ISSN: 0973-7510
E-ISSN: 2581-690X
A Streptomyces consortium was developed and investigated for biodegradation of reactive sulfonated di azo dye (Reactive Blue 222). The biodegradation of azo blue dye by the consortium was further assessed with different ratios of inoculum of two different Streptomyces potential isolates. The optimization of process conditions was further carried out with respect to the dye concentrations, pH and temperature for the potential Streptomyces consortium. The Streptomyces consortia of 2:1 ratio was proved to enhance the biodegradation of azo blue dye to 89.39%. Dye concentration of 300 mg/L, pH of 7.0 and temperature of 35 °C showed the maximum degradation of 88.33 %, 89.06 % and 89.47 % respectively.
Consortium, Streptomyces, Optimization, Azo blue, Biodegradation.
In most of the instances decolorization of dyes with pure culture was proved to be impractical, as the isolated culture would be dye specific and their application in large scale wastewater treatment plants with a variety of contaminant dyes was not feasible (Vijaraghavan, 2008). For environmental remediation, microbial consortia are found to be efficient (Senan et al, 2004). Mixed cultures and co cultures differ in their performance than from microbial monocultures. Interspecific interactions of microbes were known to be very important for their metabolic cooperation among the mixed cultures (Seneviratne et al., 2008). Microbial consortium systems can provide advantages over individual cultures as they involve the combined and inductive effects of various enzymes which can work synergistically. The complexity of microbial consortium enables them to act on a variety of pollutants (Watanabe and Baker, 2000).
Though most of the research works on dye degradation have been carried out using fungal and bacterial consortia but the work pertained to indigenously isolated Streptomyces synergism for biodegradation and detoxification of azo dye is missing. Keeping this view as well as to overcome the problems of partial degradation, long reaction time and formation of toxic metabolites, a Streptomyces consortium was developed and investigated for biodegradation of reactive sulfonated di azo dye – reactive blue 222. Degradation of reactive sulphonated di azo dye – reactive blue 222 which was known to be recalcitrant, was investigated at different ratios of Streptomyces consortia, pH and Temperature.
According to Slater and Lovatt (1984), it is a commonly accepted observation that due to the concerted activity of a multimember consortium, often the biodegradation rate of a compound is faster in nature. Watnable (2000) stated that, the complexicity of microbial consortium enables them to degrade variety of pollutants. This phenomenon of degrading the variety of pollutant is generally due to syntrophic interactions among the mixed communities which lead to complete mineralization of azo dyes (Chang et al., 2004; Fude et al., 1994; Khehra et al., 2005; Asgher et al., 2007). Degradation by consortia of microorganisms / mixed culture naturally enhances the process of biodegradation since, individual strains attack the dye molecule at different positions or they may utilize the decomposed products produced by one strain which will be further decomposed by another strain (Mohana et al., 2008; Coughlin et al., 1997; Schliephake et al., 2000). The efficacy of decomposition considerably depends on the chemical character of the synthetic dye and biodegradation capacity of the microbial consortium (Chang et al., 2001). No data were available on the biodegradation of reactive blue 222 by Streptomyces consortium but limited data were found on biotreatment of C.I. Acid Red 119 (AR-119) acidic diazo dye and that to with an initial dye concentration of only 40 mg/ L by consortium (Khehra et al., 2005).
Formulation of consortia for effective dye degradation
The previously isolated two potential isolates of Streptomyces DJP15 and Streptomyces DJP27 were assessed in combination for effective degradation of azo blue dye. Effect of inoculum size in different combinations was mainly focused for effective dye degradation, as per the method of Senan and Abraham (2004) and Khadijah et al., (2009).
Each 1mL spore suspension of 3 days old cultures of both potential isolates of Streptomyces: (Streptomyces DJP15 and Streptomyces DJP27) were inoculated to 100 mL conical flask containing 50 mL of starch casein broth (pH 7.0) and incubated at 35 °C for 5 days to develop the consortia. Three different ratios (v/v) of consortia were formulated by inoculating both isolates of Streptomyces DJP15 and Streptomyces DJP27 at different ratios of inoculum size, namely1:1%, 2:1 % and 1:2% respectively. Azo blue dye at the concentration of 300 mg/L were added individually to the inoculated flask and incubated at 35 °C for 3 days. Samples of 5 mL culture broth were drawn at every 6 h and percent decolourisation was determined as mentioned earlier.
Optimization of conditions for effective dye degradation by consortia
Optimization of important conditions such as dye concentration, pH and temperature for effective dye degradation by the consortia of potential isolates of Streptomyces for azo blue dye was carried out as per the protocol prescribed by Dave and Dave (2009). Percent degradation of dye was determined by spectrophotometric method (Prachi and Anushree, 2009). 2:1 and 1:2 ratios of Streptomyces consortia were optimized for the degradation of azo blue dye. Effect of one parameter at a time, keeping others constant was followed.
Optimization of dye concentration
The maximum dye degradation under static state by the potential consortia of Streptomyces at different concentrations of dye was assessed following broth culture method. The azo blue dye was examined at the concentrations of 300, 350, 400, 450 and 500 mg/L. The percent dye degradation was calculated as mentioned earlier.
Optimization of pH
Various levels of pH were optimized for effective dye degradation by the potential consortia of Streptomyces, following broth culture method as mentioned above. pH 6.0, 6.5, 7.0, 7.5 and 8.0 of the medium were adjusted using dilute acidic and alkaline solution of hydrochloric acid and sodium hydroxide respectively. Percent dye degradation was calculated in different pH, at every 6 h of incubation, up to 3 days.
Optimization of temperature
Different ranges of temperatures were optimized for effective dye degradation by potential consortia of Streptomyces, following broth culture method. The effect of temperature on maximum dye degradation was examined by keeping the inoculated broth at 25, 30, 35, 40 and 45 °C. The percent dye degradation by the test isolates at different ranges of temperatures was calculated, at every 6 h, as mentioned earlier.
Our study demonstrates the efficient degradation of azo blue (Reactive Blue 222) by specific consortium of Streptomyces, developed by mixing two indigenously isolated strains of Streptomyces at different ratios. The three different ratios (1:1, 2:1 and 1:2) of consortium were developed by mixing two indigenously isolated strains of Streptomyces; Streptomyces DJP15 and Streptomyces DJP27 and particularly tested for their ability to check the enhanced degradation of blue dye.
The Streptomyces consortia of 1:1, 2:1 and 1: 2 ratios showed 69.69%, 89.39% and 73.23 % of azo blue degradation respectively (Fig. 1). All the three ratios of consortia showed their maximum percent degradation in less time of 36 h than the pure potential culture Streptomyces DJP15 which took 48 h of time for its maximum degradation. Among three ratios of consortia, least percent degradation (69.69 %) was exhibited by the consortia of ratio 1:1 at 36 h. The ratio 2:1 of the Streptomyces consortia proved to be the best one showing highest percent degradation (89.39 %) in short time of 36 h whereas the consortia of ratio 1:2 showed 73.23% degradation keeping its place between the best and the least degraders.
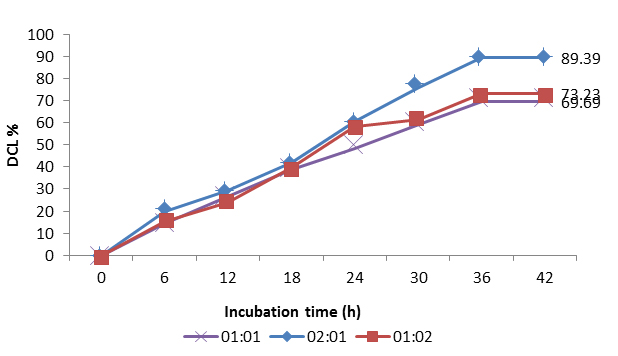
Fig. 1. Degradation of azo blue by different ratios of Streptomyces consortia
It was found from the study that, consortia with a ratio 2:1 are potential degrader for blue dye. It was noted that, in the biodegradation of blue dye, in all the ratios of consortia at every interval of 6 h incubation time, the percent degradation gradually increased indicating the degradation was synchronously related to growth of the isolates in the consortia.
Both isolates of Streptomyces were successfully existed as mixed cultures / co cultures at all the three different ratios of consortium in starch casein broth. The three ratios of consortia (1: 1, 2:1 and 1:2) of Streptomyces DJP15 and DJP27 showed varied degree of percent decolorization for azo blue. Significant reduction in the time (12h) for final degradation of blue dye by all three consortia was noted.
Optimization of conditions for effective dye degradation by consortium
Optimization of dye concentration
Figure 2 shows the effect of dye concentration on the degradation of azo blue by potential 2:1 ratio of Streptomyces consortium. It was observed that, at concentration of 300, 350, 400, 450 and 500 mg/L of reactive blue 222; 89.50 %, 81.81 %, 74.40 %, 66.07 % and 59.33 % degradation was observed. Increase in the concentration of dye, decreased the degradation efficiency of the potential consortia. Moreover, the consortia ratio showed considerable percent degradation (59.33 %) at high concentration (500 mg/L), was an significant nature of the consortia proving its tolerance property and ability to degrade more than 50 % at higher concentration of dye which was naturally lethal and toxic to most of the organisms. By this data and results it can be postulated that “Higher the concentration of the dye, lesser the degradation efficiency of the organisms”.
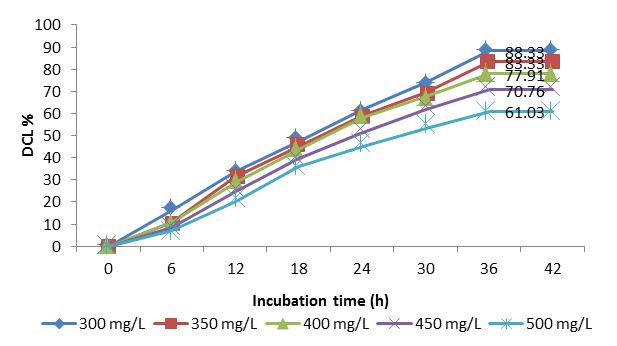
Fig 2: Degradation of azo blue by potential consortium at different dye concentration
Optimization of pH
Effect of pH on the degradation of azo blue by consortium of Streptomyces at 2:1 ratio was represented in Figure 3. 32.81 %, 42.70%, 89.06 %, 79.16 % and 68.22 % of azo blue degradation was recorded respectively over a pH range of 6.0, 6.5, 7.0, 7.5 and 8.0 at an incubation time of 36 h. Poor degradation activities of 32.81 % and 42.70 % were observed at acidic pH range of 6.0 and 6.5 respectively. Moderate percent degradation (79.16 %) was at pH 7.5 and good degradation (68.22 %) was recorded at alkaline pH 8.0; whereas the maximum degradation (89.06 %) was recorded at neutral pH for the consortia of 2:1 ratio.
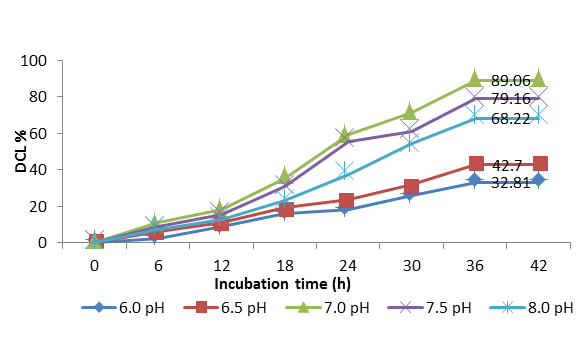
Fig 3: Degradation of azo blue by potential consortium at different pH
Figure 4 represents the effect of temperature on the degradation of azo blue by Streptomyces consortium at 2:1 ratio. Consortia exhibited moderate percent degradation over the temperature ranges of 30 °C – 40 °C; highest being 89.47 % at 35 °C. Beyond the 40 °C the degradation efficiency of the consortium decreased which was 60.52 % at 45 °C. Lowest percent degradation (35.78 %) was noticed at low temperature 25 °C. At 30 °C and 40 °C the percent degradation recorded were 65.78 % and 73.15 % respectively.
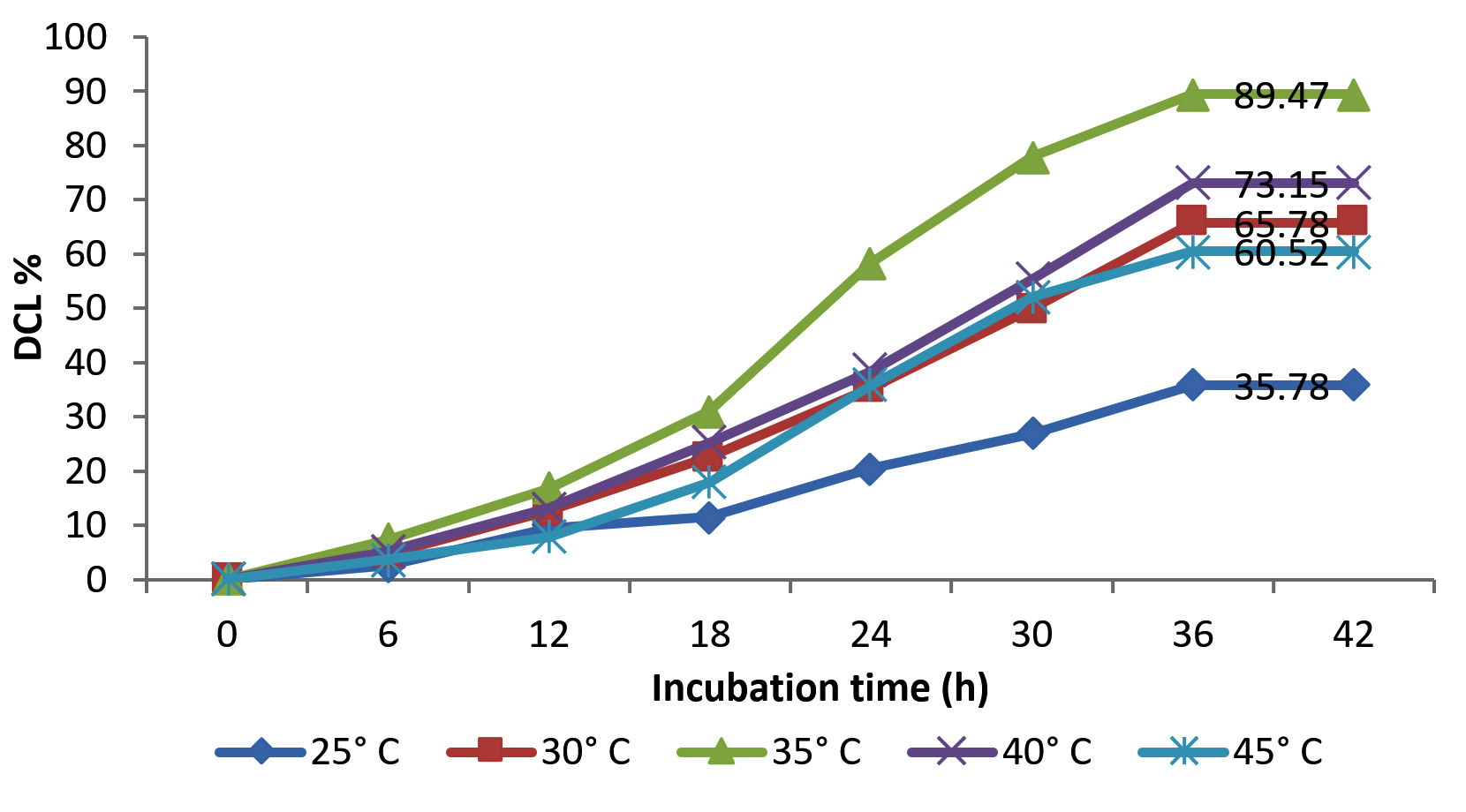
Fig. 4. Degradation of azo blue by potential consortium at different temperature
All the three of Streptomyces consortia developed in the present study exhibited their maximum final degradation at 36 h for the dye failing in their way to perform complete degradation whereas; the complete decolorisation of Acid Brilliant Scarlet GR was reported by Tan et al., (2012 a, b). Similar finding were also reported in the investigations of Buitro´n et al., (2004) and Davies et al., (2006), who investigated only one dye. Tan et al., (2012 a, b) also reported the further degradation of most of the aromatic intermediates by the consortium. Enhanced decolorization and degradation of azo dye Rubine GFL (50 mg/L within 30 h) using defined consortium GG-BL of Galactomyces geotrichum MTCC 1360 yeast and Brevibacillus laterosporus MTCC 2298 bacterium was reported by Waghmode et al., (2012) whereas, the individual cultures fails to completely decolorize the dye. Decolorization of Navy blue HE2R by developed consortium-PA of A. ochraceus NCIM-1146 fungi and Pseudomonas Sp SUK1 bacterium was reported (Kadam et al., 2011). The degradation efficiency of the consortium may be due to the synergetic actions of oxidoreductases (Gou et al., 2009; Telke et al., 2009b).
A striking observation was made with respect to the Streptomyces consortia of ratio 2:1 which exhibited an enhanced degradation of 11.50%, showing its pronounced capacity to degrade the blue dye with a maximum percent degradation of 89.39 % at 36 h at a concentration of 300 mg/L.
Pearce et al., (2003) reported that dye concentration could affect the efficiency of microbial decolorization through a combination of factors including the toxicity from higher dye concentrations and the accumulation of toxic decolorization intermediates.
Decolorization of azo dyes and simulated dye bath waste water using microbial consortium at 1 to 5 % inoculum size was reported by Dafale et al., (2008).
Synchronized action of microorganisms for the biodegradation of textile dye has been reported (Moosvi et al, 2005, Chen et al, 2006, Bafana et al, 2007, Yang et al., 2009, Patil et al., 2010, Phugare et al., 2011).
Our finding were in agreement with the reports of Nigam et al., (1996) and Sharma et al., (2004) who mentioned that, the higher degree of biodegradation and mineralization can be expected when metabolic activities of mixed cultures within a microbial community complement each other. They also stressed that the advantages of mixed cultures are apparent as some microbial consortia can collectively carry out biodegradation that cannot be achieved by pure culture.
Waghmode et al. (2012) reported the decolorization and biodegradation at 150-200 mgL-1 Rubin GEL concentration by microbial consortium GG-BL. In our study all the three ratios of consortia showed better degradation of blue dye. It was also observed that, the consortia have ability to degrade higher concentration of dye as reported by Ayed et al. (2010).
It is important to study the effect of pH on decolorization process, as transport of dye molecule into the cell is pH dependent and thought to be rate limiting step for decolorization of dyes (Lourenco et al., 2000). The potential consortia exhibited maximum degradation at pH 7.0 which was in accordance with the findings of Phugare et al., (2011) who studied the bacterial consortia SDS with maximum decolorization and degradation efficiency at pH 7.0. Optimum pH for the degradation activity for both the consortia were found to be in the narrow range of 7.0 to 8.0 pH as reported by Chaube et al., (2010). Kumar et al., (2009) recorded 96 % and 50% degradation of Remazol Black B at 7.0 pH and 5.0 pH respectively using pH sensitive consortium.
It was noted that, the ability of the consortia to show considerable degradation activity even at high temperature of 45°C. Maximum decolorization at 37°C by bacterial consortium was reported by Saratale et al., (2010).
Our finding were in agreement with the reports of Phugare et al., (2011 a) who found that, the bacterial consortium of P. aeruginosa BCH and Providencia Sp SDS had a potential to produce all the four dye decolorizing enzymes and gave a biodegradation advantage compared to the single bacterial strains.
In the bioremediation of xenobiotic anthropogens, application of mixed microbial consortium offers an advantage of complemented catabolic versatility. With a better understanding, this synergistic activity of Streptomyces consortia would be further exploited for the degradation of wide range of reactive azo dyes.
- Vijaykumar MH, Vaishampayan PA, Shouche YS, Karegouda TB. Decolourization of naphthalene-containing sulfonated azo dyes by Kerstersia sp. strain VKY1. Enzym Microb Technol, 2007; 40: 204–211.
- Senan, R.C., Abraham, T. E., Bioremediation of textile azo dyes by aerobic bacterial Consortium, Biodegradation, 2004; 15: 275-280.
- Seneviratne G, Zavahir JS, Bandara WMMS, Weerasekara MLMAW. Fungal–bacterial biofilms: their development for novel biotechnological applications. World J Microbiol Biotechnol, 2008; 24: 739–743
- Watanabe K & Baker PW. Environmentally relevant microorganisms. J. Biosci. Bioeng. 2000; 89: 1–11
- Slater JH, Lovatt D (1984) Biodegradation and the significance of microbial communities. In: Gibson DT (ed) Microbial degradation of organic compounds. Marcel Dekker, NY and Basel, pp 439–485
- Watanabe K & Baker PW. Environmentally relevant microorganisms. J. Biosci. Bioeng, 2000; 89: 1–11
- Chang, Jo-Shu.; Chen, Bor-Yann.; Lin, YS. Stimulation of bacterial decolorization of an azo dye by extracellular metabolites from Escherichia coli strain NO3. Bioresource Technology. 2004, 91: 243-48.
- Fude L, Harris B, Urrutia MM, Beveridge TJ. Reduction of Cr (VI) by a Consortium of Sulfate-Reducing Bacteria (SRB III). Appl Environ Microbiol, 1994; 60: 1525–1531.
- Khehra MS, Saini HS, Sharma DK, Chadha BS, Chimni SS. Decolorization of various azo dyes by bacterial consortium. Dyes Pigments, 2005; 67: 55–61. doi:10.1016/j. dyepig.2004.10.008
- Asgher, M., Bhatti, H. N., Shah, S. A. H., Asad, M. J., & Legge, R. L. Decolourization potential of mixed microbial consortia for reactive and disperse textile dyestuffs. Biodegradation, 2007; 18: 311–316.
- Mohana S, Srivastava S, Divecha J, Madamwar D. Response surface methodology for optimization of medium for decolorization of textile dye direct black 22 by novel bacterial consortium. Bioresour Technol, 2008; 99: 562– 569. doi:10.1016/j.biortech.2006.12.033
- Coughlin, M.F., Kinkle, B.K., Tepper, A., & Bishop, P.L. Characterization of aerobic azo dye degrading bacteria and their activity in biofilms. Water Sci Technol, 1997; 36: 215-220.
- Schliephake K, Mainwaring DE, Lonergan GT, Jones IK, Baker WL. Transformation and degradation of the disazo dye Chicago Sky Blue by a purified laccase from Pycnoporus cinnabarinus. Enzyme Microb Technol, 2000; 27:100–107.
- Chang, J.S., Chou, C., Lin, Y.C., Lin, P.J., Ho, J.Y. and Lee, Hu., T. Kinetic characteristics of bacterial azo dye decolorization by Pseudomonas luteola. Water Research., 2001; 35: 2841–2850.
- Khehra MS, Saini HS, Sharma DK, Chadha BS, Chimni SS. Decolorization of various azo dyes by bacterial consortium. Dyes Pigments, 2005; 67: 55–61. doi:10.1016/j. dyepig.2004.10.008.
- Senan, R.C., Abraham, T. E., Bioremediation of textile azo dyes by aerobic bacterial Consortium, Biodegradation, 2004; 15: 275-280.
- Khadijah, O., Lee K.K. and Abdullah, M.F., Isolation, screening and development of local bacterial consortia with azo dyes decolorizing capability, Mal. J. Microbiol. 2009; 5(1): 25- 32.
- Dave, S.R. and Dave, R.H. Isolation and characterization of Bacillus thuringiensis for Acid red 119 dye decolourization. Bioresource Technology., 2009; 100: 249- 253.
- Prachi Kaushik and Anushree Malik., Microbial decolourisation of textile dyes through isolates obtained from contaminated sites. Journal of scientific and industrial research, 2009; 68: 325-331.
- Tan HQ, Li TT, Zhu C, Zhang XQ, Wu M, Zhu XF. Parabacteroides chartae sp. nov., an obligately anaerobic species from wastewater of a paper mill. Int J Syst Evol Microbiol, 2012a; 62(Pt11):2613–2617
- Tan L, Ning S, Wang Y, Cao X. Influence of dye type and salinity on aerobic decolorization of azo dyes by microbial consortium and the community dynamics. Water Sci Technol, 2012b; 65(8):1375–1382.
- Buitro´n G, Quezada M, Moreno G. Aerobic degradation of the azo dye acid red 151 in a sequencing batch biofilter. Bioresour Technol, 2004; 92(2):143–149.
- Davies LC, Pedro IS, Novais JM, Martins-Dias S. Aerobic degradation of Acid Orange 7 in a vertical-flow constructed wetland. Water Res, 2006; 40(10):2055–2063
- Tan HQ, Li TT, Zhu C, Zhang XQ, Wu M, Zhu XF. Parabacteroides chartae sp. nov., an obligately anaerobic species from wastewater of a paper mill. Int J Syst Evol Microbiol, 2012a; 62(Pt11):2613–2617
- Tan L, Ning S, Wang Y, Cao X. Influence of dye type and salinity on aerobic decolorization of azo dyes by microbial consortium and the community dynamics. Water Sci Technol, 2012b; 65(8):1375–1382.
- Waghmode, T.R., Kurade, M.B., Lade, H.S., Govindwar, S.P., Decolorization and Biodegradation of Rubine GFL by microbial consortium GG-BL in sequential aerobic / microaerophilic process. Applied Biochemistry and Biotechnology. 2012; http://dx.doi.org/10.1007/s12010-012-9585-z.
- Kadam, A.A., Telke, A.A., Jagtap, S.S., Govindwar, S.P., Decolorization of adsorbed textile dyes by developed consortium of Pseudomonas sp. SUK1 and Aspergillus ochraceus NCIM-1146 under solid state fermentation. Journal of Hazardous Materials 2011; 189: 486-494.
- Gou M, Qu Y, Zhou J, Ma F, Liang T. Azo dye decolorization by a new fungal isolate, Penicillium sp. QQ and fungal–bacterial cocultures. J Haz Mat, 2009; 170:314–319.
- Telke, A.A., Kalyani, D., Dawkar, V. and Govindwar, S. Influence of organic and inorganic compounds on oxidoreductive decolourization of sulfonated azo dye CI Reactive Orange 16. J Hazard Mater, 2009; 172:298–309.
- Pearce CI, Lloyd JR, Guthrie JT. The removal of colour from textile wastewater using whole bacterial cells: a review. Dyes Pigments, 2003; 58: 179–196
- Dafale N, Rao NN, Meshram SU and Wate RS. Decolorization of azo dyes and simulated dye bath wastewater using acclimatized microbial consortium- biostimulation and halo tolerance. Bioresource Technol. 2008; 99: 2552-2558.
- Moosvi S, Keharia H, Madamwar D. Decolorization of textile dye reactive violet 5 by newly isolated bacterial consortium RVM-11.1. World J Microbiol Biotechnol, 2005; 21:667–672.
- Chen, B.Y., Chen, S.Y., Lin, M.Y., Chang, J.S., Exploring Bioaugmentation strategies for azo-dye decolorization using a mixed consortium of Pseudomonas luteola and Escherichia coli, Process Biochemistry, 2006; 41: 1574-1581
- Bafana, A., Devi, S.S., Krishnamurthy, K., Chakrabarti, T., Kinetics of decolorization and biotransformation of direct black 38 by C.hominis and P.stutzeri, Applied Microbiology Biotechnology, 2007; 31: 94- 97.
- Yang, Q., Li, C., Li, H., Li, Y., Yu, N., Degradation of synthetic reactive azo dyes and treatment of textile wastewater by a fungi consortium reactor, Biochemical Engineering Journal, 2009; 43: 225-230.
- Patil, P.S., Phugare, S.S., Jadhav, S.B., Jadhav, J.P., Communal action of microbial cultures for RED HE3B degradation, Journal of Hazardous Materials, 2010; 181: 263-270.
- Phugare, S.S., Kalyani, D.C., Patil, A.V., Jadhav, J.P., Textile dye degradation by bacterial consortium and subsequent toxicological analysis of dye and dye metabolites using cytotoxicity, geneotoxicity and oxidative stress studies, Journal of Hazardous Materials, 2011; 186: 713-723.
- Nigam P, Banat IM, Singh D, Marchant R. Microbial process for the decolorization of textile effluent containing azo, diazo and reactive dyes. Process Biochem, 1996; 31: 435–442
- Sharma DK, Saini HS, Chimni SS, Chadha BS. Biological treatment of textile dye Acid violet-17 by bacterial consortium in an up-flow immobilized cell bioreactor. Lett Appl Microbiol, 2004; 38: 345–350.
- Waghmode, T.R., Kurade, M.B., Lade, H.S., Govindwar, S.P., Decolorization and Biodegradation of Rubine GFL by microbial consortium GG-BL in sequential aerobic/microaerophilic process. Applied Biochemistry and Biotechnology, 2012. http://dx.doi.org/10.1007/s12010-012-9585-z.
- Ayed L, Harbi B, Cheref A, Bakhrouf A and Achour S. Application of the mixture design to optimize the formulation of active consortia to decolorize azo dye methyl red. Water Sci. Technol. 2010; 62(12): 2837-2845.
- Lourenco, N.D., Novais, J.M., Pinheiro, H.M., Reactive textile dye colour removal in a sequencing batch reactor. Water Science and Technology, 2000; 42: 321-328.
- Phugare SS, Kalyani DC, Patil AV, Jadhav JP. Textile dye degradation by bacterial consortium and subsequent toxicological analysis of dye and dye metabolites using cytotoxicity, genotoxicity and oxidative stress studies. J Haz Mat, 2011a; 186(1):713–723.
- Chaube P, Indurkar H and Moghe S. Biodegradation and decolorization of dye by mix consortia of bacteria and study of toxicity on Phaseolus mungo and Triticum aestivum. Asiatic J. Biotechnol. Resources. 2010; 1: 45-56.
- Kumar K, Dastidar MG, Sreekrishnan TR. Effect of process parameters on aerobic decolourization of reactive azo dye using mixed culture. World Acad Sci Eng Tech, 2009; 58: 962–965.
- Saratale RG, Saratale GD, Chang JS and Govindwar SP. Decolorization and degradation of reactive dyes and dye wastewater by a developed bacterial consortium. Biodegradation, 2010; 21: 999-1015.
- Phugare SS, Kalyani DC, Surwase SN, Jadhav JP, Ecofriendly degradation, decolorization and detoxification of textile effluent by a developed bacterial consortium. Ecotoxicol Environ Saf, 2011b; 74:1288–1296
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


