ISSN: 0973-7510
E-ISSN: 2581-690X
Enterococci are responsible for serious diseases such as urinary tract infections (cystitis, urethritis, pyelonephritis and prostatis), bacteremia, intra- abdominal, pelvic and soft tissue infections. Infection on burn surfaces were also commonly caused by Enterococcus. The ability of enterococci to cause such diseases is due to acquisition of certain virulence factors such as hemolysin, gelatinase, Biofilm formation and enterococcus surface protein (esp). This study has been conducted to investigate the occurrence of virulence factors between Enterococcus isolated from nosocomial infections and from stool samples (commensal isolates).Both clinical and stool specimens were collected and isolates were identified by microscopic, culture and standard biochemical tests. Virulence factors were examined by phenotypic tests and esp gene were detected by PCR method. There is no significant difference in the virulence factors like hemolysin and gelatinase production between clinical and commensal isolates. Biofilm production and presence of esp gene is very high among clinical isolates. Study of virulence factors associated with invasiveness and disease producing capacity of nosocomial pathogen like multidrug resistant Enterococcus will be very useful in treating them.
Enterococcus, biofilm, esp gene, virulence
Enterococcus, the gram positive cocci are part of normal flora of human intestine, biliary tract and to lesser extent vagina and male urethra. However the incidence of Enterococcal infections, especially nosocomial infection, has dramatically increased over last 25 years1, 2. Enterococci have been implicated in clinical conditions like urinary tract infections (cystitis, urethritis, pyelonephritis and prostatis), bacteremia, intra- abdominal, pelvic and soft tissue infections. Infection on burn surfaces were also commonly caused by Enterococcus.
Enterococcus faecalis is the most common species found in clinical specimens when compared with Enterococcus faecium. Enterococcus faecium is more drug resistant than Enterococcus faecalis3. Most strains of Enterococcus are resistant to penicillin, aminoglycosides and sulphonamides. They show intrinsic resistance to cephalosporins and Cotrimoxazole.
Enterococcus exhibit a number of virulence factors. These virulence factors may contribute to the ability of Enterococcus to cause diseases4. Adherence of Enterococcus to host cell is the important step in the pathogenesis of infection, which is achieved by adhesion factor called Enterococcal surface protein (esp)5. Enterococcus also secrete hemolysin (cytolysin protein), a bacterial toxin that produce haemolytic activity against erythrocytes of human, horse and rabbit. Another secreted molecule is gelatinase which hydrolyse collagen, casein, haemoglobin and other peptides6.
Esp is found to be one of the important virulence factor to enhance the persistence of Enterococcus in urinary bladder during experimental urinary tract infections7. Gelatinase plays an important role in the pathogenesis of endocarditis in an animal model6. Hemolysin producing strains were found to be associated in severe infections. All these factors have been associated with the virulence of Enterococcus in animal models. It is not very clear, the presence of virulence factors in Enterococcus from clinical and commensal isolates contribute to the virulence in humans.
The present study aims to compare the production of these virulence factors between Enterococcus isolated from nosocomial infections and from stool samples, i.e comparison between clinical and commensal isolates.
One hundred and twenty clinical isolates of Enterococcus species were isolated over a period of six months (January 2015- June 2015) from a tertiary care centre, Chennai. 100 commensal isolates of Enterococcus collected from stool samples (faecal flora) were included in this study.
Enterococcus species were isolated from clinical samples such as exudates (postoperative wound infection, diabetic foot ulcers, infected fractures, and burns), septicaemia, endotracheal infection and urinary tract infection. Enterococcus is confirmed by microscopic examination, standard biochemical tests and by culture methods8.
Virulence factors detection
Gelatinase assay
Gelatinase production was detected by inoculation of the organism into tubes containing 12% gelatine in 0.8% nutrient broth. These tubes were incubated for 24 hours at 370c and then placed in the refrigerator for approximately 30 minutes. The liquefaction of gelatine was considered as a positive test9.
Hemolysin production
To determine hemolysin (cytolysin protein) production, isolates were inoculated on brain- heart infusion gar supplemented with 5% sheep blood. These agar plates were incubated at 370c for 24 hours. A clear zone of beta hemolysis around the bacterial colonies indicates the hemolysin activity10.
Biofilm formation
Quantitative adherence assay was the method used to detect the Biofilm formation11. Enterococcus species were inoculated into trypticase soy broth with 0.5% glucose and incubated at 370c for overnight. 200µl of 1: 40 dilution of overnight culture in trypticase – soy broth with 0.5% glucose was inoculated into the wells of micro titre plate and incubated for 48 hours at 37oc. Then the wells were gently washed three times with phosphate buffered saline (PBS). The adherent bacterial film was fixed by air drying at 600c for one hour and then stained with 0.1% safranin and allowed to stand for 20 minutes at room temperature. The absorbance of the Biofilm was measured at 49nm in an ELISA (Enzyme linked immunosorbent assay) reader. Biofilm measurements were repeated for three times in triplicate for each strain. Mean optical density (OD) value of the positive control was considered as standard. Ability of the strain to produce Biofilm were scored as follows, if the OD value is more than 0.2, they are strong Biofilm producer. OD value which is below 0.120 is Biofilm non producer and if OD value is between 0.120 and 0.2 then it is considered as weak Biofilm producer.
Enterococcal surface protein (esp) gene detection
DNA was extracted from Enterococcus species using Purefast bacterial DNA minispin purification kit accordance with manufacturer’s protocol. PCR reaction was performed for the amplification of the 300bp fragment of esp gene using following primer.
esp gene
Forward primer 5’- AAGTGTGGGTGTTG CATCAG- 3’
Reverse primer 5’- TCCCAGTTTGCAGTTGAAGC- 3’
PCR amplification reaction mixture (25µl) contained 10µl probe PCR master mix, 5µl primer forward, 5µl of primer reverse and 5µl of purified genomic DNA. PCR conditions were as follows: Taq enzyme activation at 940c for 3 minutes, 35 cycles of denaturation at 940c for 1 minute, annealing at 580c for 1minute, extension at 720c for 1 minute. Then the final extension is done at 720c for 5 minutes.
The study was undertaken in a tertiary care centre, Chennai for a period of six months(January 2015- June 2015) .One hundred and twenty isolates of Enterococcus species isolated from various clinical infections and 100 commensal isolates of Enterococcus collected from stool samples (faecal flora) were included in this study.
Among the 120 clinical isolates of Enterococcus species , n=78(65%) were from post operative wound infection, followed by diabetic foot ulcer, urinary tract infection, endotracheal infection, infected fracture, burns and septicaemia. Enterococcus strains isolated from various clinical infection shown in Table 1.
Table (1):
Enterococcal strains isolated from clinical infection
| S. No. | Nosocomial infection | Enterococcus strains isolated | |
|---|---|---|---|
| Number | Percentage | ||
| 1 | Post operative wound infection | 78 | 65% |
| 2 | Diabetic foot ulcer | 19 | 15.83% |
| 3 | Urinary tract infection | 11 | 9.16% |
| 4 | Endotracheal infection | 4 | 3.33% |
| 5 | Infected fracture | 3 | 2.5% |
| 6 | Burns case | 2 | 1.66% |
| 7 | Septicaemia | 3 | 2.5% |
Out of 120 clinical Enterococcus isolates 31 (25.83%) produced hemolysin and 57 (47.5%) produced gelatinase. Among 100 commensal Enterococcus isolates, 17 (17%) strains produced hemolysin and 39 (39%) strains produced gelatinase.
51 (42.5%) clinical Enterococcal isolates had the ability to produce Biofilm. Out of which 9 strains were strong Biofilm producers and the remaining were weak Biofilm producers. 29 strains out of 51 Biofilm producing clinical Enterococcus isolates showed the presence of esp gene.
Among the commensal isolates, 19 (19%) were Biofilm producers with 3 strains producing strong and 16 strains producing weak amount of Biofilm. 2 strains out of 19 biofilm producing commensal Enterococcus isolates had esp gene. Production of virulence factors between clinical and commensal Enterococcus species are shown in Table 2.
Table (2):
Distribution of virulence factors between clinical and commensal Enterococcus isolates
S. No. |
Virulence factors |
Clinical isolates (n=120) |
Commensal isolates (n=100) |
|---|---|---|---|
1 |
Gelatinase |
31 |
17 |
2 |
Hemolysin |
57 |
39 |
3 |
Biofilm formation |
51 |
19 |
4 |
Presence of esp gene |
29 |
2 |
Clinical sample wise production of virulence factors like Gelatinase, hemolysin, Biofilm formation and esp gene expression are shown in Table 3. Esp gene expression is shown in Fig 1. Almost all strains causing urinary tract infection, endotracheal infection, infected fracture and septicaemia produced more virulent factors.
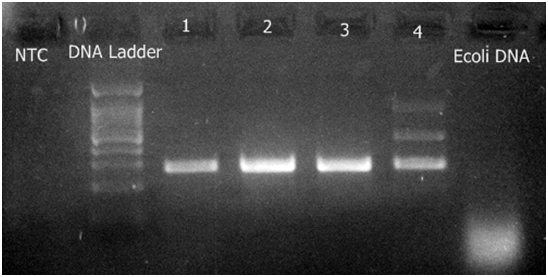
Fig. 1. Detection of gene esp (Enterococcal surface protein)
Table (3):
Infection wise distribution of virulence factors among clinical Enterococcus isolates
Clinical samples |
Gelatinase |
Hemolysin |
Biofilm production |
esp gene expression |
|---|---|---|---|---|
Post operative wound infection (n= 78) |
31 |
20 |
29 |
13 |
Diabetic foot ulcer (n= 19) |
10 |
4 |
4 |
– |
Urinary tract infection (n= 11) |
10 |
3 |
10 |
10 |
Endotracheal infection (n= 4) |
2 |
1 |
4 |
3 |
Infected fracture (n= 3) |
2 |
2 |
2 |
1 |
Burns case (n= 2) |
1 |
1 |
– |
– |
Septicaemia (n= 3) |
1 |
– |
2 |
2 |
Among the clinical Enterococcus isolates, 16 strains produced all 3 virulence factors, Gelatinase and hemolysin were produced by 9 strains, Gelatinase and Biofilm were produced by 15 strains and Hemolysin and Biofilm production is seen in 3 strains. Only Gelatinase is produced by 17 strains, hemolysin produced by 3 strains and Biofilm production is seen in 17 strains (Table 4).
Table (4):
Production of combination of virulence factors in clinical isolates
Clinical samples |
G&H&B |
G&H |
G & B |
H&B |
G |
H |
B |
|---|---|---|---|---|---|---|---|
Post operative wound infection (n= 78) |
10 |
6 |
11 |
1 |
4 |
3 |
7 |
Diabetic foot ulcer (n= 19) |
2 |
1 |
– |
1 |
7 |
– |
1 |
Urinary tract infection (n= 11) |
2 |
– |
2 |
1 |
6 |
– |
5 |
Endotracheal infection (n= 4) |
1 |
– |
1 |
– |
– |
– |
2 |
Infected fracture (n= 3) |
1 |
1 |
– |
– |
– |
– |
1 |
Burns case (n= 2) |
– |
1 |
– |
– |
– |
– |
– |
Septicaemia (n= 3) |
– |
– |
1 |
– |
– |
– |
1 |
Among commensal Enterococcus isolates, 5 strains produced all 3 virulence factors, Gelatinase and hemolysin were produced by 6 strains, Gelatinase and Biofilm were produced by 10 strains and Hemolysin and Biofilm production is seen in 3 strains. Only Gelatinase is produced by 18 strains, hemolysin produced by 3 strains and Biofilm production is seen in 2 strains.
Enterococcus is an important cause of nosocomial infections12. We studied the prevalence of virulence factors like gelatinase, hemolysin, Biofilm formation and presence of esp gene between clinical isolates and commensal isolates of Enterococcus. Both clinical isolates and commensal isolates showed the presence of the virulence factors singly or in combination. Although these factors are very much important in the pathogenesis of disease, their contribution in the clinical isolates and commensal isolates were yet to be researched. In our study, with respect to the virulence factors hemolysin and gelatinase production, there is no significant difference between clinical and commensal isolates which is similar to other studies6, 20.
But nowadays many studies on the virulence factors of Enterococcus mainly focussed on biofilm formation and the presence of a gene coding for enteococcal surface protein (esp), besides the well known aggregative substance, gelatinase and hemolysin15, 20. Biofilm production is an important factor which makes the organism to adhere onto surfaces, which in turn help it to invade and cause disease5. In this study, Biofilm production is very high among clinical isolates from urinary tract infection, endotracheal infection and septicaemia, which correlate with other studies15. Thus biofilm production in nosocomial strains of organism is an important pathogenic factor in causing infection in the hospital environment15, 21.
In some studies, the ability of the Enterococcus to produce Biofilm was linked to the presence of esp13, 14, while in other studies, there is no much association between biofilm formation and presence of esp gene 16, 17, 18. In our study more than 70% strains producing biofilm showed the presence of esp gene in clinical isolates.
In our study the clinical isolates also predominantly produce the combination of gelatinase and biofilm formation, which correlate with a study of Hancock & Perga19. In Hancock’s study, he had explained the association of gelatinase and esp with biofilm formation in Enterococcus species. He identified a two component system, the fsr locus that affected the formation of biofilm through the regulation of gelatinase, which depend on cell density. The gelatinase and esp gene with biofilm formation may act in syngery to establish the infection.
Hence Enterococcus is an important nosocomial pathogen with increasing prevalence of MDR Enterococcus (Multidrug resistant)21.
Our data says there is no significant difference between clinical and commensal isolates in the production of virulence factors like hemolysin and gelatinase. Biofilm production is very high among clinical isolates from urinary tract infection, endotracheal infection and septicaemia. Combination of gelatinase and biofilm formation is high among clinical isolates. Thus the identification of virulence factors associated with invasiveness and disease severity has become an important subject for research. Development of some mechanisms to inhibit the virulence factors like preventing Biofilm production will be very useful in treating the nosocomial pathogen like multidrug resistant Enterococcus.
- Tendulkar PM, Baghdayan AS, Shankar N. Pathogenic Enterococci: New developments in the 21st Century. Cell Mol Life Sci., 2003; 60: 2622-36.
- Vergis EN, Shankar N, Chow JW, Hayden MK, Snydman DR, Zervos MJ, et al. Association between the presence of Enterococcal virulence factors gelatinase, haemolysin and enterococcal surface protein and mortality among patients with bacteremia due to Enterococcus faecalis. Clin Infect Dis., 2002; 35: 570-5.
- Franz CM, Holzapfel WH, Stiles ME. Enterococci at the crossroads of food safety? Int J Food Microbiol., 1999; 47: 1-24.
- S.S.M.Umamageswari, M. Jeya and J.Perumal. virulence factors and susceptibility pattern of isolates of tribe proteae from clinical samples in a tertiary care hospital. Int J Pharm Bio Sci ., 2013; 4(4): (B) 456 – 461.
- Cosentino S, Podda GS, Corda A, Fadda ME, Deplano M, Pisano MB. Molecular detection of virulence factors and antibiotic resistance pattern in clinical Enterococcus faecalisstrains in Sardinia. J Prev Med Hyg., 2010; 51: 31-6.
- Coque TM, Patterson JE, Steckelberg JM & Murray BE. Incidence of hemolysin, gelatinase, and aggregation substance among enterococci isolated from patients with endocarditis and other infections and from feces of hospitalized and community-based persons. J Infect Dis., 1995; 171: 1223–1229.
- Creti R, Imperi M, Bertuccini L, Fabretti F, Orefici G, Di Rosa R &Baldassarri L. Survey for virulence determinants among Enterococcus faecalis isolated from different sources. J Med Microbiol., 2004; 53: 13–20.
- Ross PW.: Streptococcus and Enterococcus. In: Practical medical microbiology.( Collee JG, Fraser AG, Marmion BP, Simmons A, ed.) New Delhi: Curchill Livingstone, 2006; pp 263-73.
- Andrea M, Dib-Hajj F, Lamb L, Kaczmarek F, Shang W, Beckius G, et al. Enterococcal virulence determinants may be involved in resistance to clinical therapy. Diagn Microbiol Infect Dis., 2007; 58: 59-65.
- Chow JW, Thal LA, Perri MB, Vazquez JA, Donabedian SM, Clewell DB, et al. Plasmid-associated hemolysin and aggregation substance production contribute to virulence in experimental enterococcal endocarditis. Antimicrob Agents Chemother., 1993; 37: 2474-7.
- Jayanthi S, Ananthasubramanian N, Appalaraju B. Assessment of pheromone response in biofilm forming clinical isolates of high level gentamycin resistant Enterococcus faecalis. Indian J Med Microbiol., 2008; 23: 248-51.
- Murray BE & Weinstock GM. Enterococci: new aspects of an old organism. Proc Assoc Am Physicians.,1999; 111: 328–334.
- Tendolkar PM, Baghdayan AS, Gilmore MS & Shankar N. Enterococcal surface protein Esp, enhances biofilm formation by Enterococcus faecalis. Infect Immun., 2004; 72: 6032–6039.
- Toledo-Arana A, Valle J, Solano C, et al. The Enterococcal surface protein, Esp, is involved in Enterococcus faecalis Biofilm formation. Appl Environ Microbiol., 2001; 67: 4538–4545.
- Baldassarri L, Bertuccini L, Ammendolia MG, Gherardi G & Creti R. Variant esp gene in vancomycin-sensitive Enterococcus faecium. Lancet.,2001; 357: 1802.
- Hufnagel M, Koch S, Creti R, Baldassarri L & Huebner J . A putative sugar-binding transcriptional regulator in a novel gene locus in Enterococcus faecalis contributes to production of biofilm and prolonged bacteremia in mice. J Infect Dis., 2004; 189: 420–430.
- Kristich CJ, Li YH, Cvitkovitch DG & Dunny GM . Esp-independent biofilm formation by Enterococcus faecalis. J Bacteriol., 2004; 186: 154–163.
- Mohamed JA & Murray BE . Lack of correlation of gelatinase production and biofilm formation in a large collection of Enterococcus faecalis isolates. J Clin Microbiol., 2005; 43: 5405–5407.
- Hancock LE & Perego M . Systematic inactivation and phenotypic characterization of two-component signal transduction systems of Enterococcus faecalis V583. J Bacteriol., 2004; 18: 7951–7958.
- Eaton TJ & Gasson MJ. Molecular screening of Enterococcus virulence determinants and potential for genetic exchange between food and medical isolates. Appl Environ Microbiol., 2001; 67: 1628–1635.
- Gilmore MS, Huycke MM, Daniel FS. Multidrug-resistant Enterococci. The nature of the problem and an agenda for the future. Emerg Infect Dis 1998; 4: 239-49.
© The Author(s) 2016. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


