ISSN: 0973-7510
E-ISSN: 2581-690X
Turcicum leaf blight (TLB) is the most important disease of maize growing areas of India. To identify new resistance sources and establish durability of known resistance sources, 30 maize inbred lines were evaluated against TLB under artificial inoculation during kharif 2013 and kharif 2014. Ten inbred lines were found resistant against TLB. Based on per se performance and resistance to TLB nine inbred lines viz., DMIT 105, DMIT 106, DMIT 111, DMIT 113, DMIT 118, DMIT 121, DMIT 123, DMIT 124 and DMIT 125 were used to develop 36 single cross hybrids and were screened to identify resistant hybrids. Thirty two hybrids were found resistant with disease score 1 and 2. Based on percent disease index (PDI) and area under disease progress curve (AUDPC) values, the components of blighting, inbred lines viz., DMIT 105, DMIT 113, DMIT 118 and DMIT 126 and hybrids viz., DMIT 105 × DMIT 125, DMIT 106 × DMIT 111, DMIT 106 × DMIT 121, DMIT 106 × DMIT 125, DMIT 111 × DMIT 113, DMIT 111 × DMIT 121, DMIT 113 × DMIT 118, DMIT 113 × DMIT 121, DMIT 113 × DMIT 125, DMIT 118 × DMIT 121, DMIT 118 × DMIT 124 and DMIT 121 × DMIT 123 were identified as slow blighters. These hybrids should be involved in the crop improvement programmes.
Hybrids, Inbred lines, Maize, PDI, Turcicum leaf blight.
Maize (Zea mays L.) is one of the world’s three most important cereal crops along with rice and wheat. India is rich in maize germplasm particularly of tropical and subtropical types with maximum variability and adaptability. Due to moderate low temperature and high humidity during the maize growing period, turcicum leaf blight of maize (syn. Northern leaf blight) caused by Exserohilum turcicum (Pass.) Leonard and Suggs is recurrent problem in most maize growing regions of India. Most of the cultivated genotypes are more or less susceptible to this disease and the loss in yield has been reported to vary from 28 to 91 per cent (Harlapur et al., 2000). Various options are available to control maize leaf blight such as the use of host plant resistance, cultural practices and fungicides. Host plant resistance is the cheapest and most effective way to control leaf blight disease because chemical treatments are expensive and often ineffective.
Slow blighting is a form of resistance, where despite a susceptible host reaction, the rate of disease development is very slow. Slow blighting is expressed in the reduced infection of a plant by a blight fungus, late appearance of blight in the life cycle of the host and retarded development of the fungus. Partial resistance is a form of incomplete resistance, characterized by a reduced rate of epidemic development (Parlevliet, 1979). The phenomenon of slow blighting was observed in Helminthosporium leaf blight of wheat (Patil, 2000) and partial resistance to TLB of maize (Mallikarjuna, 1998). The utilization of resistance source in breeding programme requires detailed information on various components of resistance under field conditions. Hence a field study was undertaken to determine the turcicum leaf blight response of 30 maize inbred lines to development of resistant single cross maize hybrids and to evaluate the resultant hybrids against TLB to identify resistant hybrids and blighting reaction.
The experiments were conducted at Main Agricultural Research Station, maize Scheme, University of Agricultural Sciences, Dharwad, Karnataka (15° 25’ N latitude, 70° 25’ East longitude) with an altitude of 678 m above mean sea level. Each test line was sown in 2 rows of 4m length and rows were spaced at 60 cm in randomized block design with two replications. The test genotypes were inoculated with E. turcicum multiplied on sorghum grain culture by whorl drop method. Inoculation was done twice at 35 and 45 days after sowing (DAS) followed by water spray so as to maintain required humidity for successful infection. Spreader rows of highly susceptible inbred CM 202 were planted at the border and at regular interval as a source of secondary inoculum for disease development. Observations on blight severity was recorded at the time of tasselling, 20 days after tasselling and at maturity using 1-5 scale (Payak and Sharma, 1983). Based on this scale, the genotypes were classified into three groups viz., resistant (disease score ≤2), moderately resistant (disease score 3) and susceptible (disease score >4). Further, PDI (Wheeler, 1969) and AUDPC (Wilcoxson et al., 1975) were calculated by using the following formula.
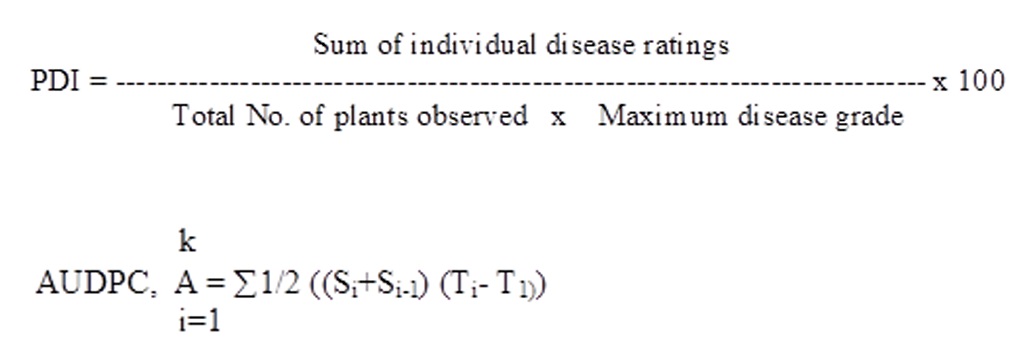 Where,
Where,
K= Number of successive observations
Si = Severity of disease at ith period
Si-1 = Severity of disease proceeding to ith period
Ti– T1 = time intervals between two observations
Screening of inbred lines
The experimental materials in the study composed of sixty inbred lines developed at Main Agricultural Research Station (MARS), maize Scheme, University of Agricultural Sciences, Dharwad and forty inbred lines received from Indian Institute of Maize Research, New Delhi. From one hundred lines, thirty resistant lines were selected based on disease reaction and per se performance during kharif-2013 and were selfed. These lines along with susceptible check (CM 202) were again grown during kharif-2014 under artificial epiphytotic condition to identify new sources of resistance against TLB. The list of selected resistant inbred lines and their pedigree are presented in Table 1.
Table (1):
List of inbred lines along with their pedigrees.
SI No |
Line No |
Pedigree/source population |
SI No |
Line No |
Pedigree/source population |
|---|---|---|---|---|---|
1 |
DMIT101 |
WNCDMR 19 RYDWS 1592 |
16 |
DMIT 116 |
KS × 4901 –X-X-X-X-14 |
2 |
DMIT 102 |
DMH 8255-6-8-4-48 |
17 |
DMIT 117 |
NK 6240 × CML 162-3-5-9-8 |
3 |
DMIT 103 |
900 Gold × NE 1412004-X-X-X-13 |
18 |
DMIT 118 |
D 9081-6-4-8-20 |
4 |
DMIT 104 |
900 Gold × NE 1412004-X-X-X-20 |
19 |
DMIT 119 |
900 GOLD × NE1412004-X-X-X-X-20 |
5 |
DMIT105 |
WNCDMR6RYFWS 8008 |
20 |
DMIT 120 |
CML146/CML176-B-29-1-3 |
6 |
DMIT 106 |
NK 6240 × K-155-X-X-X-18 |
21 |
DMIT 121 |
NK 6240 × CML 412-6-5-20 |
7 |
DMIT 107 |
BIO 6891-16-5-6 |
22 |
DMIT 122 |
WNCDMR11R 4788 |
8 |
DMIT108 |
SOS1YQBB26-B |
23 |
DMIT 123 |
CM 290 × CML 160-X-X-X-3 |
9 |
DMIT 109 |
CM 215 × CM 145-1-5-8-28 |
24 |
DMIT124 |
WNCDMR19RYDWS 1396 |
10 |
DMIT 110 |
NK 6240 × K-128-X-X-X-15 |
25 |
DMIT125 |
WNCDMR6RYFWS 8053 |
11 |
DMIT 111 |
NK 6240 × K-128-X-X-X-18 |
26 |
DMIT126 |
WNCDMR19RYDWS 1712A |
12 |
DMIT 112 |
CML 332 × CML 325-6-5-25 |
27 |
DMIT 127 |
VA-6-9-66 |
13 |
DMIT 113 |
PINNACLE × K148-X-X-X-X-X-16 |
28 |
DMIT128 |
WNCDMR6RYFWS 8105 |
14 |
DMIT 114 |
30V92 × K148-X-X-X-15 |
29 |
DMIT 129 |
WNCDMR11R6362 |
15 |
DMIT 115 |
NK 6240 × K-132-X-X-X-11 |
30 |
DMIT 130 |
WNCDWR10RYFWS 8627 |
Evaluation of hybrids
Among thirty six inbred lines evaluated, nine resistant (disease score £ 2) and good per se performance inbred lines viz., DMIT 105, DMIT 106, DMIT 111, DMIT 113, DMIT 118, DMIT 121, DMIT 123, DMIT 124 and DMIT 125 were selected for the production of single crosses. A total of 36 hybrids were produced in half diallel fashion during summer- 2014-15 and were screened for TLB under artificial epiphytotic condition during kharif 2015.
The inbred lines and hybrids of maize in present investigation were evaluated under artificial epiphytotic conditions during kharif 2014 and kharif 2015 to identify resistance source and to develop resistant maize hybrids against turcicum leaf blight (Helminthosporium turcicum).
Screening of inbred lines
Out of thirty inbred lines, ten lines viz., DMIT 105, DMIT 106, DMIT 111, DMIT 113, DMIT 118, DMIT 121, DMIT 123, DMIT 124, DMIT 125 and DMIT 126 recorded less than or equal to score 2 and grouped them as resistant; twelve inbred lines viz., DMIT 101, DMIT 103, DMIT 104, DMIT 107, DMIT 108, DMIT 109, DMIT 110, DMIT 112, DMIT 119, DMIT 122, DMIT 127 and DMIT 129 possessing score 3.0 were categorized as moderate resistant and eight inbred lines viz., DMIT 102, DMIT 114, DMIT 117, DMIT 120, DMIT 130, DMIT 115, DMIT 116 and DMIT 128 exhibited TLB score 4 were categorized as susceptible and susceptible check (CM202) had score 5 was categorized as highly susceptible to TLB (Table 2). These are in agreement with results obtained by earlier workers (Chandrashekara et al., 2014 and Singh et al. 2014) while working with turcicum leaf blight of maize.
Table (2):
Categorization of maize inbred lines based on the reaction to E. turcicum under artificial epiphytotic condition.
Reaction |
Score |
Inbred lines |
|---|---|---|
Resistant |
≤2 |
DMIT105, DMIT106, DMIT111, DMIT113, DMIT118, DMIT121, DMIT123, DMIT124, DMIT125, DMIT126 |
Moderately resistant |
3 |
DMIT101, DMIT103, DMIT104, DMIT107, DMIT108, DMIT109, DMIT110, DMIT112, DMIT119, DMIT122, DMIT127, DMIT129 |
Susceptible |
4 |
DMIT102, DMIT114, DMIT117, DMIT120, DMIT130, DMIT115, DMIT116, DMIT128, DMIT135 |
Highly susceptible |
5 |
DMIT131, DMIT132, DMIT133, DMIT134, DMIT136 |
Evaluation of hybrids
The disease score at maturity ranged from 1 to 3 in hybrids and resistant check compared to susceptible check (score 5). Out of 36 hybrids, fourteen hybrids possessed a disease score of 1 which were found to be highly resistant. Eighteen hybrids scored disease score of 2 indicating that they were resistant to the disease while, remaining four hybrids recorded disease score 3, which were found to be moderately resistant to the disease (Table 3). Similar results were reported by Kumar and Salgotra (2015).
Table (3):
Categorization of maize hybrids based on the reaction to E. turcicum under artificial epiphytotic condition.
Reaction |
Score |
Hybrids |
|---|---|---|
Highly resistant |
1 |
DMIT106×DMIT121,DMIT106×DMIT125, DMIT111×DMIT113, DMIT111×DMIT118, DMIT111×DMIT121, DMIT111×DMIT124, DMIT113×DMIT118, DMIT113×DMIT121, DMIT113×DMIT125, DMIT118×DMIT121, DMIT118×DMIT123, DMIT118×DMIT124, DMIT118×DMIT125, DMIT121×DMIT123 |
Resistant |
2 |
DMIT105×DMIT111, DMIT105×DMIT113, DMIT105×DMIT121, DMIT105×DMIT124, DMIT105×DMIT125, DMIT106×DMIT111, DMIT106×DMIT113, DMIT106×DMIT118, DMIT106×DMIT123, DMIT106×DMIT124, DMIT111×DMIT123, DMIT111×DMIT125, DMIT113×DMIT124, DMIT121×DMIT124, DMIT121×DMIT125, DMIT123×DMIT124, DMIT123×DMIT125, DMIT124×DMIT125 |
Moderately resistant |
3 |
DMIT105×DMIT106, DMIT105×DMIT118, DMIT105×DMIT123, DMIT113×DMIT123 |
Per cent disease index (PDI)
Significant differences in disease severity were observed among the inbred lines and hybrids between days to tasselling and at maturity. The difference of genotypes in disease severity was due to diversity in their genetic makeup as reported by Williams and Hallauer (2000) and Kraja et al. (2000). Per cent disease index (PDI) and area under disease progress curve (AUDPC), the components of blighting, were calculated to identify slow blighting genotypes.
Per cent disease index shows the cumulative value of disease in the target genotypes. Its value is based on disease severity. Four inbred lines viz., DMIT 105, DMIT 113, DMIT 118 and DMIT 126, fifteen hybrids which were developed from resistant inbreds viz., DMIT 106 × DMIT 111, DMIT 106 × DMIT 121, DMIT 106 × DMIT 125, DMIT 111 × DMIT 113, DMIT 111 × DMIT 118, DMIT 111 × DMIT 121, DMIT 111 × DMIT 124, DMIT 113 × DMIT 118, DMIT 113 × DMIT 121, DMIT 113 × DMIT 125, DMIT 118 × DMIT 121, DMIT 118 × DMIT 123, DMIT 118 × DMIT 124, DMIT 118 × DMIT 125 and DMIT 121 × DMIT 123 and resistant check P 3051 showed not only delayed onset of the disease but also ended up with significantly lower disease severity (Table 4 and 5). In these genotypes, blight development was more or less the same during different periods of observation. Result indicates the resistance is oligogenic or polygenic with partial and race-nonspecific resistance. Hossain (1987) reported that maize genotypes Thaltzapam-8146 was found partially resistant to TLB. Sharma and Payak (1990) recorded durable resistance in two maize inbred lines CM-104 and CM-105 against Exserohilum turcicum and observed that these inbred lines were potential in transmitting resistance in hybrid combinations through additive gene action. Durability of their resistance was associated with polygenic control.
Table (4):
Per cent disease index (%) and area under disease progress curve of turcicum leaf blight in inbred lines of maize.
Inbred line |
PDI at tasselling |
PDI at 20 days after tasselling |
PDI at maturity |
AUDPC |
|---|---|---|---|---|
DMIT101 |
6.54 |
21.84 |
24.50 |
812.64 |
DMIT102 |
7.00 |
14.56 |
26.17 |
692.86 |
DMIT103 |
7.88 |
20.25 |
25.84 |
820.87 |
DMIT104 |
9.35 |
22.33 |
34.78 |
981.44 |
DMIT105 |
3.65 |
10.28 |
14.58 |
424.47 |
DMIT106 |
8.83 |
13.83 |
18.99 |
643.24 |
DMIT107 |
9.64 |
19.08 |
30.19 |
876.31 |
DMIT108 |
12.48 |
25.37 |
33.90 |
1095.9 |
DMIT109 |
8.18 |
19.00 |
25.73 |
800.98 |
DMIT110 |
10.43 |
22.69 |
28.96 |
951.99 |
DMIT111 |
7.48 |
16.08 |
22.37 |
694.94 |
DMIT112 |
14.69 |
27.15 |
36.63 |
1202.97 |
DMIT113 |
4.42 |
8.97 |
15.94 |
427.03 |
DMIT114 |
13.82 |
27.72 |
37.41 |
1204.81 |
DMIT115 |
16.43 |
30.05 |
41.49 |
1344.41 |
DMIT116 |
10.85 |
20.36 |
31.92 |
943.46 |
DMIT117 |
14.31 |
26.84 |
36.09 |
1183.75 |
DMIT118 |
2.95 |
8.06 |
14.66 |
366.65 |
DMIT119 |
5.51 |
22.50 |
27.10 |
831.26 |
DMIT120 |
14.42 |
29.01 |
51.27 |
1381.17 |
DMIT121 |
6.54 |
10.00 |
16.30 |
493.64 |
DMIT122 |
13.92 |
23.03 |
37.89 |
1117.82 |
DMIT123 |
8.48 |
12.99 |
18.81 |
617.42 |
DMIT124 |
8.45 |
13.32 |
21.44 |
649.75 |
DMIT125 |
7.09 |
15.49 |
19.27 |
644.32 |
DMIT126 |
3.03 |
9.26 |
14.18 |
387.62 |
DMIT127 |
17.34 |
23.13 |
28.88 |
1098.17 |
DMIT128 |
22.08 |
31.13 |
36.64 |
1430.55 |
DMIT129 |
14.27 |
25.53 |
40.35 |
1199.55 |
DMIT130 |
15.64 |
27.00 |
41.18 |
1264.73 |
CM 202 (Check) |
31.75 |
51.27 |
82.32 |
2483.56 |
CV |
9.07 |
12.76 |
7.15 |
|
CD@5% |
2.02 |
5.45 |
4.41 |
|
CD@1% |
2.71 |
7.33 |
5.93 |
Area Under Disease Progress Curve (AUDPC)
The AUDPC estimates the area under the actual infection curve. It is expressed as accumulation of daily percent infection values and interpreted directly without transformation. The higher the AUDPC, the more susceptible is the clone or variety. The AUDPC is calculated from all the three ratings at different time thus leading to a more accurate phenotypic evaluation. The loss of active leaf area results in less photosynthetic available region during the grain filling stage which eventually results in producing smaller kernels. This reduction may eventually contribute to the overall yield losses.
Area under disease progress curve was calculated for inbred lines, 36 hybrids and resistant and susceptible checks and it is presented in Table 4 and 5. The AUDPC values differed considerably for genotypes. The lowest AUDPC values were noticed in five inbred lines viz., DMIT 105 (424.47), DMIT 113 (427.03), DMIT 118 (366.65), DMIT 121 (493.64) and DMIT 126 (387.62). Among hybrids and checks, twelve hybrids viz., DMIT 105 × DMIT 125 (399.10), DMIT 106 × DMIT 111 (292.74), DMIT 106 × DMIT 121 (185.46), DMIT 106 × DMIT 125 (379.34), DMIT 111 × DMIT 113 (333.75), DMIT 111 × DMIT 121 (285.69), DMIT 113 × DMIT 118 (173.59), DMIT 113 × DMIT 121 (193.19), DMIT 113 × DMIT 125 (143.66), DMIT 118 × DMIT 121 (245.52), DMIT 118 × DMIT 124 (300.10) and DMIT 121 × DMIT 123 (289.25) and resistant check P 3051 (364.35) showed low AUDPC values. The high AUDPC values among hybrids were observed in the hybrid DMIT 113 × DMIT 123 (823.75) and in susceptible check CM 202 (2442.93). This is in accordance with the work of earlier reports (Mallikarjuna, 1998). In general, AUDPC values took care of initial and terminal severity and also rate of infection. Hence, genotypes with lower AUDPC values can be considered as slow blighters. It indicated that such genotypes were more tolerant to pathogen without any economic damage.
Table (5):
Per cent disease index (%) and area under disease progress curve of turcicum leaf blight in maize hybrids.
HYBRID |
PDI at tasselling |
PDI at 20 days after tasselling |
PDI at maturity |
AUDPC |
|---|---|---|---|---|
DMIT105×DMIT106 |
3.09 |
9.53 |
20.88 |
461.23 |
DMIT105×DMIT111 |
4.09 |
12.37 |
20.37 |
532.70 |
DMIT105×DMIT113 |
1.23 |
9.79 |
19.74 |
417.77 |
DMIT105×DMIT118 |
9.19 |
17.17 |
27.34 |
800.62 |
DMIT105×DMIT121 |
4.03 |
9.12 |
17.22 |
435.29 |
DMIT105×DMIT123 |
8.92 |
17.82 |
27.59 |
810.70 |
DMIT105×DMIT124 |
3.03 |
11.75 |
20.89 |
504.56 |
DMIT105×DMIT125 |
4.28 |
8.08 |
15.18 |
399.10 |
DMIT106×DMIT111 |
2.33 |
6.25 |
12.13 |
292.74 |
DMIT106×DMIT113 |
5.26 |
10.70 |
19.69 |
516.09 |
DMIT106×DMIT118 |
3.06 |
7.97 |
18.93 |
409.97 |
DMIT106×DMIT121 |
0.32 |
3.79 |
10.32 |
185.46 |
DMIT106×DMIT123 |
0.32 |
9.56 |
20.94 |
407.04 |
DMIT106×DMIT124 |
3.70 |
7.85 |
18.57 |
416.78 |
DMIT106×DMIT125 |
3.71 |
8.56 |
13.39 |
379.34 |
DMIT111×DMIT113 |
4.72 |
5.54 |
12.86 |
333.75 |
DMIT111×DMIT118 |
7.23 |
7.38 |
12.23 |
414.55 |
DMIT111×DMIT121 |
1.65 |
6.66 |
11.94 |
285.69 |
DMIT111×DMIT123 |
6.38 |
11.05 |
20.67 |
555.26 |
DMIT111×DMIT124 |
7.12 |
8.63 |
11.97 |
434.68 |
DMIT111×DMIT125 |
4.49 |
9.27 |
18.08 |
456.04 |
DMIT113×DMIT118 |
0.32 |
4.21 |
8.29 |
173.59 |
DMIT113×DMIT121 |
0.32 |
4.67 |
9.33 |
193.19 |
DMIT113×DMIT123 |
9.90 |
17.62 |
27.33 |
823.75 |
DMIT113×DMIT124 |
7.18 |
12.40 |
21.53 |
606.71 |
DMIT113×DMIT125 |
0.32 |
2.59 |
8.55 |
143.66 |
DMIT118×DMIT121 |
0.32 |
6.69 |
10.53 |
245.52 |
DMIT118×DMIT123 |
5.92 |
9.48 |
14.35 |
451.39 |
DMIT118×DMIT124 |
1.62 |
7.24 |
12.29 |
300.10 |
DMIT118×DMIT125 |
7.43 |
7.70 |
13.17 |
434.32 |
DMIT121×DMIT123 |
1.23 |
7.02 |
12.42 |
289.25 |
DMIT121×DMIT124 |
7.54 |
12.87 |
22.76 |
635.76 |
DMIT121×DMIT125 |
5.86 |
11.48 |
20.09 |
547.82 |
DMIT123×DMIT124 |
7.52 |
14.72 |
24.01 |
684.82 |
DMIT123×DMIT125 |
4.27 |
13.80 |
23.02 |
591.38 |
DMIT124×DMIT125 |
8.00 |
14.63 |
22.56 |
678.17 |
P3051 (Resistant check) |
4.16 |
7.92 |
12.28 |
364.35 |
CM 202 (Susceptible check) |
28.87 |
54.43 |
77.68 |
2442.93 |
CV |
16.93 |
6.64 |
4.53 |
|
CD@ 5 % |
1.78 |
1.51 |
1.79 |
|
CD@1 % |
2.38 |
2.02 |
2.40 |
Based on the results of the present study, it is concluded that the inbred lines viz., DMIT 105, DMIT 113, DMIT 118, DMIT 121 and DMIT 126 categorized as highly resistant and resistant can be well utilized successfully for developing hybrids and composites in future breeding programme.
In the cross combinations, the hybrids with disease score less than 2 for turcicum leaf blight along with desirable specific combining ability for grain yield and its component traits appear to be suitable for cultivation in blight predominant areas. In the present study, it is observed that the inbred lines viz., DMIT 105, DMIT 113, DMIT 118, DMIT 121 and DMIT 126 and hybrids viz., DMIT 105 × DMIT 125, DMIT 106 × DMIT 111, DMIT 106 × DMIT 121, DMIT 106 × DMIT 125, DMIT 111 × DMIT 113, DMIT 111 × DMIT 121, DMIT 113 × DMIT 118, DMIT 113 × DMIT 121, DMIT 113 × DMIT 125, DMIT 118 × DMIT 121, DMIT 118 × DMIT 124 and DMIT 121 × DMIT 123 possessed slow blighting characters. Thus, the slow blight resistant character is very important and can be used in selection process for developing the hybrid or used as such for cultivation. If slow blighting genotypes are widely used in a disease control strategy, the rate of leaf blight development will not only be reduced during the rainy season, but also during the subsequent rabi / summer seasons when the resistance of slow blighting genotype in adult plant stage of growth is operating. Therefore, deployment of the identified slow turcicum leaf blighting genotypes could be an important TLB management strategy in maize.
ACKNOWLEDGMENTS
The authors are grateful to Department of Plant Pathology, University of Agricultural Sciences, Dharwad, Karnataka for providing lab facilities.
- Harlapur, S.I., Wali, M.C., Anahosur, K.H. and Muralikrishna, S. A report on survey and surveillance of maize diseases in northern Karnataka. Karnataka. J. Agric. Sci., 2000; 13: 750-1.
- Parlevliet, J.E. Components of resistance that reduce the rate of epidemic development. Ann. Rev. Phytopath., 1979; 17: 203-22.
- Patil, V.S. Epidemiology and management of leaf blight of wheat caused by Exserohilum hawaiienesis (Bugnicourt) Subram and Jain, Ex. Ellis, M.B. Ph.D. Thesis, University of Agricultural Sciences, Dharwad, 2000; pp 97.
- Mallikarjuna, N. Studies on partial resistance to turcicum leaf blight (Exserohilum turcicum) in maize. M.Sc. (Agri.) Thesis, University of Agricultural Sciences, Bangalore, 1998; pp 116.
- Payak, M.M. and Sharma, R.C. Disease rating scales in maize in India. In: Techniques of Scoring for Resistance to Important Diseases of Maize. All India Coordinated Maize Improvement Project, Indian Agricultural Research Institute, New Delhi, 1983; pp 1-4.
- Wheeler, B.E.J. (ed): An Introduction to Plant Diseases. London, United Kingdom: John Wiley and Sons Ltd., 1969; pp 62-65.
- Wilcoxson, R.D., Skovmand, B. and Atif, A. Evaluation of wheat cultivars for ability to retard development of stem rust. Ann. Appl. Biol.. 1975; 80: 275-81.
- Chandrashekara, C., Jha, S.K., Arunkumar, R. and Agrawal, P.K. Identification of new sources of resistance to turcicum leaf blight and maydis leaf blight in maize (Zea mays L.). SABRAO J. Breed. Genet., 2014; 46 (1): 44-55.
- Singh, R., Srivastava, R.P., Mani, V.P., Khandelwal, R.S. and Lekha Ram. Screening of maize genotypes against northern corn leaf blight. The bioscan, 2014; 9: 1689-93.
- Kumar and Salgotra, S. K. Evaluation of maize hybrids against leaf blight (Helminthosporium maydis and H. turcicum) and brown spot diseases (Physoderma zea maydis) of maize under mid hills of North Western Himalayas. Maize Genomics Genet., 2015; 6(1): 1-5.
- Williams, T.R. and Hallauer, A. R. Genetic diversity among maize hybrids. Maydica, 2000; 45: 163-71.
- Kraja, A., Dudley, J.W and White, D.G. Identification of tropical and temperate maize populations having favorable alleles for disease resistance. Crop Sci., 2000; 40: 948-54.
- Hossain, M. Screening of maize varieties / lines against leaf blight disease. Bangladesh J. Agric., 1987; 12: 213-15.
- Sharma R.C. and Payak, M.M. Durable resistance to two leaf blights in two maize inbred lines. Theor. Appl. Genet., 1990; 80: 542-44.
© The Author(s) 2017. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


