ISSN: 0973-7510
E-ISSN: 2581-690X
Avian paramyxovirus serotype-1 (AMPV1) infects a wide range of avian species leading to broader range of clinical symptoms. The ease of transmission has allowed the virus to spread worldwide with varying degree of virulence depending upon virus strain and host range. Rapid detection is an important step to prevent an outbreak of the disease. The present study was carried out to detect APMV-1 from chicken reared in Kashmir Valley. Out of 12 suspected disease outbreaks, all were positive for AMPV-1. APMV-1 was detected using Matrix Protein gene (M gene) by RT-PCR. Detection by M gene is used for primary screening of the APMV-1 in chicken with both virulent and avirulent forms.
Avian paramyxovirus serotype 1, Matrix protein gene, RT-PCR.
Avian paramyxovirus serotype-1 (APMV-1) is a member of the family Paramyxoviridae and is the causative agent of virulent Newcastle disease (vND)1,6. The virus is able to infect wide range of avian species and virulent strains can cause significant clinical signs. Due to the extensive range of susceptible hosts, the virus has been able to establish itself world-wide. Infection by virulent strains has resulted in several panzootics since 19261,6,7. This disease can have devastating effects on the poultry industry due to the high morbidity and mortality associated with virulent strains of the virus2,3,4. Clinical signs of vND include drop in egg production, respiratory distress, listlessness, weakness and central nervous system signs6. Diagnostic testing and rapid detection are important steps to prevent an outbreak of the disease. The matrix rRT-PCR assay is able to detect APMV-1 RNA within 3 hours of sample receipt in the laboratory. The developed laboratories use this as an important screening assay allowing for a quick turn-around time for reporting results. Lack of detection by the matrix assay can result in a 7 to 14 day delay in reporting detection of the virus.
In this study we standardized a rapid and inexpensive method to detect APMV-1 from chicken based on M gene assay on conventional RT-PCR. This method is naturally less sensitive when compared to real-time PCR assays, however it may be a good alternative for laboratories without RTqPCR (reverse transcriptase real time PCR) equipment.
A total of 12 suspected outbreaks of APMV-1 infection from chicken reared in Kashmir valley were attended. The samples collected from the dead birds included tracheal swabs, cloacal swabs, caecal tonsils, spleen, brain, proventriculus and Payer’s patches. The samples were preserved in 50% glycerol saline at -20ºC or directly in TRIzol reagent (Sigma, USA) at – 80ºC. The tissue was homogenized and RNA was extracted from the homogenized tissue using TRIzol Reagent Total RNA was extracted using TRIzol Reagent. Complementary DNA (cDNA) synthesis was carried out using random hexamer primers and Revert-Aid First-Strand cDNA Synthesis kit. A primer set, M+4100 /M-42208 was used to detect APMV-1 by reverse transcriptase PCR (RT-PCR) for the presence of M gene. PCR assay was performed at a 25 µl total reaction volume with Nuclease Free Water 12.6 µl,10x PCR Buffer 2.5µl, 25mM MgCl2 2 µl, 25mM dNTP mix 0.2 µl, forward primers & reverse primer (12pM)1.2 µl each, Taq DNA Polymerase 0.3 µl, cDNA (2 µg/20 µl) 5 µl. The thermal profile used for this PCR was to heat samples to 95°C for 5 min, followed by 40 cycles of 30 sec at 94°C, 30sec at 52°C and 30 sec at 72°C, with the final extension at 72°C for 10 min. At the end of the run, the amplification was checked by Agar gel electrophoresis.
The extracted RNA from the samples of all the suspected outbreaks on subjecting to RT-PCR for Matrix protein gene amplified a fragment with the expected size (~121 bp) [Fig:1] and confirmed the presence of APMV-1. Avian paramyxovirus is a major threat to the poultry industry worldwide. The disease particularly affects the livelihood of people across the developing world where poultry and poultry products are the major source of protein and income. The samples from all the 12 suspected outbreaks screened by RT-PCR targeting Matrix (M) protein were positive for APMV-1. The M gene is found to have more conserved regions, particularly at 5´ end of the region. These conserved regions have been exploited for the development of various PCR based diagnostic assays intended to screen a wide variety of APMV genomes. The intended use of the assay based on conserved M gene was primary screening2,4. The advantage of the RT-PCR is the ability to detect a broad spectrum of isolates both virulent and avirulent forms, as the primers bind to the conserved fragment of M genes5. This method is naturally less sensitive when compared to real-time PCR assays, however it may be a good alternative for laboratories without RTqPCR equipment.
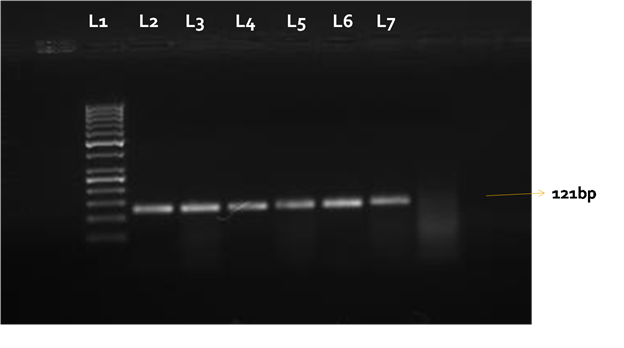 Fig. 1. Matrix protein gene (121bp) was amplified in the positive samples. L1= 50 bp DNA ladder, L2-L7 samples from disease outbreaks suspected for APMV-1
Fig. 1. Matrix protein gene (121bp) was amplified in the positive samples. L1= 50 bp DNA ladder, L2-L7 samples from disease outbreaks suspected for APMV-1ACKNOWLEDGMENTS
The ICAR-NAIP and NAE (anaerobic bacteriology)Projects are highly acknowledged.
- Alexander, D.J., Bell, J.G. and Alders, R.G. 2004 “A Technology Review: Newcastle disease – with special emphasis on its effects on village chickens,” Chapter 1-3. Food and Agriculture Organization of the United Nations. Rome.
- Kim, L.M., Suarez, D.L. and Afonso, C.L. “Detection of a broad range of class I and II Newcastle disease viruses using a multiplex real-time reverse transcription polymerase chain reaction assay,” Journal of Veterinary Diagnostic Investigation, 2008; 20, pp. 414-425.
- Lee, Y.J., Sung, H.W. and Choi, J.G. “Protection of chickens from Newcastle disease with a recombinant baculovirus subunit vaccine expressing the fusion and hemagglutinin-neuraminidase proteins,” Journal of Veterinary Science, 2008; 9(3) : 301-308.
- Miller, P.J., Decanini, E.L. and Afonso, C.L. “Newcastle disease: evolution of genotypes and the related diagnostic challenges,” Infection, Genetics and Evolution, 2010; 10(1): 26-35.
- Nidzworski, D., Smietanka, K., Minta, Z. and Szewczyk , B. “Detection of avian influenza virus and Newcastle Disease virus by duplex One Step RT PCR,”Central European Journal of Biology, 2013; 8: 520-526.
- Saif, Y.M., Fadly, A.M., Glisson, J.R. and McDougald, L.R. “Newcastle disease, other avian paramyxoviruses, and pneumovirus infections” in Diseases of Poultry. pp.75-93, Blackwell Publishing Professional, Ames, IA, USA, 12th edition 2008.
- Seal, B.S., King, D.J. and Sellers, H.S. “The avian response to Newcastle disease virus,” Developmental and Comparative Immunology, 2000; 242-3: 257-268.
- Wise M.G., Sellers H.S., Alvarez R. and Seal B.S. “RNA-dependent RNA polymerase gene analysis of worldwide Newcastle disease virus isolates representing different virulence types and their phylogenetic relationship with other members of the paramyxoviridae” Virus Research, 2004 ; 101 pp.71–80.
© The Author(s) 2017. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


