ISSN: 0973-7510
E-ISSN: 2581-690X
Nine dairy cows seven months gestation received via the milk ducts formulation inactivated antigen protein adhesion to fibronectin of Staphylococcus aureus, Streptococcus agalactiae and Escherichia coli cells. The analysis of the immunoglobulins G by indirect ELISA against the applied antigens, showed that five of nine cows were stimulated with Escherichia coli, the case for the antigenic stimulus for Streptococcus agalactiae showed that eight of nine cows responded with good indices of significance. Finally, stimulus virulence proteins of Staphylococcus aureus adhesion to fibronectin, revealed response rates all cows (P <0.0002). Intraductal response to antigens promises to be a way for immunization to dairy cows in the drying stage.
Acute mastitis, inactivated antigens and analysis of immunoglobulins.
In order for the mammary gland to become infected with some bacteria, a physical contact between the pathogen and the host is necessary; Where the recognition receptors are exhibited between them, at that time, the innate immune system of the host responds through the interaction of molecules referred to as Pathogen Associated Molecular Patterns (PAMPs) [Ulevitch et al., 2000]. Mastitis cases by Gram negative bacteria are more common, being Escherichia Coli the most predominante; because it contains in its cell wall lipopolysaccharide (LPS) which is common for the whole family (Yoshimura et al., 1999). For the group of Gram positive bacteria, and peptidoglycan (PGN) and lipoteichoic acid (LTA) cell wall representing the repertoire of compounds recognition similar to (LPS) effects, but with other mediators recognition (Schroder et al., 2003).
Staphylococcus aureus is one of the bacteria colonizing the mammary gland tissue destruction effects with high quality and low inflammatory response milk. Its reservoirs scarified skin of the nipple and the stratified epithelium of the nasal mucosa (Kluymants et al., 1997). Current vaccines for Staphylococcus aureus are made with whole bacteria and against the PGN (Baselga et al., 1994; Garcia et al., 1996; Kerro-Dego et al., 2006) and the immune response is limited given the numerous factors virulence that this bacterium exhibits (Anderson et al., 2012). However, facilitates recognition of PAMPs low immune response to infection, also facilitates persistent virus infections immunosuppressants as bovine viral diarrhea BVD, IBR Rhinotraqueitis Infectious Bovine viral Bovine Leukemia BVL (Wilson et al., 1997; Barkema et al., 1998).
The persistence of high risk bact erias in glandular acids such as Staphylococcus aureus, Streptococcus agalactiae, Mycoplasma bovis and some strains of Escherichia coli adherent and invasive, are associated with subclinical mastitis cases high (Dopfer et al., 2002). In turn, prepartum mediated immunosuppression stress and chain hormones are in part the result of dysfunction of phagocytosis of polymorphonuclear neutrophil leukocytes which leads to facilitation of colonization of bacteria in glandular structures generates loss and poor quality of milk production (Burvenich et al., 2003; Fox et al., 2005).
The purpose of this study was to analyze the immune response g enerada application by intra – ductal three bacterial antigens in dairy roof racks to determine the presence of specific immunoglobulins against antigens of the formula.
Bacterial antigens
The application of E coli obtained from cases of acute mastitis was carried out, as well as four serotypes previously characterized as producers of Pilis K-99, 39 and 35 ATCC collection of low and high production of labile thermosensitive and thermosensitive toxins. For Gram + bacteria such as Staphylococcus aureus and Streptococcus agalactiae, they were obtained from clinical cases of acute mastitis and their respective morphological and biochemical characterization until their characterization. The previously obtained recombinant adhesion protein to the fibronectin (Fnb) of Lactococcus lactis previously obtained on the wall of Lactococcus lactis, was also incorporated as a virulence protein that participates for adhesion to cells and host tissue.
Selection criteria for pre-calving and intramammary cows
In the region of Atlixco, Puebla-Mexico nine next to dry gestation diagnosed by palpation of about 7 months and low milk production in lactating cows were selected earlier. The herd of 200 breeding receives annual immunization against the virus of the respiratory and reproductive cattle complex, however, n or applied biological control of mastitis, this depends on control of hygiene and milking management on a daily basis.
Analysis of total protein and Immunoglobulins
Of each cow s blood samples were given Vacutainer tubes in obtaining d serum before and after 30 days of immunization. The serum is centrifuged do for 10 minutes at 1,000 rpm, was later deposited in Ependorff tubes and frozen at minus 20oC. Each sample of 2 cc was evaluated relative to the total protein concentration using a refractometer Brix ATAGO Master-M. For the determination of total protein busied 0.5 mL of serum, diluted 1:2 in buffer solution of phosphates (PBS), serum was transferred to a beaker on ice with magnetic stirring co ntinua, adding 1.5mL of ammonium sulfate at pH 7.8 up to saturation, subsequently conducted the analysis of the total protein concentration by the Bradford method which is based on the binding of blue dye Coomassie (G-250) to proteins. The acid solution dye is bound to form a protein-dye complex with a higher extinction coefficient than the free dye. This method is sensitive 1-15mg.
For precipitac ion total immunoglobulin was used 0.5 mL of serum from each sample of nine dairy cows, ammonium sulfate was incorporated in slowly and under constant stirring for 30 minutes at 4°C for precipitation of globulins. The precipitate was re – suspended by gentle agitation for five minutes and centrifuged at 10,000 G for 15 minutes at 4°C. E l supernatant was discarded and the button is re Cupero in 0.5 mL of PBS with a final saturation of 40% salt. Subsequently the fraction of globulins g was transferred to a dialysis bag against saline 0.85% making 6 changes using barium chloride in a 1:1 with water for 24 to 36 hours at 4°C until the ammonium sulfate it was no longer detectable. Globulins were pr eservadas frozen at 20oC.
Quantification of immunoglobulins in animals immunized by the ELISA method
The ELISA its acronym English Linked Immuno Sorbent-Enzyme Assay: is a specific technique to detect Bound antibodies an enzyme capable of generating a product (Lequin, 2005). Indirect ELISA test was developed for the quantitative determination of antibodies in serum in each of dairy cows for the different antigens. 100 µL of a 108 suspension of each bacterium to be tested was added in triplicate on 96-well ELISA plates (Costar). It was left at 4°C for 12 hours. subsequently they were washed with PBS and applied 100 µL of purified immunoglobulins as the first antibody, incubated at room temperature with moderate stirring for one hour. Do that time passes washed three times with PBS solution and applied 100 µL bovine IgG peroxidase – labeled goat as second antibody. It was read in spectrophotometer for ELISA, Diagnostic Pasteur model LP 400.
Immune System immune ma participates significantly in the resolution of infections of viral or bacterial origin. Some germs are producers of compounds that inhibit the immune system, so the chances of the germ (s) in colonizing and invading the host (Nakawa et al., 1993). In the particular case of bacterial infection in the mammary gland in the immediate postpartum period, polymorphonuclear neutrophils are inhibited in its function of phagocytosis, probably mediated by the stress of labor (Lee et al., 2003). Udders of dairy cows receiving immunization in the drying period, did not have any physical alteration, besides the electrical conductivity of the milk on the fifth day after postpartum, it turned with measurements above the cutpoint (300 ± 50), which means that the milk had no alterations of bacterial origin.
E. coli is one of the common pathogens found in milk immediate postpartum especially in cows second and third lactation where injury to the sphincter through the nipple are more frequent and loss of function predisposes to colonization of environmental bacteria (Russol et al., 2007). In this research c inco nine cows stimulated E antigens. coli showed higher antibody concentration in statistically significant (Fig. 1). The use of antibiotic products as a curative and / or preventive treatment in the drying stage is a common practice among milk producers and there is little control over their use (Russol et al., 2007). Also the control of antimicrobial resistance of bacteria that infect the mammary gland of cattle is nil or limited in Mexico.
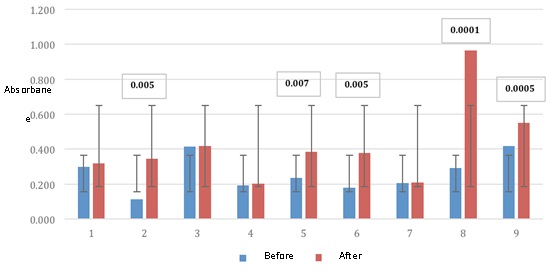 Fig. 1. ELISA absorbance values to E. coli
Fig. 1. ELISA absorbance values to E. coliThe production of specific immunoglobulins against epitopes of pili and fimbriae of E. coli mediated immunization are factors internal support for the facilitation of opsonization, control and resolution to challenges of E. coli in cattle milk production (EcoStaph PM + 3 2015) [González et al., 1989]. (EcoStaph PM + 3 2015) [González et al., 1989]. Although E. coli serotypes among animals livestock production species maintain their niches regarding colonization, it is necessary to mention that artisanal producers of fresh bovine milk cheese curdling made without pasteurization what it defines as potential zoonosis to those infections.
Streptococcus agalactiae is a bacterium streptococcus group B and other family that prevails in the intestinal tract of various mammals, including humans. In bovines it induces mastitis considered within the pathogens of importance by the reduction of milk production from which its surname derives. It is a germ with high sensitivity to antimicrobial beta lactams, so its treatment both lactating and the drying period eliminates infection (Rossitto et al., 2002), however, the antimicrobial resistance what little Is known in Mexico, is a factor that drives the improvement of hygiene and the application of vaccines.
Yokomizo and Norcross (1978), described the benefits in immunizing vaccine Streptococcus by way intra- parenteral and intro-mamaria, other experimental groups developed significant increase in IgG, 1 and 2, IgM and IgA. Furthermore, Lindahl et al. (2005) has developed vaccines for Streptococcus agalactiae neonatal infections linked to the men, where bovine strains involved are linked to virulence and capsule formation.
In this paper we find that the antigenic stimulus for S. agalactiae by way intra tanker, was favorable in eight of nine cows, which responded with good titers of antibodies to this antigen with gnificancia of p <0.0002 (Fig. 2) which confirms previous work by (Yokomizo and Norcross, 1978). Mastitis for S. agalactiae is common in ruminants such as; Cows, sheep, goats and monogastricos as; Bristles, bitches, cats, rabbits, etc. significantly reducing the quality of milk (Bergonier, 2014).
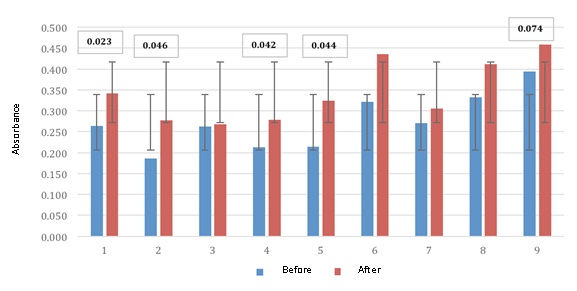 Fig. 2. ELISA absorbance values to Streptococcus agalactiae
Fig. 2. ELISA absorbance values to Streptococcus agalactiaeThe absorbance values in the ELISA test for Staphylococcus aureus were concentration values for inmunoglobulinas with some statistical significance for 2 of 9 cows (Fig. 3). These results are in agreement with previous studies where the low response.
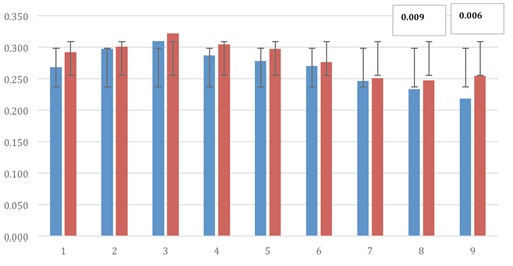 Fig. 3. ELISA absorbance values Staphylococcus aureus
Fig. 3. ELISA absorbance values Staphylococcus aureusHowever, stimulation with the virulence protein adhesion to fibronectin S. aureus, revealed response in all cows with signi cance rates between p <0.0002 (Fig. 4).
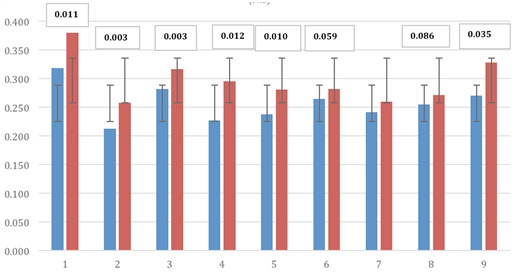 Fig. 4. ELISA absorbance values at (Fnb), S. aureus protein Recombinant fibronectin binding
Fig. 4. ELISA absorbance values at (Fnb), S. aureus protein Recombinant fibronectin bindingOn the other hand, the immuno stimulation via the milk duct, resulted in production of immuno globulins specific G with significance values in; E coli, Streptococcus agalactiae and Protein FNB of the S. aureus.
Antigens applied and its response indicates that the tank via intra can be used for the induction of immune response in the drying step. Uncapsulated lymphoid tissue located in the Fürstenberg rosette and permeability of the epithelium of that region suggest sites of antigen presentation to immune cells. It remains to be studied the production of IgA by this route where the mucosal response does with synthesis of this immunoglobulin. Staphylococcus aureus bacteria from the group of Gram positive found more frequently in cases of bovine mastitis (Barkema et al., 1998). The infections whose origin was S. aureus infections occur with sub clinical or chronic medical that usually persist for the life of the infected animal.
The results described in table 1, show that the sera prepartum of 5 cows were lower compared to sera obtained after delivery. The study by refractometer is often used to assess concentrations of immunoglobulins in the colostrum milk, however, their sensitivity limits are not as accurate as others.
Table (1):
Protein measurement by refractometer.
Sample number |
Brix Pre-delivery whey |
Brix Postpartum Sera |
|---|---|---|
10 |
8 |
10 |
19 |
9 |
10 |
23 |
10 |
11 |
65 |
7 |
9 |
307 |
9 |
9 |
410 |
10 |
10 |
484 |
7 |
12 |
798 |
11 |
11 |
990 |
10 |
10 |
The age of the cow, nutritional status, its environment and vaccination schedules will determine the quality and quantity of serum immunoglobulins present in the bloodstream. During the pre-natal period, the cow disseminate their blood capillaries, into the light immunoglobulins of glandular acinos of the mammary gland, forming the protein rich secretion, hormonal factors, lipids, known as colostrum. In cattle, there are particular conditions of immune suppression “stress” pre and post delivery, which favors the invasion of opportunistic pathogens to different organs such as the uterus, vagina and mammary gland (Lioyd, 1993).
- Lee, J.W., Paape, M.J., Elsasser, T.H. Zhao, X. Recombinant soluble CD14 reduces severity of intramammary infection by Escherichia coli. Infect. Immun, 2003; 71:4034.
- Lindahl, G.M. Surface proteins of Streptococcus agalactie and related proteins in other bacterial pathogens. Clinical Microbiology Reviews, 2005; 18(1): 102-127.
- Lioyd, S.C. Effect of pregnancy and lactation upon infection. Vet Imm and Immpath, 1993; 4: 153-176.
- Burvenich, C., Van Merris, V., Mehrzad, J., Diez-Fraile, A., Duchateau, L. Severity of E. coli mastitis is mainly determined by cows factors. Vet res, 2003; 34: 521-564.
- Schroder, N.W., Morath, S., Alexander, C., Hamann, L., Hartung, T., Zahringer, U., Gobel, U., Weber, J.R., Schumann, R. Lipoteichoic acid (LTA) of Streptpcoccus pneumoniae and Staphylococcus aureus activates immune cells via Toll-like receptor (TLR)-2, lipopolysaccharide-binding protein (LBP), and CD14, whereas TLR-4 and MD-2 are not involved. J.Biol. Chem, 2003; 278:15587.
- Wilson, D.J., Gonzalez, R.N., Das, H.H. Bovinemasitis pathogens in New York and Pennsylvania: prevalence and effects on somatic cell count and milk production. J. Dairy Sci, 1997; 80:2592-2598.
- Yoshimura, A., Lien, E., Ingalls, R.R., Tuomanen, E., Dziarski, R., Golenbock, D. Cutting edge: recognition of Gram-positive bacterial cell wall components by the innae immune system occurs via Toll-like receptor 2. J. Immunol, 1999; 163(1):1-5.
- Yokomizo, Y., Norcross, N.L. Bovine antibodie against Streptococcus agalactie, type Ia, produced by prepaturient intramamary and systemic vaccination. Am JVet Res, 1978; 39: 511-516.
- Anderson, A.S., Miller, A.A., Donald, R.G., Scully, I.L., Manura, J.S., Cooper, D., Jansen, K.U. Development of a multicomponent Saphylococcal aureus vaccine designed to counter multiple bacterial virulence factors. Human vaccines and immunotherapeutics, 2012; 8(11): 1585-1594.
- Burvenich, C.V. Severity of E. coli mastitis is mainly determinated by cow factors. Vet. Res, 2003; 34(5): 521-564.
- Baselga, R., Alvizu, I., Amorena, B. Staphylococcus aureus capsule and slime as virulence factors in ruminant mastitis. A review. Vet Microbiol, 1994; 39: 195-204.
- Barkema, H.W., Schukken, Y.H., Lam, T.JM., Beiboer, L., Wilmink, H., Benedictus, G., Brand, A. Incidence of clinical mastitis in dairy herds grouped in three categories by bulk: milk somatic cell counts. J. Dairy Sci, 1998; 81:411.
- Bergonier, D.D. Staphylococcus aureus from 152 cases of bovine, ovine and caprine mastitis investigated by Multiple-locus variable number of tandem repeat analysis (MLVA). BioMed Central, 2014; 45(97): 1-8.
- Dopfer, D.R., Almeida, T.J., Lam, H., Nederbragt, S.P., Oliver, W.G. Adhesion and invasion of Escherichia coli from single and recurrent clinical cases of bovine mastitis in vitro. Vet Microbiol, 2002; 74: 331-343.
- Fox, L.K., kirk, J.H., Britten, A. Mycoplasma mastitis: a review of transmission and control. J Vet Med B Infect Dis Vet Public Health, 2005; 52:153.
- García, V., Gomez, M., Iglesias, M., Sanjuan, N., Gherardi, M., Cerquetti, M.C., Sordeli, D. Intramammary inmunization with liv-attenuated Staphylococcus aureus: microbiological and inmmunological studies in a mouse mastitis model. FEMS Immunol Med Microbiol, 1996; 14: 45-51.
- Kluymants, J., Velcum, V.A., Verbrugh, H. Nasal carriage of Staphylococcus aureus. Epidemiology underlying mechanims and associated risk. Clinical Microbiology Reviews, 1997; 505-520.
- Kehrli, M.E., Shuste, D.E. Factors affecting milk somatic cells and their role in health of bovine mammary gland. J. Dairy Sci, 1994; 7(2):619-627.
- Kerro-Dego, O., Prysliak, T., Potter, A.A, Pérez-Casal, J. DNA-protein inmmunization agains de GapB and GapC proteins of mastitis isolate of Staphylococcus aureus. Vet immuno Immunophatol, 2006; 113: 125-38.
- Nakawa, J.S., Araki, S., Kimura, M. Effects of active egg white product on neutrophil funtion in calves. J Vet Med Sci, 1993; 5:259-263.
- Russo, T.A., Beanan, J.M., Olson, R.,. Genagon, S.A., MacDonald, U., Cope, J.J, Davidson, B.A., Johnston, B., Johnson, R. A killed, genetically Engineered derivative of a wild-type extraintestinal pathogenic E. coli strain is a vaccine candidate, Vaccine, 2007; 25(19): 3859-3870.
- Rossitto, P.V. Antibiotic susceptibility patterns for environmental Streptococci isolated from bovine mastitis in central California dairies. Journal of Dairy Sci, 2002; 85(1): 132- 138.
- Lequin, R.M. Enzyme immunoassay (EIA)/enzyme-linked immunosorbent assay (ELISA). Clinical Chemistry, 2005; 51(12): 2415-2418.
© The Author(s) 2017. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


