ISSN: 0973-7510
E-ISSN: 2581-690X
Plant growth promoting bacteria (PGPB) are known for their ability to produce plant hormones and antimicrobial substances, to induce plant resistance, and to solubilize phosphate. In this study, two bacterial isolates Bacillus sp. ST02 and Lactobacillus plantarum ST03, one endophytic bacteria Sp 1E, and one rhizospheric actinomycetes Sp 10R were assayed for their ability in solubilizing phosphate and producing indole acetic acid (IAA), and in affecting chili growth and health. Bacterial ability in solubilizing phosphate was assayed in Pikovskaya’s agar, while the ability in producing IAA was spectrophotometrically measured using Salkowski reagent. To know bacterial potential in promoting plant growth and in reducing fungal disease of Fusarium oxysporum, bacterial isolates were applied in chili seedling grown in small scale field. The results showed that only two isolates Bacillus sp. ST02 and Lactobacillus plantarum ST03 were capable of solubilizing phosphate, but all isolates produced IAA to some extent. Single and combination of ST02 and Sp 10R showed to affect more in increasing plant performance and reducing fungal disease caused by F. oxysporum. This result indicated that the PGPB isolates were considered as a promising microbial inoculant for plant health and growth.
Indole acetic acid, phosphate solubilizing bacteria, fungal control, plant performance.
To control plant pathogenic fungi and to increase plant performance, ones mainly use chemical compounds such as pesticides and fertilizer. Recently, many efforts have been conducted to reduce chemical pesticide and fertilizer application by exploring a possibility of utilization of biological agent as biofertilizer and biocontrol agent. Among biocontrol and biofertilizer, bacteria are the most interesting group of organisms that have already been utilized as beneficial microorganism. A diverse group of beneficial microorganisms have been found, but their roles in environments have not been fully understood. Many studies have been conducted to isolate and to utilize plant-benefit microbes such as plant growth promoting bacteria (PGPB). These include free-living, specific symbiotic, and cyanobacteria (Glick, 2012).
PGPB are group of bacteria enhancing and protecting plant growth through a wide variety of mechanisms such as by producing plant hormones, increasing the availability of soil nutrient, and controlling plant pathogen by inducing plant resistance or by producing antimicrobial compounds (Chakraborty et al., 2013; Przemienieck et al., 2015; de Sauza et al., 2015). Furthermore, they may assist plants to cope with a variety of abiotic stresses (da Silva Sousa et al., 2008; Thijs et al., 2014; Gagné-Bourque et al., 2015), and reduce environmental problems (Glick 2012; Thijs et al., 2014). Among the bacteria Bacillus, Pseudomonas, and Acinetobacter (Chakraborty et al., 2013; Dias et al., 2013; Ngoma et al., 2013; Thijs et al., 2014), and actinomycetes (Doumbou et al., 2001; Franco-Correa et al., 2010; Gopalakrishnan et al., 2013; de Sauza et al., 2015) are known as PGPB.
Chili is one of the most important spices in our tradition. The consumption of fruit of this crop is very high everyday, and even higher in special holidays, with raised price tremendously. Since it is considered as important plant, chili is planted in almost all area in this region, from area close to the sea to highland area.
The chili fruit production depends on good culture practices such as to keep pest and disease controlled, to provide sufficient nutrient to the plant, and to growth the plant in proper season. To control pest and disease, ones utilize synthetic pesticides which may harm to environment and health, increase pest and disease resistant, and kill non target organisms. The need of free chemical on agricultural product together with the awareness of environmental issues increases the attempt to replace the chemicals with more environmentally friendly ones. In this study, PGPB of local isolates were assayed for their ability to solubilize phosphate, to produce indole acetic acid (IAA), and to assay the isolate in controlling fungal disease and enhancing chili productivity.
Microbial isolates and culture condition
Two potential phosphate solubilizing bacterial isolates Bacillus sp. ST02 and Lactobacillus plantarum ST03 isolated from guano of bat cave, one endophytic bacteria Sp 1E and one actinomycete Sp 10R of white lettuce rhizosphere of potential in inhibiting fungal growth, and a chili pathogenic fungi Fusarium oxysporum were used in this study. Microbial isolates were kept in refrigerator on either chitin contained medium or Pikovskaya’s agar for bacteria and actinomycete, and Sabaroud dextrose agar for fungal isolate. All isolates were grown in proper media at ambient temperature for further study.
Assay on Phosphate Solubilizing Potential of Bacterial Isolates
Assay on phosphate solubilizing potential of bacterial isolates was done by dropping a 10 µl of bacterial suspension of OD600 » 0.5 in the center of Pikovskaya’s agar in petridish. Culture was incubated at ambient temperature for 7 days. Phosphate solubilizing index (PSI) was calculated as the ratio of diameter of clear zone divided by diameter of colony.
Assay on Potential of Indole Acetic Acid Production of Bacterial Isolates
IAA production was measured as described by Gordon and Webber (1951). A 10ml of bacterial culture of OD600 » 0.5 grown in broth medium was centrifuged at 10.000 rpm for 15 minutes. Supernatant was taken and mixed with Salkowski reagent in ratio of 1:1. Mixture was incubated at ambient temperature for 60 minutes. IAA was detected as pink color in the mixture. IAA concentration was measured as absorbance using spectrophotometer at 530nm.
Assay on Effect of Bacterial and Actinomycetes Isolates in Controlling Fusarium oxysporum and Chili Performance in Field
All bacterial and actinomycete isolates were grown in nutrient broth. Chili seedling of 2 weeks were planted in soil mixed with compost in small scale field of Kebun Percobaan Tanaman Sayuran Berastagi, Balai Penelitian Tanaman Sayuran, Tongkoh, Deli Serdang, North Sumatra. No fertilizer and pesticide were applied, and no prior soil sterilization was carried out. Bacterial suspension of » 108 cell/ml was applied next to chili seedling 2 weeks after planting, while fungal suspension of » 108 conidia/ml was infested one week after bacterial infestation. Treatment was applied as described in Table 1.
Table (1):
PGPB treatments in chili.
Treatments |
Type of treatments |
|---|---|
(+) control |
Soil was applied with F. oxysporum |
(-) control |
No microbial infestation |
Sp 1E |
Soil was applied with F. oxysporum and Sp 1E |
Sp 10R |
Soil was applied with F. oxysporum and Sp 10R |
ST02 |
Soil was applied with F. oxysporum and Bacillus sp. ST02 |
ST03 |
Soil was applied with F. oxysporum and Lactobacillus plantarum ST03 |
Sp 1E+Sp 10R |
Soil was applied with F. oxysporum, Sp 1E, and Sp 10R |
Sp 1E+ST02 |
Soil was applied with F. oxysporum, Sp 1E, and Bacillus sp. ST02 |
Sp 1E+ST03 |
Soil was applied with F. oxysporum, Sp 1E, and Lactobacillus plantarum ST03 |
Sp 10R+ST02 |
Soil was applied with F. oxysporum, Sp 10R, and Bacillus sp. ST02 |
Sp 10R+ST03 |
Soil was applied with F. oxysporum, Sp 10R, and Lactobacillus plantarum ST03 |
ST02+ST03 |
Soil was applied with F. oxysporum, Bacillus sp. ST02, and Lactobacillus plantarum ST03 |
Observation on control of Fusarium oxysporum and chili performance
Observation on plant health and performance included chlorophyll content, fresh-weight and dry-weight, and frequency of infected plant, disease coverage, and rotten fruit of chili fruit was conducted at the end of study of 16 weeks, while plant height and number of branch were observed at 14 weeks after planting. Chlorophyll content was measured as described by Wintermans and de Mots (1965).
Phosphate Solubilizing Assay
A diverse group of bacteria and actinomycetes has been reported to solubilize phosphate to make it available to plants (da Silva Sousa et al., 2008; Dias et al., 2013; Ngoma et al., 2013). By growing them in Pikovskaya’s agar, these microorganisms show clearing zone around their colony measured as PSI. In this study, bacterial isolates Bacillus sp. ST02 and Lactobacillus plantarum ST03 showed to solubilize phosphate, whether the Sp 1E and Sp 10R did not showed the ability to solubilize phosphate (Figure 1). PSI of that two bacterial isolates was considerably higher compared to that of previous studies by Kasmita (2010) and Ruwandani et al. (2014). Lactobacillus plantarum ability in solubilizing phosphate has not been reported so far. However, the ability of Lactobacillus plantarum in solubilizing phosphate was believed due to its ability in producing organic acids. Previous studies have shown that Lactobacillus species was isolated as endophyte and had potential as biocontrol agent for plant disease pathogens. Pereira et al. (2012) reported that 6 out of 45 isolated endophytic bacteria from strawberry plants belong to the genera Lactobacillus. In addition, Limanska et al. (2015) demonstrated that Lactobacillus plantarum might reduce the percentage of tumor formation by Agrobacterium tumefaciens on carrot plants up to 53%.
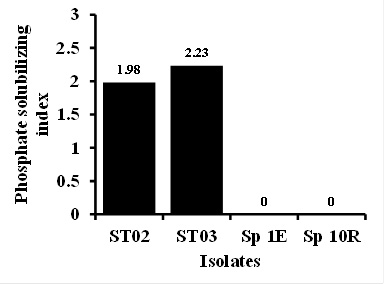 Fig. 1. PGPB isolate ability to solubilize phosphate
Fig. 1. PGPB isolate ability to solubilize phosphateIAA Production Assay
All four isolates of bacteria and actinomycetes used in this study showed to vary in producing IAA (Figure 2). Sp 10R showed to have relatively higher IAA production. IAA is one of the most physiologically active auxins (Yasmi et al., 2016). The higher production of IAA by the isolates is expected to promote plant growth. Many bacteria and actinomycetes have been reported to produce plant hormones such as auxine and giberelic acid (Ponmurugan and Gopi 2006; da Silva Sousa et al., 2008; Saharan and Nehra 2011; Dias et al., 2013; Ngoma et al., 2013; Przemienieck et al., 2015).
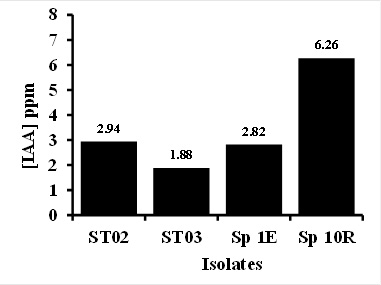 Fig. 2. PGPB isolate ability to produce IAA
Fig. 2. PGPB isolate ability to produce IAAEffect of Bacterial Treatment in Controlling of Fusarium oxysporum and Chili Performance
Application of bacterial isolates and actinomycete, both singly or in combination showed to have different effect in controlling chili disease caused by F. oxysporum compared to that of (+) control and (-) control. It seemed that combination of Sp 10R+ST02 showed to have lower frequency of infected plant, disease coverage, and rotten fruit. This was also shown with application of the single isolates of both (Table 2), in which lower infected plant and rotten fruit were observed. It was observed that chili with no microbial application still got fungal disease with mild disease coverage, which was susceptible to be infected by any fungal pathogen of unsterilized soil.
Table (2):
Effect of PGPB application on chili disease.
| Percentage of | |||
|---|---|---|---|
| Treatments | Frequency of infected plant | Disease coverage | Rotten Fruit |
| (+) control | 26.7 | 93.3 | 3.5 |
| (-) control | 13.3 | 40 | 0 |
| Sp 1E | 16.7 | 41.7 | 4.3 |
| Sp 10R | 6.7 | 53.3 | 3.3 |
| ST02 | 0 | 73.3 | 3.7 |
| ST03 | 33.3 | 100 | 4.1 |
| Sp 1E+Sp 10R | 20 | 53.3 | 19.2 |
| Sp 1E+ST02 | 13.3 | 100 | 4.1 |
| Sp 1E+ST03 | 13.3 | 40 | 1 |
| Sp 10R+ST02 | 5.6 | 27.8 | 0.9 |
| Sp 10R+ST03 | 20 | 66.7 | 4.9 |
| ST02+ST03 | 26.7 | 66.7 | 1.4 |
Application of phosphate solubilizing bacteria of isolate ST02 both singly and in combination to chili plant rhizosphere seemed to noticeably increase plant height, number of branch, and fresh-weight and dry-weight of chili fruit which was similar to that of single SP 10R of IAA producer (Figure 3, 4, and 5). Sufficient of nutrient such as phosphate, reduction of fungal disease, and adequate of plant hormones contributed to this result. IAA may directly influence of shoot growth and development (Yasmi et al., 2016). However, no effect was shown in chlorophyll content of chili plant (Figure 6). Dias et al. (2013) isolated and screened of bacterial strains producing IAA and of capable of phosphate solubilization. It was found that 59 out of 79 strains had both abilities, in which 7 strains promoted crop growth under greenhouse conditions in unsterilized soil. However, he observed that growth promoting capability was not consistently related in vitro to IAA production and phosphate solubilization. In this study, it seemed that isolate both producing more IAA and more capable of solubilizing phosphate promoted more chili growth and health.
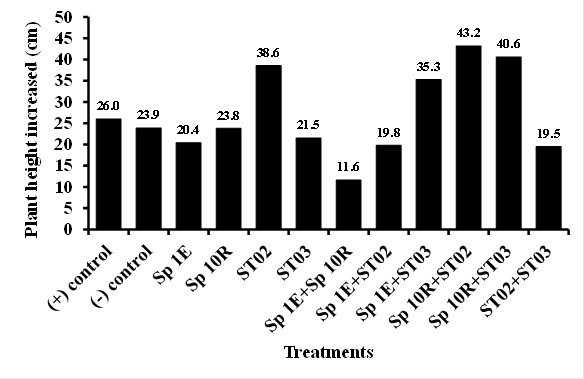 Fig. 3. Effect of PGPB isolate application in increasing plant height
Fig. 3. Effect of PGPB isolate application in increasing plant height 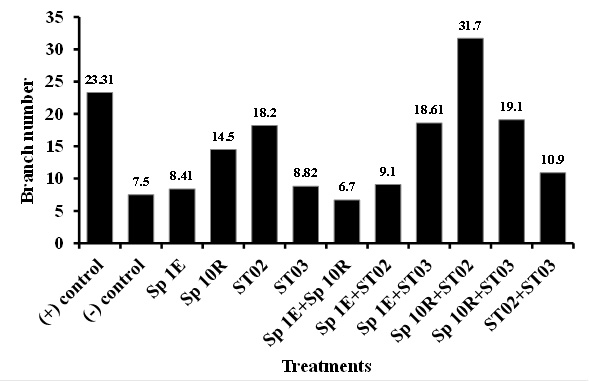 Fig. 4. Brach number of chili treated with PGPB
Fig. 4. Brach number of chili treated with PGPB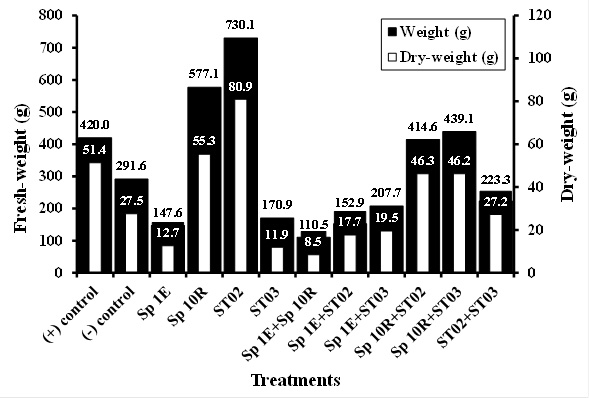 Fig. 5. Effect of PGPB isolate application in fresh-weight and dry-weight of chili fruit
Fig. 5. Effect of PGPB isolate application in fresh-weight and dry-weight of chili fruit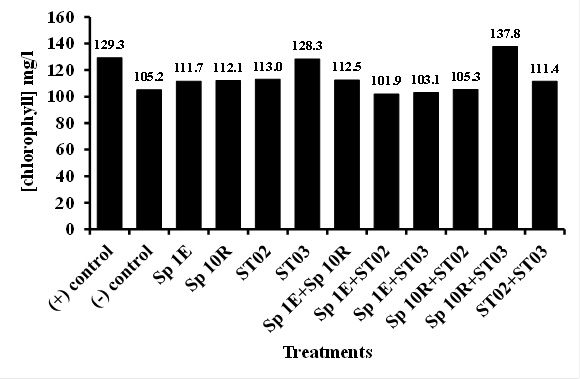 Fig. 6. Effect of PGPB isolate application in chlorophyll content
Fig. 6. Effect of PGPB isolate application in chlorophyll contentMany actinomycetes especially of the genus Streptomyces have been utilized as biocontrol agent of plant disease. Their ability to control plant disease is due to adequate production secondary metabolites, such as antibiotics, siderophores, and extracellular enzymes (Dias et al., 2013; H Etesami et al., 2014). They have also been reported to produce plant hormone and to solubilize phosphate, and play a role in decomposition and nutrient mineralization (Ngoma et al., 2013; H Etesami et al., 2014; Laid et al., 2014). Moreover, actinomycetes has been reported to produce degrading enzymes such as pectinase, xylanase, cellulase, lipase, amylase that may help it to decompose organic material in soil and in turn provide nutrient for plant (Ogbo and Okonkwo 2012).
Beneficial microorganisms like PGPB can be used as an alternative to substitute agrochemical compounds in agriculture in controlling plant disease. In addition, these microorganisms might provide sufficient plant nutrient by solubilizing phosphate and fixating nitrogen (Dias et al., 2013; Ngoma et al., 2013; Yasmi et al., 2016). Moreover, these microorganisms act as plant growth regulator by producing amino acids, vitamin, and plant hormones such as auxine and giberelic acid, as seen in the enhancement of seedling germination, stand health, plant vigor, plant height, shoot weight, nutrient content of shoot tissues, early bloom, chlorophyll content, and increased nodulation in legumes (Ponmurugan and Gopi 2006; Saharan and Nehra 2011).
Phosphor (P) is the second key important macronutrient after nitrogen for plant growth. Plants absorb phosphorus mainly in the form of phosphate ion, H2PO4– and HPO42-. Phosphate availability in soil is usually limited. In acidic soil, phosphate bond together with aluminum (Al) and iron (Fe), while in alkaline soil they tend to bond with calcium (Ca) and magnesium (Mg). An alternative in phosphate solubilization for improving the efficiency of natural phosphate utilization is through microbial metabolism. The main mechanism to solubilize phosphate is related to organic acids synthesized by microorganisms (da Silva Sousa et al., 2008). The ability of PGPB to solubilize phosphate is also due to production of alkaline or acid phosphatase and other enzymes, and makes it available to the roots (Franco-Correa et al., 2010; Przemieniecki et al., 2015).
It is very important to protect plant growth in the early stage to yield good agricultural product (Przemieniecki et al., 2015). Up until now, many agrochemical compounds are applied for that purpose. One alternative to decrease the agrochemical application in controlling plant disease is to use biological control agent. The ability of biological control agent to control plant disease is due to their ability to product antimicrobial substances such as antibiotics, siderophore, and hydrogen cyanide (Ngoma et al., 2013; H Etesami et al., 2014; Laid et al., 2016), lytic enzymes such as chitinase, glucanase, and other antifungal protein (Ogbo and Okonkwo 2012), and to induce plant resistance (Chakraborty et al., 2013; Przemieniecki et al., 2015).
ACKNOWLEDGMENTS
We would like to thank to University of Sumatera Utara for fully supporting this research.
- Chakraborty U, Chakraborty BN, Chakraborty AP, Sunar K, Dey PL. Plant growth promoting rhizobacteria mediated improvement of health status of tea plants. Indian Journal of Biotechnology, 2013; 12: 20-31.
- da Silva Sousa C, Soares ACF, da Silva Garrido M. Characterization of Streptomycetes with potential to promote plant growth and biocontrol. Sci. Agric. (Piracicaba, Braz.), 2008; 65(1): 50-55.
- de Souza R, Ambrosini A, Passaglia LMP. Plant growth-promoting bacteria as inoculants in agricultural soils. Review Article. Gene. Mol. Biol., 2015; 38 (4): 401-419.
- Dias A, dos Santos SG, da Silva Vasconcelos VG, Radl V, Xavier GR, Rumjanek NG, de Lucena Duarte Ribeir R. Screening of plant growth promoting rhizobacteria for the development of vegetable crops inoculants. Afr. J. Microbiol. Res., 2013; 7(19): 2087-2092.
- Doumbou CL, Hamby Salove MK, Crawford DL, Beaulieu C. Actinomycetes, promising tools to control plant diseases and to promote plant growth. Phytoprotection, 2001; 823; 85-102.
- Franco-Correa M, Quintana A, Duque C, Suarez C, Rodriguez MX, Barea J-M. Evaluation of actinomycete strains for key traits related with plant growth promotion and mycorrhiza helping activities. App. Soil Ecol., 2010; 45: 209-217.
- Gagné-Bourque F, Mayer BF, Charron J-B, Vali H, Bertrand A, Jabaji S. Accelerated growth rate and increased drought stress resilience of the model grass Brachypodium distachyon colonized by Bacillus subtilis B26. Plos one, 2015; 10(6): e0130456. doi:10.1371/journal.pone.0130456.
- Glick BR. Plant growth-promoting bacteria: Mechanisms and applications. Review article. Scientica. 2012; Article ID 963401, 15 pages.
- Gopalakrishnan S, Srinivas V, Vidya MS, Rathore A. Plant growth-promoting activities of Streptomyces spp. in sorghum and rice. SpringerPlus 2013, 2: 574.
- Gordon SA, Weber RP. Colometric estimation of indole acetic acid. Plant Physiol. 1951; 26(1): 192-195.
- H Etesami H, Hosseini M, Alikhani HA. In planta selection of plant growth promoting endophytic bacteria for rice (Oryza sativa L.). J Soil Scie. Plant Nutri. 2014; 14(2): 491-503.
- Kasmita R. Isolasi, karakterisasi, dan identifikasi molekuler bakteri pelarut fosfat (BPF) dari beberapa sampel tanah di Bogor, Nusa Tenggara Barat (NTB), dan Nusa Tenggara Timur (NTT). [Thesis]. Institut Pertanian Bogor 2010.
- Laid B, Kamel K, Mouloud G, Manel S, Walid S, Amar B, Hamenna B, Faiçal B. Effects of plant growth promoting rhizobacteria (PGPR) on in vitro bread wheat (Triticum aestivum L.) growth parameters and biological control mechanisms. Adv. Microbiol., 2016; 6: 677-690.
- Limanska N, Korotaeva N, Biscola V, Ivanytsia T, Merlich A, Franco BDGM, Chobert JM, Ivanytsia V, Haertle T. Study of the potential application of lactic acid bacteria in the control of infection caused by Agrobacterium tumefaciens. J. Plant. Pathol. Microb., 2015; 6(8): 292-301.
- Ngoma L, Esau B, Babalola OO. Isolation and characterization of beneficial indigenous endophytic bacteria for plant growth promoting activity in Molelwane Farm, Mafikeng, South Africa . Afr. J. Biotechnol., 2013; 12(26): 4105-4114.
- Pereira MGV, Magalhaes KT, Lorenzetii ER, Souza TP, Schwan RF. A multiphasic approach for the identification of endophytic bacterial in strawberry fruit and their potential for plant growth promotion. Microb. Ecol., 2012; 63(2): 405-417.
- Ponmurugan P, Gopi C. In vitro production of growth regulators and phosphate activity by phosphate solubilizing bacteria. Afri. J. Biotechnol., 2006; 5(4): 348-350.
- Przemieniecki SW, Kurowski TP, Karwowska A. Plant growth promoting potential of Pseudomonas sp. SP0113 isolated from potable water from a closed water well. Arch. Biol. Sci., 2015; 67(2): 663-673.
- Ruwandani MN, Rakhmawati A, Yulianti E. Isolasi, karakterisasi, dan identifikasi bakteri pelarut fosfat dari Guano di Gua Anjani, Jawa Tengah. Jurnal Biologi UNY. 2014; 3(2): 1-11.
- Saharan BS, Nehra V. Plant growth promoting rhizobacteria: A critical review. Life Sciences and Medicine Research, 2011; LSMR-21. 30 pages.
- Thijs S, Weyens N, Sillen W, Panagiotis. Potential for plant growth promotion by a consortium of stress-tolerant 2,4-dinitrotoluene-degrading bacteria: isolation and characterization of a military soil. Microbial. Biotechnol., 2014; 7(4): 294-306.
- Wintermans JFGM, de Mots A. Spectrophotometric characterization of chlorophyll and their pheophytins in ethanol. Biochem. Biophys. Acta, 1965; 109: 448-453.
- Yasmi A, Radziah O, Hawa ZE. J., Zainal Abidin MA, Panhwar QA. Characterisation of plant growth-promoting bacteria from Kacip Fatimah (Labisia pumila) under natural tropical forest. Pertanika J. Trop. Agric. Sci., 2016; 39 (4): 557-575.
© The Author(s) 2017. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


