The human microbiota comprises several microbial communities, which play key roles in maintaining host health, especially through enhancing immunity. Therefore, we aimed to examine the influence of the microbiota on immune function, particularly the mechanisms through which these microbes modify immune responses and provide protection against disease. We found that commensal bacteria maintain mucosal barrier integrity, control mucosal immune responses, and produce antimicrobial peptides. This helps protect the bloodstream and digestive tract from dangerous bacteria and antigens. Notably, the majority of human microflora are located in the gut. Research has shown that using synbiotics, which contain both probiotics and prebiotics, increases the effectiveness of microbiota-based treatments in altering immune responses and reducing immunological disorders because they result in synergistic benefits that increase the persistence and activity of microbes in the digestive system. Prebiotics typically consist of indigestible plant cells that promote the development and activity of healthy bacteria in the large intestine. They help strengthen the immune system by facilitating the proliferation of advantageous microorganisms such as bifidobacteria and lactobacilli. Additionally, prebiotics provide fermentation substrates for the production of short-chain fatty acids (SCFAs), which have anti-inflammatory properties, regulate immune cell activity, and maintain intestinal homeostasis. Microbiota-based supplements provide new possibilities for therapeutic intervention through the prevention and management of immune-related diseases. Insights into the intricate connection between the immune system and the microbiota have substantial implications for human health and highlight new opportunities for enhancing immunity and preventing disease.
Microbiome, Innate Immunity, Adaptive Immunity, Microbiome Interplay, Gut Health, Antimicrobial Resistance
The human microbiome includes all the naturally occurring viruses, bacteria, fungi, and other microorganisms on and inside human bodies.1 Although microbes are so small that they must be observed under a microscope, they have profound effects on human health and well-being. Specific microbes help to develop our immune system, protect us from infection, and allow us to digest food and produce energy.1 The various microorganisms present in and on all multicellular animals and plants, known as the microbiota, can exhibit mutualistic, commensal, or pathogenic relationships with their host.2 These microorganisms, which include bacteria, archaea, protists, fungi, and viruses, play essential roles in maintaining host hormonal, metabolic, and immune homeostasis.3 The term microbiome refers to the genetic material of bacteria and their surrounding environments or the collective genomes of microorganisms occupying an ecological niche.4 It also includes all molecules produced by microbes, such as their metabolites, toxins, signaling molecules, and organic and inorganic compounds, in addition to chemicals that are produced by coexisting hosts and shaped by the environment in which they live.5 The complex network of innate and adaptive components that constitute the immune system is capable of responding to a variety of stimuli.6 This cellular network acts as a potent regulator of host homeostasis in response to microbial and environmental interactions, thereby allowing tissue function to be maintained or restored.7 The mechanisms of host-microorganism interactions likely evolved to maintain symbiotic relationships with these diverse microbial communities through the emergence of complex microbiota and the evolution of distinct immune system functions, responses, and components, especially those related to adaptive immunity.8 The microbiota supports and regulates all aspects of the immune system. The first line of defense in the immune system is provided by physical barriers such as the skin and mucous membranes and chemical barriers such as stomach acid, tears, and sweat.1 These structural components serve as fundamental barriers between the outer environment and internal body structures. Tight junctions serve as barriers that block pathogens from entering the skin, while proteolytic cleavage activates the production of antimicrobial peptides, such as cathelicidins and β-defensins, which combat infections through multiple mechanisms.9
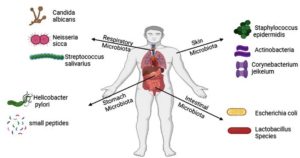
Location of the human microbiota
The human body contains a wide variety of microorganisms, including bacteria, viruses, eukaryotes, and archaea. The complex ecological community comprised of these microorganisms affects human health and disease.4 Communities of bacteria that live on the skin or in the oral cavity, respiratory system, gastrointestinal tract, urinary tract, and reproductive tract comprise the human microbiota. Billions of microorganisms reside in or on the human body. In particular, intestinal microflora have a considerable influence on the physiology of their host.4 Firmicutes and Bacteroidetes are the most prevalent bacterial phyla in the small intestine, accounting for approximately 90% of the gut microbiota. Other microorganisms present in the gut include lactic acid-producing bacteria, protozoa, and fusobacteria.4 They play essential roles in host physiology, including supporting metabolic processes, defending against infection, and providing nutrients and energy. However, some microbes in the human microbiome may be pathogenic.10 Twelve bacterial strains comprise the oral microbiota: bacteroids, actinobacteria, fusobacteria, saccharibacteria, gracilibacteria, chlamydia, chloroflexi, spirochetes, SR1, synergists, fusobacteria, and bacteroidetes.11 Other microorganisms found in the oral cavity include viruses, fungi, and protozoa. Together, these microorganisms protect the oral cavity, help maintain oral homeostasis, and delay the onset of disease. When the natural balance of microorganisms in the mouth is disrupted, several types of bacterial infections can occur.12 Diverse microorganisms also inhabit the human urinary system and genitalia. The most common type of bacteria found in the vagina are lactobacilli. However, identifying the exact species and number of bacteria present in the vaginal canal is challenging. The vaginal microflora play essential roles in maintaining vaginal health and preventing disease.13 Approximately 90% of human cells are connected to microbiota. Figure 1 shows the locations of the human microbiota.
The genetic profile and number of different microbial cells present in the gut is regulated by the local climate and anatomy of the body site.4 At birth, babies acquire a substantial amount of microflora from their mothers. However, the composition of this flora is extremely dynamic and constantly changes during the first three years of life, as well as during adolescence, middle age, and old age.14
Types of microorganisms in the microbiota
Microbiota consist of different types of microorganisms, including bacteria, fungi, viruses, archaea, and protists.15 These microorganisms play important roles in supporting immunological, hormonal, and metabolic processes in host organisms, including humans. They are indispensable for maintaining the overall health and homeostasis of their hosts.16 As bacteria are the most common microorganisms in the human gut, they have been the subject of much research and are linked to both the development of pathology and maintenance of homeostasis.17 Some bacteria are useful and serve vital purposes in the human body. For example, they protect the body from dangerous bacteria, increase the absorption of nutrients from food, and help to break down cellular debris.18 Commensal bacteria break down various polysaccharides and proteins that the stomach cannot metabolize to produce short-chain fatty acids (SCFAs), vitamins, and vital amino acids.19 In addition, they support the structural, mechanical, and barrier functions of the intestinal mucosa and protect the intestines.20 Commensal bacteria in the gut support a healthy immune system by aiding intestinal immune cells and maintaining a balanced microflora composition. They play an important role in regulating specific immunological functions and ensuring immune system homeostasis.20 Meanwhile, fungi constitute approximately 0.1% of microorganisms in the human gut.21 Through toll-like receptors (TLR2 and TLR4), Dectin-1 (CLEC7A), the scavenger receptor family, and other mechanisms, fungi interact with the host immune system (CD5, CD36, and SCARF1), and fungal cell wall components, such as beta-glucans, chitin, and mannans, are activated by glycoprotein components of the complement system.22 Similarly, this interaction results in immunological signaling through molecules including IL9, IL17, IL22, NF-κB, NFAT, and ITAM-containing receptors. Notably, viruses can only multiply within live cells and are smaller than bacteria and fungi.23 A wide variety of viruses, including bacteriophages and eukaryotic viruses, reside in the human body and have an immediate impact on health.24 A species persists when the immune response against one type of antigen does not affect another, and mutations are another means of immune escape.25
Role of the microbiota in strengthening the immune system
The two types of immune systems are partially trained and developed by the gut microbiota. Research conducted on germ-free animals has demonstrated that the growth and functionality of the immune system are hampered by the absence of intestinal microflora.26 Microbiota signals are essential for the proper development and control of immune cells in both animals and humans.27 A coordinated antibody response is supported by the intestinal microflora to prevent excessive inflammation or autoimmune reactions. This cellular network functions as a powerful regulator of host homeostasis in response to pathogenic and external factors, allowing tissue function to be maintained or restored. The mechanisms of microbiome-immune system interactions likely evolved to maintain symbiotic associations between hosts and diverse bacteria, as evidenced by the mechanisms associated with resistant immunity. The microbiome supports and regulates every element of the body’s defense system.28
Regulation of immune response
Some beneficial bacteria can stimulate the growth of regulatory cells that suppress overactive immunological responses. In addition, certain gut microflora stimulate the production of secretory IgA and antimicrobial proteins, supporting intestinal barrier maintenance and preventing pathogen invasion.26 The development of autoimmune and inflammatory diseases is associated with disruption of the gut microbiome (i.e., dysbiosis). Changes in the diversity and composition of gut bacteria can lead to immune dysregulation and a loss of tolerance to self-antigens.2 Notably, modulating the microbiota through the intake of probiotics, prebiotics, and postbiotics can enhance the body’s defenses.28 Interventions can help restore a healthy and balanced microbiome and immune response.29 Intestinal epithelial cells (IECs), which form the mucosal barrier, are tightly packed and coated with a gel layer moistened with mucus containing several antiseptic compounds, such as secretory IgA and peptides, which fight bacteria. Most intestinal bacteria cannot penetrate the mucosal barrier.30
As fundamental components of the innate immune system, IECs use both passive and active mechanisms to identify SCFAs that affect the intestinal environment.30 Among SCFAs, butyrate is mainly metabolized by IECs, propionate is mainly absorbed by the liver, and acetate primarily enters the bloodstream. Butyrate produced by commensal bacteria induces the production of transforming growth factor β (TGF-β) in IECs through its HDAC inhibitory activity and transcription factor-specific protein binding to the core promoter. This in turn drives the expression of TGF-β1 in IECs and the subsequent convergence of regulatory T cells in the gut.26 Different gut microbial communities are found between several intestinal mucous layers and across the length and breadth of the gastrointestinal tract. Compared with that of the ileum and colon, the duodenum exhibits significantly lower immunological activity.31 A more permeable gut allows the passage of macromolecules and antigens, which can lead to immune-mediated diseases or disorders in which gut microbes colonize the small intestine.32 Notably, IgA is a major antibody in the gut that is essential for maintaining mucosal homeostasis, and its production by gut-associated lymphoid tissues is a distinct response of the host immune system to microbial colonization of the gut.33
Production of antimicrobial substances
Microbiota also stimulate the production of secretory IgA, an important antibody that ensures the immune exclusion of pathogens on mucosal surfaces. This helps strengthen the intestinal barrier and prevent microbial translocation.34 Microbiota produce metabolites, such as SCFAs, which can stimulate epithelial cells to produce mucus and antibacterial peptides. These antimicrobials create an inhospitable environment for pathogens.35 Disruption of the gut microbiome is associated with weakened antimicrobial defenses and an increased susceptibility to infections.36 Supplementation with probiotics and prebiotics can restore a balanced microbiome, enhancing antimicrobial defenses and supporting the immune system.37 The absorption of iron and other vital nutrients may be inhibited during intestinal infections. “Nutritional immunity” refers to the host’s attempt to starve the infection by sequestering iron.38 It is believed that human nutrition, metabolism, epithelial growth, immunological modulation, control of fat storage, and defense against infections are greatly influenced by the gut microbiota.39 Indeed, alterations in the gut ecosystem have been associated with a number of diseases, such as cancer, inflammatory immunological diseases, and obesity.40 Probiotic therapies can positively influence the gut microbial ecology, gut homeostasis, metabolism, and, ultimately, the general well-being of the host.41 Many of these interactions within the gastrointestinal tract may be mediated by strain-specific, microbial produced bioactive compounds, sometimes known as pharmbiotics.42 These molecules can be living or dead microorganisms as well as bacterial components and metabolites. The synthesis of antimicrobial peptides, such as bacteriocins, can positively modify gut ecology by selectively inhibiting infections.43
Competition with pathogens for nutrients
Nutrient availability strongly influences the type and abundance of microorganisms that colonize the gut.2 Pathogens can more easily penetrate the gut when there is a limited diversity of gut microbiota that utilize limited amount of resources (e.g., after antibiotic therapy), and they must compete with gut commensals for resources to colonize the gut.2 Bacteria in the intestine compete with each other for carbon and nitrogen sources essential for their growth. They can also compete with pathogens for nutrients required to thrive, such as iron and zinc.44 However, some commensal gut flora, such as Bacteroides thetaiotaomicron, have the ability to release proteins that bind to iron-binding compounds such as XusB. As a result, the pathogen has access to iron while it is protected from host sequestration.45 Pathogens are unable to access nutrients, such as iron, as efficiently as commensal microorganisms.1 The struggle for resources among the host, pathogen, and commensal microbiota is complicated by the fact that other Bacteroidetes species encode proteins capable of binding to iron-binding compounds.46 Microbes have an innate ability to feed and reproduce even in nutrient-rich environments. Notably, a key component of the host’s innate defense against bacterial infection is the limitation of essential resources, such as through iron sequestration or tryptophan degradation.47 Thus, nutrient uptake is one of the most important elements of bacterial pathogenesis.48 While immune-educational and immunosuppressive processes allow the pathogen to maintain access to the host’s nutritional stores, colonization provides the organism with continuous access to nutrient sources.2
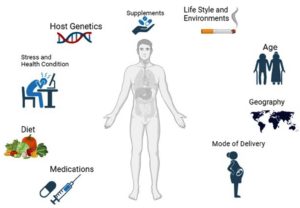
Factors affecting microbiota balance and its impact on immunity
Role of diet, lifestyle, and probiotics
The body obtains nutrients and energy from food, and nutrient absorption and energy production are influenced by intestinal conditions, which in turn affect the homeostasis of the host’s immune system. Billions of bacteria reside in the gut and are vital for several physiological functions, including those of the immune, neurological, endocrine, and metabolic systems.49 Notably, food has a considerable impact on the gut microbiota, which in turn influences the health of the host. Owing to the close connection between gut microflora, nutrition, and overall health, much research has been conducted to determine the underlying processes.50 Many noncommunicable diseases and disorders, including obesity, inflammatory bowel, allergies, dementia, and memory loss, have become serious health problems, especially in developed and developing countries. This highlights the need to understand how to address these issues, with dietary interventions appearing to be the most palatable and effective strategy.51 From birth, dietary practices have a significant influence on the gut microbiota. Microbes begin to colonize the gut immediately after birth, and certain substances in breast milk are thought to play a key role in the early establishment of the gut microbiome.14 During the fetal and postnatal periods, a variety of host and external factors influence the development of immunity. However, few are as important as interactions with commensal microorganisms, which pose a challenge for host evolution, in addition to being the most direct environmental exposure.7 Figure 2 illustrates the diet and lifestyle factors that influence the microbiome. Diet and lifestyle have a fundamental influence on the intestinal microflora balance and its influence on immunity.52 Diet has a major impact on the gut microbiome. For example, breastfeeding promotes the growth of beneficial bacteria, such as Escherichia and Bacilli, which support the development of the immune system.53 In contrast, formula feeding can lead to an increase in potentially harmful bacteria such as Clostridia and Salmonellae, which can weaken the immune system.54
Antibiotic use
The intestinal microbiota affects the wellness of the host and is affected by several factors such as antibiotic use.55 Host health can be negatively affected by antibiotic-induced changes in the microbial composition.56 These include modification of the core features of the microbiota, a decrease in microbial diversity, and the development and selection of antibiotic-resistant strains, thereby increasing the host’s susceptibility to infections by pathogens such as Clostridium difficile.5 There is a global epidemic of antibiotic resistance, and as antibiotic use has increased over time, additional research is required to ascertain how this affects human microbiota and health.57 The composition of the fecal microbiota can be disrupted by lifestyle factors that affect immune function, such as antibiotic use and a sedentary lifestyle. Medicines can reduce the diversity and abundance of microflora, which in turn reduces the efficiency of antibody synthesis.55 To support the maintenance of a healthy microbiome during antibiotic therapy, high-dose probiotics should be prescribed in addition to antibiotics.56 Recurrent Clostridium difficile infection and antibiotic-associated diarrhea are two conditions that can result from changes in the gut flora.58 In addition, antibiotic exposure during early childhood may be associated with immune, neurological, and gastrointestinal disorders.59 Antibiotics significantly alter the intestinal microbiota, reducing species diversity, modifying metabolic processes, and selecting for antibiotic-resistant microorganisms.60 Disruption of the microbiome by antibiotics can compromise immune function by inhibiting the activation of mast cells in the intestinal mucosa, reducing dietary lipid absorption, hyperactivating intestinal macrophages, and increasing the number of pro-inflammatory T cells.55 Antibiotic exposure in early childhood is associated with several alimentary, immunological, and neuropsychological disorders.61 Diet has a major impact on microbiome composition. For example, beneficial bacteria are promoted by breastfeeding, whereas detrimental bacteria are associated with poor nutrition.62
Environmental factors
The microbiota balance can be affected by many environmental factors, and this in turn affects the immune system.1 Maintaining a strong immune response depends on a balanced microbiota, as this offers a variety of beneficial bacteria that can boost immunity and stop the proliferation of harmful pathogens.8 Allergies, autoimmune diseases, and chronic inflammation are immune-related conditions that can result from dysbiosis or microbiota imbalance.63 Environmental exposure to pathogens toxins, and pollutants can affect the microbiota balance, which in turn affects the immune system. Environmental pollutants can directly or indirectly alter microbial populations by inducing oxidative stress and inflammation, which can affect immunological responses.64 However, exposure to diverse microbial communities in the natural environment can strengthen the immune system and increase disease resistance.64 A complex network of interactions between different exposures, affecting different structures and activities of the microbiome, constitutes the way in which the environment affects people’s health.65 Environmental exposures directly affect the human microbiome and are associated with several disorders and diseases in humans such as oropharyngeal malignancies, neurological disorders, diabetes, and obesity.66 Both the macroenvironment, which includes architectural, social, toxicological, and chemical environments, and the microenvironment, which includes factors such as alcohol, smoking, and diet, influence and are associated with environmental exposures.66 Even after accounting for external environmental influences, individual factors such as age, sex, and genes interact to ultimately determine exposure, dose, and subsequent response and function.67 For example, many toxic substances found in cigarette smoke come into direct contact with bacteria in the mouth and upper respiratory tract. These agents can disrupt the microbial ecology through the action of antibiotics, lack of oxygen, and other possible processes.68 The established association between cigarettes and the onset and development of microbe-dependent periodontitis offers strong evidence that the disappearance of beneficial oral agents can lead to the spread of infectious agents and the eventual development of disease.69 Alcohol can have different effects on the human oral microflora. Oral bacteria and fungi can produce carcinogenic nitrosamines, interact with cigarette smoke condensates, convert alcohol and dietary carbohydrates into carcinogenic acetaldehyde, and thereby promote oral carcinogenesis.70 The oral microflora of a smoker is typically robust in its ability to produce acetic acid from ethanol, both across assays and across species. Animal and in vitro studies have suggested possible mechanisms through which oral microbiota may influence the development of oral cancer.70 These processes include the initiation of expanding cells, establishment of chronic inflammation, stimulation of cell invasion, prevention of apoptosis, and cooperation between bacteria and fungi in multilayered oral communities.69
The microbiota enables the immune system to distinguish beneficial microorganisms from dangerous pathogens and further aids the immune system. The immune reaction is regulated by the microbiota, which helps balance the immunologic response. By ensuring that the microbiota maintains an equilibrium, individuals can be protected from illnesses including infections, immune diseases, and inflammation. Metabolites, such as vitamins and SCFAs, are substances produced by the microbiota that indirectly affect the activity of the immune system. For example, SCFAs have anti-inflammatory properties and stimulate the growth of regulatory T cells. Therefore, probiotics targeting the immune system are used to combat bacterial and viral infections. Certain bacterial strains suppress the growth of pathogens or compete with them, thereby inhibiting infection. Furthermore, probiotics regulate immunological processes and maintain the normal microflora, supporting immunity. The ingestion of prebiotic, probiotic and synbiotic, is a viable option for modifying the gut flora composition and enhancing immune function. This treatment plan acts cohesively to reinforce the barrier to infection and aids in the proliferation of beneficial microbes. Additionally, understanding the subtle interplay of microbial equilibrium and immune function with other important variables such as nutrition, antibiotic usage, and environmental factors is essential for creating programs to improve health and defend against disease. Probiotic supplements also aid in maintaining a healthy mix of gut flora, contributing to enhanced immunity and general well-being. Intestinal microflora, when combined with probiotic supplements, play a vital role in boosting the inflammatory response. Thus, probiotics are recommended to help maintain a beneficial mixture of gut flora, which can enhance immunity and boost overall health.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
Not applicable.
ETHICS STATEMENT
Not applicable.
- Lambring CB, Siraj S, Patel K, Sankpal UT, Mathew S, Basha R. Impact of the Microbiome on the Immune System. Crit Rev Immunol. 2019;39(5):313-328.
Crossref - Pickard JM, Zeng MY, Caruso R, Nunez G. Gut Microbiota: Role in Pathogen Colonization, Immune Responses and Inflammatory Disease. Immunol Rev. 2017;279(1):70-89.
Crossref - Matijasic M, Mestrovic T, Eipeic Paljetak H, Peric M, Baresic A, Verbanac D. Gut Microbiota beyond Bacteria—Mycobiome, Virome, Archaeome, and Eukaryotic Parasites in IBD. Int J Mol Sci. 2020;21(8):2668.
Crossref - Dekaboruah E, Suryavanshi MV, Chettri D, Verma AK. Human microbiome: an academic update on human body site specific surveillance and its possible role. Arch Microbiol. 2020;202(8):2147-2167.
Crossref - Aggarwal N, Kitano S, Puah GRY, Kittelmann S, Hwang IY, Chang MW. Microbiome and Human Health: Current Understanding, Engineering, and Enabling Technologies. Chem Rev. 2022;123(1):31-72.
Crossref - Poon MML, Farber DL. The Whole Body as the System in Systems Immunology. iScience. 2020;23(9):101509.
Crossref - Tan CY, Ramirez ZE, Surana NK. A modern world view of host–microbiota–pathogen interactions. J Immunol. 2021;207(7):1710-1718.
Crossref - Belkaid Y, Hand TW. Role of the Microbiota in Immunity and inflammation. Cell. 2014;157(1):121-141.
Crossref - Lefevre-Utile A, Braun C, Haftek M, Aubin F. Five Functional Aspects of the Epidermal Barrier. Int J Mol Sci. 2021;22(21):11676.
Crossref - Passalacqua KD, Charbonneau ME, O’Riordan MXD. Bacterial metabolism shapes the host:pathogen interface. Microbiol Spectr. 2016;4(3)
Crossref - Dewhirst FE, Chen T, Izard J, et al. The Human Oral Microbiome. J Bacteriol. 2010;192(19):5002-5017.
Crossref - Deo PN, Deshmukh R. Oral microbiome: Unveiling the fundamentals. J Oral Maxillofac Pathol. 2019;23(1):122-128.
Crossref - El-Sayed A, Aleya L, Kamel M. Microbiota’s role in health and diseases. Environ Sci Pollut Res Int. 2021;28(28):36967-36983.
Crossref - Sarkar A, Yoo JY, Dutra SVO, Morgan KH, Groer M. The Association between Early-Life Gut Microbiota and Long-Term Health and Diseases. J Clin Med. 2021;10(3):459.
Crossref - Mestrovic T, Matijasic M, Peric M, Paljetak HC, Baresic A, Verbanac D. The Role of Gut, Vaginal, and Urinary Microbiome in Urinary Tract Infections: From Bench to Bedside. Diagnostics. 2020;11(1):7.
Crossref - Thomas S, Izard J, Walsh E, et al. The Host Microbiome Regulates and Maintains Human Health: A Primer and Perspective for Non-Microbiologists. Cancer Res. 2017;77(8):1783-1812.
Crossref - Manos J. The human microbiome in disease and pathology. APMIS. 2022;130(12):690-705.
Crossref - Zhang YJ, Li S, Gan RY, Zhou T, Xu DP, Li HB. Impacts of Gut Bacteria on Human Health and Diseases. Int J Mol Sci. 2015;16(4):7493-7519.
Crossref - Fusco W, Lorenzo MB, Cintoni M, et al. Short-Chain Fatty-Acid-Producing Bacteria: Key Components of the Human Gut Microbiota. Nutrients. 2023;15(9):2211.
Crossref - Gierynska M, Szulc-Dabrowska L, Struzik J, Mielcarska MB, Gregorczyk-Zboroch KP. Integrity of the Intestinal Barrier: The Involvement of Epithelial Cells and Microbiota—A Mutual Relationship. Animals. 2022;12(2):145.
Crossref - Hallen-Adams HE, Suhr MJ. Fungi in the healthy human gastrointestinal tract. Virulence. 2016;8(3):352-358.
Crossref - Aldars-Garcia L, Marin AC, Chaparro M, Gisbert JP. The Interplay between Immune System and Microbiota in Inflammatory Bowel Disease: A Narrative Review. Int J Mol Sci. 2021;22(6):3076.
Crossref - Wang Y, Zhang D, Hou Y, Shen S, Wang T. The adaptor protein CARD9, from fungal immunity to tumorigenesis. Am J Cancer Res. 2020;10(8):2203-2225
- Bai GH, Lin SC, Hsu YH, Chen SY. The Human Virome: Viral Metagenomics, Relations with Human Diseases, and Therapeutic Applications. Viruses. 2022;14(2):278.
Crossref - Zimmermann P, Curtis N. Factors That Influence the Immune Response to Vaccination. Clin Microbiol Rev. 2019;32(2):e00084-18.
Crossref - Yoo JY, Groer M, Dutra SVO, Sarkar A, McSkimming DI. Gut Microbiota and Immune System Interactions. Microorganisms. 2020;8(10):1587.
Crossref - Wu HJ, Wu E. The role of gut microbiota in immune homeostasis and autoimmunity. Gut Microbes. 2012;3(1):4-14.
Crossref - de Sire A, de Sire R, Curci C, Castiglione F, Wahli W. Role of Dietary Supplements and Probiotics in Modulating Microbiota and Bone Health: The Gut-Bone Axis. Cells. 2022;11(4):743.
Crossref - Hitch TCA, Hall LJ, Walsh SK, et al. Microbiome-based interventions to modulate gut ecology and the immune system. Mucosal Immunol. 2022;15(6):1095-1113.
Crossref - Soderholm AT, Pedicord VA. Intestinal epithelial cells: at the interface of the microbiota and mucosal immunity. Immunology. 2019;158(4):267-280.
Crossref - Schroeder BO. Fight them or feed them: how the intestinal mucus layer manages the gut microbiota. Gastroenterol Rep (Oxf). 2019;7(1):3-12.
Crossref - Kim KS. Regulation of T cell repertoires by commensal microbiota. Front Cell Infect Microbiol. 2022;12:1004339.
Crossref - Gutzeit C, Magri G, Cerutti A. Intestinal IgA production and its role in host-microbe interaction. Immunol Rev. 2014;260(1):76-85.
Crossref - Pietrzak B, Tomela K, Olejnik-Schmidt A, Mackiewicz A, Schmidt M. Secretory IgA in Intestinal Mucosal Secretions as an Adaptive Barrier against Microbial Cells. Int J Mol Sci. 2020;21(23):9254.
Crossref - Portincasa P, Bonfrate L, Vacca M, et al. Gut Microbiota and Short Chain Fatty Acids: Implications in Glucose Homeostasis. Int J Mol Sci. 2022;23(3):1105.
Crossref - Hrncir T. Gut Microbiota Dysbiosis: Triggers, Consequences, Diagnostic and Therapeutic Options. Microorganisms. 2022;10(3):578.
Crossref - Żolkiewicz J, Marzec A, Ruszczynski M, Feleszko W. Postbiotics—A Step Beyond Pre- and Probiotics. Nutrients. 2020;12(8):2189.
Crossref - Cassat JE, Skaar EP. Iron in Infection and Immunity. Cell Host Microbe. 2013;13(5):509-519.
Crossref - Liu J, Tan Y, Cheng H, Zhang D, Feng W, Peng C. Functions of Gut Microbiota Metabolites, Current Status and Future Perspectives. Aging Dis. 2022;13(4):1106-1126.
Crossref - Gagliardi A, Totino V, Cacciotti F, et al. Rebuilding the Gut Microbiota Ecosystem. Int J Environ Res Public Health. 2018;15(8):1679.
Crossref - Hemarajata P, Versalovic J. Effects of probiotics on gut microbiota: mechanisms of intestinal immunomodulation and neuromodulation. Therap Adv Gastroenterol. 2013;6(1):39-51.
Crossref - Baishya J, Bisht K, Rimbey JN, et al. The Impact of Intraspecies and Interspecies Bacterial Interactions on Disease Outcome. Pathogens. 2021;10(2):96.
Crossref - Darbandi A, Asadi A, Mahdizade Ari M, et al. Bacteriocins: Properties and potential use as antimicrobials. J Clin Lab Anal. 2021;36(1):e24093.
Crossref - Rowland I, Gibson G, Heinken A, et al. Gut microbiota functions: metabolism of nutrients and other food components. Eur J Nutr. 2018;57(1):1-24.
Crossref - Zafar H, Saier MH Jr. Gut Bacteroides species in health and disease. Gut Microbes. 2024;13(1):1848158.
Crossref - Botta A, Barra NG, Lam NH, et al. Iron Reshapes the Gut Microbiome and Host Metabolism. J Lipid Atheroscler. 2021;10(2):160-183.
Crossref - Best A, Abu Kwaik Y. Nutrition and bipartite metabolism of intracellular pathogens. Trends Microbiol. 2019;27(6):550-561.
Crossref - Sharma AK, Dhasmana N, Dubey N, et al. Bacterial Virulence Factors: Secreted for Survival. Indian J Microbiol. 2017;57(1):1-10.
Crossref - Bo S, Fadda M, Fedele D, Pellegrini M, Ghigo E, Pellegrini N. A Critical Review on the Role of Food and Nutrition in the Energy Balance. Nutrients. 2020;12(4):1161.
Crossref - Spiga L, Fansler RT, Perera YR, et al. Iron acquisition by a commensal bacterium modifies host nutritional immunity during Salmonella infection. Cell Host Microbe. 2023;31(10):1639-1654.e10.
Crossref - Xia X, Jiang Q, McDermott J, Han JJ. Aging and Alzheimer’s disease: Comparison and associations from molecular to system level. Aging Cell. 2018;17(5):e12802.
Crossref - Zhang P. Influence of Foods and Nutrition on the Gut Microbiome and Implications for Intestinal Health. Int J Mol Sci. 2022;23(17):9588.
Crossref - Rinninella E, Raoul P, Cintoni M, et al. What is the Healthy Gut Microbiota Composition? A Changing Ecosystem across Age, Environment, Diet, and Diseases. Microorganisms. 2019;7(1):14.
Crossref - Mancuso G, Midiri A, Gerace E, Biondo C. Bacterial Antibiotic Resistance: The Most Critical Pathogens. Pathogens. 2021;10(10):1310.
Crossref - Patangia DV, Ryan CA, Dempsey E, Ross RP, Stanton C. Impact of antibiotics on the human microbiome and consequences for host health. MicrobiologyOpen. 2022;11(1):e1260.
Crossref - Konstantinidis T, Tsigalou C, Karvelas A, Stavropoulou E, Voidarou C, Bezirtzoglou E. Effects of Antibiotics upon the Gut Microbiome: A Review of the Literature. Biomedicines. 2020;8(11):502.
Crossref - Aljeldah MM. Antimicrobial Resistance and Its Spread Is a Global Threat. Antibiotics. 2022;11(8):1082.
Crossref - Mullish BH, Williams HR. Clostridium difficile infection and antibiotic-associated diarrhoea. Clin Med (Lond). 2018;18(3):237-241.
Crossref - Morreale C, Giaroni C, Baj A, et al. Effects of Perinatal Antibiotic Exposure and Neonatal Gut Microbiota. Antibiotics. 2023;12(2):258.
Crossref - Gestels T, Vandenplas Y. Prenatal and Perinatal Antibiotic Exposure and Long-Term Outcome. Pediatr Gastroenterol Hepatol Nutr. 2023;26(3):135-145.
Crossref - McBurney MI, Davis C, Fraser CM, et al. Establishing What Constitutes a Healthy Human Gut Microbiome: State of the Science, Regulatory Considerations, and Future Directions. J Nutr. 2019;149(11):1882-1895.
Crossref - Furman D, Campisi J, Verdin E, et al. Chronic inflammation in the etiology of disease across the life span. Nat Med. 2019;25(12):1822-1832.
Crossref - Andersen L, Corazon SS, Stigsdotter UK. Nature Exposure and Its Effects on Immune System Functioning: A Systematic Review. Int J Environ Res Public Health. 2021;18(4):1416.
Crossref - Virolainen SJ, VonHandorf A, Viel KCMF, Weirauch MT, Kottyan LC. Gene–environment interactions and their impact on human health. Genes Immun. 2023;24(1):1-11.
Crossref - Panthee B, Gyawali S, Panthee P, Techato K. Environmental and Human Microbiome for Health. Life. 2022;12(3):456.
Crossref - Motsinger-Reif AA, Reif DM, Akhtari FS, et al. Gene-Environment Interactions within a Precision Environmental Health Framework. Cell Genom. 2024; 4(7):100591
Crossref - Conlon MA, Bird AR. The Impact of Diet and Lifestyle on Gut Microbiota and Human Health. Nutrients. 2014;7(1):17-44.
Crossref - Alwithanani N. Periodontal Disease and Smoking: Systematic Review. J Pharm Bioallied Sci. 2023;15(Suppl 1):S64-S71.
Crossref - Sedghi L, DiMassa V, Harrington A, Lynch SV, Kapila YL. The oral microbiome: Role of key organisms and complex networks in oral health and disease. Periodontol 2000. 2021;87(1):107-131.
Crossref - Leigh T, Scalia RG, Autieri MV. Resolution of inflammation in immune and nonimmune cells by interleukin-19. Am J Physiol Cell Physiol. 2020;319(3):C457-C464.
Crossref
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.