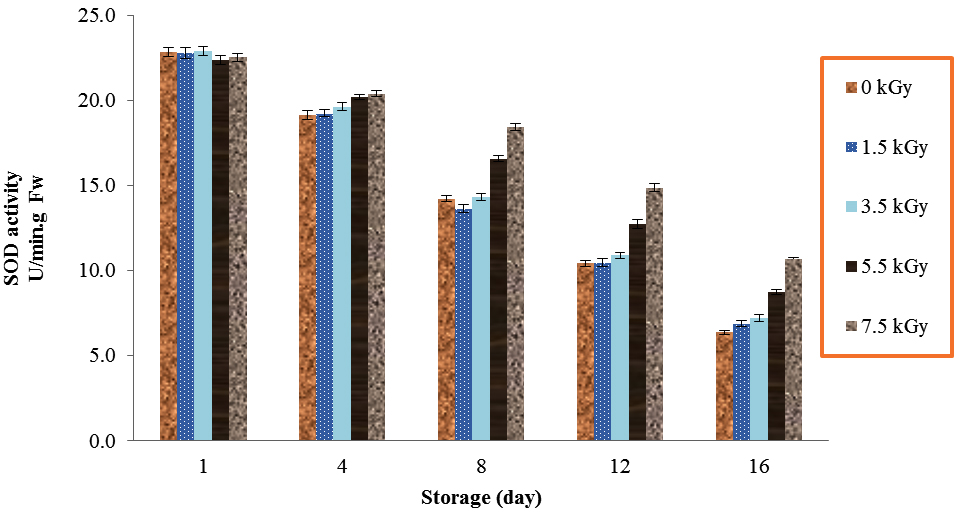
The amount of SOD activities was different during storage times by irradiation. Electron beam irradiation significantly affected on SOD activities of button mushroom (Table 1). The amounts of SOD continually decreased from day 1 to day 16. The highest activity in storage times of 4, 8, 12 and 16 in mushrooms was due to the use of 7.5 kGy (Fig. 2). At any of the time periods during the study, significant difference was observed between irradiated and non-irradiated mushrooms in SOD activities, except day 1 (P<0.01).
Fig. 2. Effect of Electron-Beam Irradiation on SOD Activity in Agaricus Brunnescens Fruit Bodies During Storage at 4 °C. Vertical Bars Represent the Standard Deviation about the Mean (r = 3)
Effect of electron-beam irradiation on polyphenol oxidase (PPO) activities
Catechol as the substrate
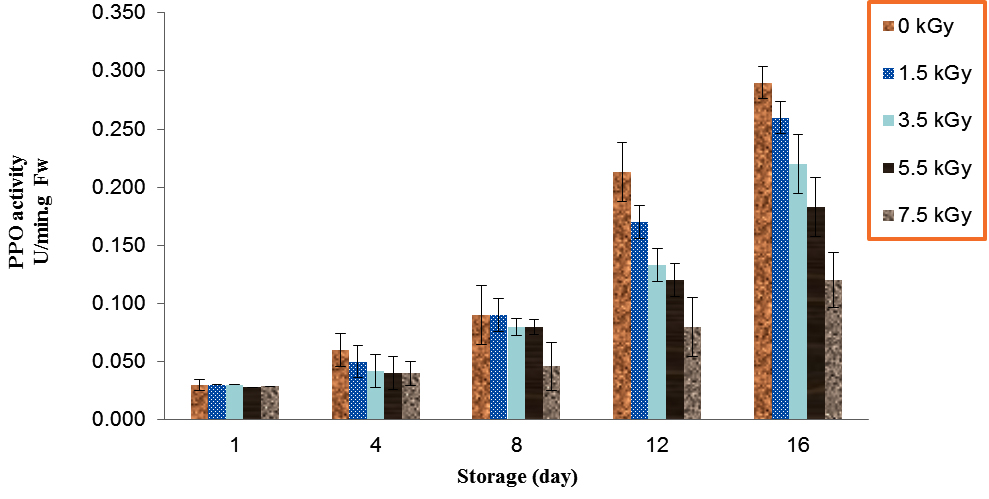
During the storage, increases of the mushroom PPO activities were observed for all mushrooms, control and treated (Table 1). There was significant differences for PPO activity in irradiated and control button mushroom after 12 days of storage (P<0.01). The mushroom which irradiated with 7.5 kGy had the lowest PPO activity as compared to treatments over 12 days storage, also treatment of control showed the higher PPO activity during storage at 4 °C. PPO activity in control (0 kGy) and dose of 7 kGy increased from 0.03 and 0.02 on day 1 to 0.29 and 0.12 on day 16 (Fig. 3).
Fig. 3. Effect of Electron-Beam Irradiation on PPO Activity Using Pyrocatechol as Substrate in Agaricus Brunnescens Fruit Bodies During Storage at 4 °C. Vertical Bars Represent the Standard Deviation about the Mean (r = 3)
Pyrogallol as the substrate
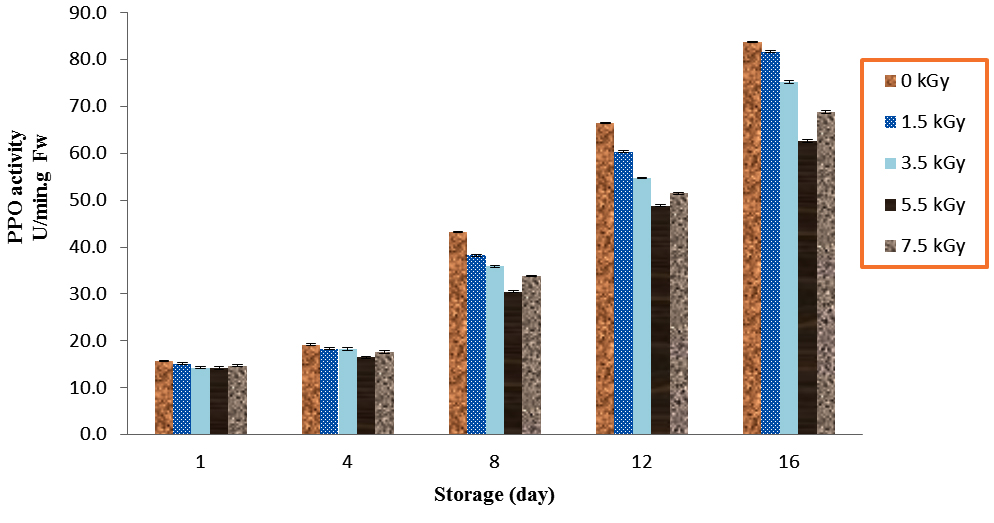
The mushroom which irradiated with 5.5 kGy had the lowest PPO activities as compared to treatments in all day’s storage at 4 °C. However, non-significant differences observed between 5.5 kGy and 7.5 kGy doses during storage times (P<0.01). The amount of PPO activities in control and 5.5 kGy mushrooms increased from 15.7 and 14.3 on day 1 to 83.8 and 62.7 on day 16, respectively (Fig. 4).
Peroxidase (POD) among antioxidant enzymes plays an important role of hydrogen peroxide (H2O2) detoxification in cells, thereby protecting cellular components such as proteins and lipids against oxidation (Wi, Chung and Kim., 2006). The PODs are also requires essentially for a variety of cellular functions such as lignification, suberization, cell elongation, growth, regulation of cell wall, biosynthesis and plasticity (Chanda and Singh., 1997). In the study by Wi, Chung and Kim (2006), the induction of POD by irradiation would be one of the defense systems activated through the ROS-mediated cellular signaling. As observed in the present study, it was suggested that the increase in electron beam doses corresponded to an increase in specific activity of peroxidase. Kiong, Lai, Hussein et al. (2008), showed the highest amount of specific activity of peroxidase was obtained in plantlets irradiated at 50 Gy. Qin, Wang, Wang et al. (2000) noticed a change in the peroxidase activity in Lathyrus sativus plants after treatment of seeds with gamma ray and EMS. Enhancement in peroxidase activity by radiation has also been reported by Omar (1988) in sunflower. It has been indicated that irradiation enhanced peroxidase activity of two Phaseolus vulgaris cultivars (Strid, Chow, Anderson., 1990). Evidence indicated that the stimulation of peroxidase development by radiation may be partly mediated through the enhanced amounts of ethylene produced by radiation, but it is mainly mediated by another unknown mechanism (Ogawa and Uritani., 1970). Moussa and Abdel-Aziz. (2008) indicated that increase in POD activity under various stress conditions has been linked with protection from oxidative damage, lignifications and cross-linking of cell wall to prevent from such adverse conditions.
Stress effect all organisms and responses to stress have been shown to be controlled at the molecular level. In white button mushroom, detachments of the mushroom and post-harvest storage are likely to induce stress. Following harvest, the mushroom continues to develop though is subject to a number of stresses besides wounding including nutritional and water deprivation. This response to water and nutrient limitation in the harvested sporophore has been termed a “post-harvest stress disorder” (Moore., 1988). Post-harvest conditions are associated with a number of physiological, molecular and biochemical changes which effect consumer quality. Harvesting is itself a wounding event that is accompanied by a massive disruption in metabolism. The isolated sporophore continues to develop similarly to the non-harvested fruit body (Hammond, Nichols., 1975). SOD has been associated with stress tolerance in button mushroom, and the gene encoding the enzyme is upregulated in postharvest sporophores (Amiot, Flueriet, Cheynier et al., 1997). Superoxide dismutase serve to neutralize the destructive effects of reactive oxygen species on cellular component by converting superoxide anions into hydrogen peroxide and are thought to alleviate postharvest deterioration by maintaining membrane integrity. The results indicate that high-doses electron beam could stimulate SOD activity (Fig. 2). However, enhancement of SOD activity alone cannot alleviate the burden of excess ROS. Peroxide is a highly toxic ROS and must be sequestered by the action of CAT and POD, which converts peroxide into oxide and oxygen (Mattes., 2000). Xiong, Xing, Feng et al. (2009) showed SOD activity in Pleurotus nebrodensis decreased throughout the postharvest storage period in both irradiated samples and non-irradiated controls although the rate of decrease in the latter was significantly slower (p< 0.05) during the first 10 days.
Such behavior is in aggrement with results reported earlier (Gautam, Sharma and Thomas., 1998). This negative shift of PPO activity provoked by the irradiation could be due to a conformational change of the enzyme or to a modification of the active site, namely a reduction of the cupric ion of the enzyme (Fry and Strothkamp., 1983). This ion is required for oxidizing phenols. As PPO is necessary to initate phenol oxidation into dark brown melanin (Skou, Bech and Lundsten., 1974), it is possible that a decrease of PPO activity would lead to an increase of phenol concentration (GHB) and, hence to a lower rate of melanin formation. Koorapati, Foley, Pilling et al. (2004) reported that exposure to electron beam irradiation at doses as high as 5.2 kGy did not affect the polyphenol oxidase activity in A. bisporus mushroom slices, whereas increase in PPO were recorded in whole A. bisporus fruit bodies gamma radiation with 0.5, 1.0 and 2.0 kGy during the first 7, 9 and 12 days postharvest, respectively (Benoit, Aprano and Lacroix., 2000). Enzyme activity in H. marmoreus fruit bodies exposed to between 1.0 and 4.0 kGy of 60Co-irradiation gradually increased in all samples during an initial 16-19 day postharvest period and then declined. Highest peak activity was recorded in nonirradiated controls, and peak activities in irradiated samples were inversely proportional to dosage (Xing, Wang, Feng et al., 2007).
This study has clearly demonstrated that the dose rate of electron beam irradiation has a significant effect on the PPO, POD and SOD activity. Result showed that irradiation at 7.5 kGy improved the enzyme activities of mushrooms during storage in 4 °C. Consequently, we can recommend the irradiation with suitable electron-beam dose in postharvest stage as a good practice to increase POD and SOD activity in Agaricus brunnescens compared with control. Also, decrease in PPO activity and finally browning can be achieved by doses of 5.5 kGy.
ACKNOWLEDGMENTS
Authors acknowledge the support of University of Guilan and Yazd Radiation Processing Center (YRPC), Yazd, from IRAN.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
- Amiot, M.J., Flueriet, A., Cheynier, V., Nicolas, J. Phenolic compounds and oxidative mechanisms in fruit and vegitables. In Phytochemistry of Fruit and Vegetables; Tomas-Barberan, F. A., Robins, R. J., Eds; Clarendon Press, Oxford, U. K. 1997 pp: 51-85.
- Bai, Z., Harvey, L. M., and Mcneil, B. Oxidative stress in submerged cultures of fungi. Crit. Rev. Biotechnol., 2003; 23: 267-302.
- Beaulieu, M., Lacroix, M., Charbonneau, R., Laberge, I., Gagon, M. Effects of irradiation dose rate on microbiological and physical quality of mushrooms (Agaricus bisporus). Sciences des aliments., 1992; 12: 289-303.
- Benoit, M.A., Aprano, G., Lacroix, M. Effect of ã-irradiation on phenylalanine ammonia-lyase activity, total phenolic content, and respiration of mushrooms (Agaricus bisporus). Journal of Agricultural and Food Chemistry., 2000; 48: 6312-6316.
- Bhat, R., Sridhar, K.R., Yokotani, K.T. Effect of ionizing radiation on antinutritional features of velvet bean seeds (Mucuna pruriens). Food Chemistry., 2007; 103: 860-866.
- Bhat, R., and Sridhar, K.R. Nutritional quality evaluation of electron beam irradiated (Nelumbo nucifera) seeds. Food Chemistry., 2008; 107: 174-184.
- Chanda, S.V., Singh, Y.D. Changes in peroxidase and IAA oxidase activities during wheat grain development. Plant Physiology and Biochem., 1997; 35(3): 245-250.
- Constantine, N.G., Stanley, K.R. Superoxidase dismutases. Plant Physiol., 1977; 59: 309-314.
- Fry, D.C., Strothkamp, K.G. Photo inactivation of Agaricus bisporus tyrosinase: Modification of the binuclear copper site. Biochemistry., 1983; 22(21): 4949-4953.
- Galeazzi, M.A.M., Sgarbieri, V., Constantinides, S. M. Isolation, purification and physicochemical characterization of polyphenoloxidase (PPO) from a dwarf variety of banana (Musa cavendishii, L). J. Food Sci., 1981; 46: 150-155.
- Gautam, S., Sharma, A., Thomas, P. Gamma irradiation effect on shelf-life, texture, polyphenol oxidase and microflora of mushrooms (Agaricus bisporus). Int J Food Sci Nutr., 1998; 49: 5-10.
- Giri, S.K., Prasad, S. Drying kinetics and rehydration characteristics of microwave vacuum and convective hot-air dried mushrooms. Journal of Food Engineering., 2007; 78: 512-521.
- Hammond, J.B.W., Nichols, R. Changes in respiration and soluble carbohydrates during the postharvest storage of mushrooms (Agaricus bisporus). J. Sci. Food Agric., 1975; 26: 835–842.
- Holm, N.W., Berry, R.J. Manual on Radiation Dosimetry. Dekker, New York, USA. 1970.
- Jamieson, D. J. Oxidative stress responses of the yeast Saccharomyces cerevisiae. Yeast., 1998; 14: 1511-1527.
- Jolivet, S., Arpin, N., Wichers, H.J., Pellon, G. Agaricus bisporus browning: a review. Mycol. Res., 1998; 102: 1459-1483.
- Kader, A. A. Potential applications of ionizing radiation. J. Am. Food Technol., 1986; 40: 117–121.
- Kiong, A., Lai, A., Hussein, S., Harun, A.R.. Physiological responses of orthosiphon stamineus plantlets to gamma irradiation. Amer- Eurasian J. Sustain. Agric., 2008; 2: 135-149.
- Koorapati, A. Foley, D. Pilling, R., Prakash, A. Electron-beam irradiation preserves the quality of white button mushroom (Agaricus bisporus) Slices. Journal of Food Science., 2004; 69(1): 25-28.
- Long, J.T., Alben, J.O. Preliminary studies of mushroom tyrosinase (Polyphenol oxidase). Mush. Sci., 1969; 5: 281-299.
- Mattes, J.M. Effect of antioxidant enzymes in the molecular control of reactive oxygen species toxicology. Toxicology., 2000; 153: 83-104.
- Mexis, S.F., Badeka, A.V., Chouliara, E., Riganakos, K.A., Kontominas, M.G. Effect of g-irradiation on the physicochemical and sensory properties of raw unpeeled almond kernels (Prunus dulcis). Innovative Food Science and Emerging Technologies., 2009; 10: 87-92.
- Moerchbacher, B.M., Noll, U.M., Flott, B.E. Relsener, H.J. Lignin biosynthetic enzymes in stem rust infected, resistant and susceptible near-isogennic wheat lines. Physiol. Mol. Plant Pathol., 1988; 33: 33-46.
- Moore, D. Fungal morphogenesis. Development and cell biology series. Cambridge, UK: Cambridge University press. 1988.
- Moussa, H., Abdel-Aziz, S.M. Comparative response of drought tolerant and drought sensitive maize genotypes to water stress. Aust. J. Crop Sci., 2008; 1: 31-36.
- Nerya, O., Ben-Arie, R., Luzzato, T., Musa, R., Khativ, S., Vaya, J. Prevention of Agaricus bisporus postharvest browning with tyrosinase inhibitors. Postharvest Biology and Technology.. 2006; 39: 272–277.
- Ogawa, M., Uritani, I. Effect of Gamma radiation on peroxidase development in sweet potato disks. Radiation Research., 1970; 41: 342-352.
- Omar, M.S. Effect of gamma ray on callus cultures and asexual embryogenesis in Phoenix dacryliferal. Plant Mutation Breeding for Crop Improvement., 1988; 6(2): 258-264.
- Park, D. Vestal, T.A. Electron Beam. 21st Century food technology. Texas A&M University., 2002.
- Qin, X., Wang, F., Wang, X., Zhou, G., Li, Z. Effect of combined treatment of 60Co gamma ray and EMS on anti-oxidase activity and ODAP content in Lathyros sativus. Ying Yong Sheng Tai Xue Bao., 2000; 11(6): 957-965.
- Robb, D.A. In: Lontie, R. (Ed.), Copper Proteins and Copper Enzymes, Vol. 2. CRC Press, Boca Raton FL, 1984 pp. 207–241.
- Robb, D.A., Gutteridge, S. Polypeptide composition of two fungal tyrosinases. Phytochemistry., 1981; 20: 1481–1485.
- Royse, D.J., Wuest, P.J. Identification and thermal sensitivity of two bacterial pathogens of Agaricus bisporus. Plant Disease., 1980; 64: 455-457.
- Skou, J.F., Bech, H., Lundsten, K. Effects of ionizing irradiation on mushrooms as influenced by physiological and environmental coniditions. Radiat. Bot., 1974; 14: 287-299.
- Strid, A., Chow, W.S., Anderson, J.M. Effect of supplementary gamma irradiation on photosynthesis in Pisum sativum. Biochem., 1990; 1020(1): 260-268.
- Stussi, H., Rats, D. The biosynthesis and possible function of gamma-glutamin1-4-hydroxybenzene in Agaricus bisporus. Phytochemistry., 1981; 20: 2347-2352.
- Vamos-Vigyazo, L., Haard, N.F. Polyphenol oxidase and peroxidases in fruits and vegetables. Crit. Rev. Food. Sci. Nutr., 1981; 15: 49-127.
- WHO. Wholesomeness of irradiated food. Technical Report Series 659, Geneva., 1981.
- Diehl, J.F. Food irradiation – past, present and future. Radiation Physics and Chemistry., 2002; 63: 211-215.
- Wi, S.G. Chung, B.Y., Kim, J.S. Effects of gamma irradiation on morphological changes and biological responses in plants. Micron., 2006; 38: 553-564.
- Xing, Z.T., Wang, Y.S., Feng, Z.Y., Zhao, Z. H., Liu, X.H. Effect of 60Co-irradiation on postharvest quality and selected enzyme activities of Hypsizygus marmoreus fruit bodies. Journal of Agricultural and Food Chemistry., 2007; 55: 8126–8132.
- Xiong, Q.L., Xing, Z.T., Feng, Z., Tan, Q., Bian, Y.B. Effect of 60Co gamma-irradiation on postharvest quality and selected enzyme activities of Pleurotus nebrodensis. Food Science and Technology., 2009; 42: 157–161.
- Zhang, X., Flurkey, W.H. Phenoloxidases in portabella mushrooms. Journal of Food Science., 1997; 62: 97-100.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.